Abstract
Background:
In December 2016, MACI (autologous cultured chondrocytes on porcine collagen membrane) received approval from the US Food and Drug Administration for the treatment of symptomatic articular cartilage defects of the knee with or without bone involvement in adults.
Purpose:
To describe the cartilage defects and patient characteristics for 1000 adult patients treated with MACI for knee cartilage repair in the United States.
Study Design:
Case series; Level of evidence, 4.
Methods:
Data collected by Vericel for adult patients treated for articular cartilage defects of the knee were reconciled and summarized. Data were collected for 1000 consecutive patients starting on July 1, 2017, when Carticel (the prior generation of autologous cultured chondrocytes) was no longer available. Patient names were removed for confidentiality, and patients were identified by MACI lot number and surgery date. Safety data were derived from the pharmacovigilance database. Patient demographics, cartilage defect characteristics, concomitant surgical procedures, and adverse events were summarized with descriptive statistics.
Results:
A total of 1000 adults and 1010 knee joints were implanted with MACI by 372 surgeons. The male (49.6%)-to-female (50.4%) ratio was evenly split, and the mean age was 34.0 years. The majority of patients (68.1%) had a single cartilage defect treated, and the mean treated defect size was 4.7 cm2. The mean total treated lesion size, including multiple defects, was 5.8 cm2. The patella was the most commonly treated joint surface (32.7%), followed by the medial femoral condyle (31.3%). Most patients (92.4%) had concomitant surgical procedures at the time of cartilage biopsy acquisition. The most common concomitant procedures at the time of biopsy procurement included cartilage debridement (83.7%) and meniscal resection (11.3%). The most common planned concomitant surgeries at the time of MACI implantation were anterior tibial tubercleplasty (7.8%) and reconstruction of dislocating patella (5.5%). Few patients (2.6%) had adverse events.
Conclusion:
Patient age and mean total MACI-treated defect size in the United States are similar to the findings of the pivotal European SUMMIT (Superiority of MACI Implant Versus Microfracture Treatment) trial and other studies from outside the United States. Treatment of multiple cartilage defects is more frequent in the United States than elsewhere.
Full-thickness articular cartilage defects of the knee do not heal spontaneously. They may cause pain and symptoms such as swelling, catching, and loss of knee function. Several cost-effective treatments for symptomatic cartilage defects are available, 3 including autologous cultured chondrocytes on porcine collagen membrane (MACI; Vericel Corporation).
In December 2016, MACI received approval from the US Food and Drug Administration (FDA) for the treatment of symptomatic articular cartilage defects of the knee with or without bone involvement in adults. No patients had been treated with this autologous cellularized scaffold product in the United States prior to this FDA approval. Of note, however, is that other types of matrix-associated chondrocytes have been in clinical development in the United States, and the prior generation of FDA-approved autologous cultured chondrocytes (Carticel; Vericel Corporation) had been used in conjunction with collagen membranes off-label. The FDA approval of MACI was supported by the results of the European SUMMIT (Superiority of MACI Implant Versus Microfracture Treatment) trial—a phase 3, two-year, prospective, multicenter, randomized, open-label, parallel-group study that enrolled a total of 144 patients who had at least 1 symptomatic Outerbridge grade III or IV focal cartilage defect on the medial femoral condyle, lateral femoral condyle, and/or trochlea. 7
MACI-treated study participants had a mean age of 34.8 years, and 63% were male. The majority of patients (82%) had single cartilage defects. The mean cartilage defect size was 4.9 cm2, and 75% of the treated defects were located on the medial femoral condyle. The treatment of symptomatic cartilage knee defects by MACI was better than treatment with microfracture, in terms of both clinical results and statistical significance. 7 All patients from the 2-year study had the option to enroll in a 3-year follow-up study (extension study), in which 128 patients participated. At 5 years after treatment, the improvement seen in MACI over microfracture with regard to the co-primary endpoint of the Knee injury and Osteoarthritis Outcome Score for pain and function was maintained and was clinically and statistically significant. 1
All clinical experience with this autologous cellularized scaffold product had been outside the United States until the first implant of MACI in the United States in January 2017. A literature report summarizing data from 2690 patients with planned MACI treatment from 9 European countries, Australia, and Singapore reported that the mean patient age was 33.7 years and 67% of patients were male. The majority of patients (81%) had single cartilage defects. The mean cartilage defect size was 5.6 cm2 in that report. 4 To evaluate whether clinical use of MACI in the United States differed from that in Europe or differed from that in clinical trial experience, the purpose of the current study was to summarize the patient demographics, cartilage lesion characteristics, concomitant surgical procedures, and adverse events for 1000 US MACI-treated patients after the discontinuation of Carticel (first-generation autologous chondrocyte implantation product).
Methods
Data were collected for 1000 consecutive patients starting on July 1, 2017, when Carticel (first-generation autologous chondrocyte implantation product) was no longer available. Patient names were removed for confidentiality, and patients were identified by MACI lot number and surgery date. Adverse events, collected as part of the FDA’s mandatory safety reporting requirements for licensed products, were derived from the pharmacovigilance database for these 1000 patients from July 1, 2017, to November 30, 2019.
MACI Manufacturing
Vericel provided a cartilage biopsy kit for acquisition of cartilage. All cartilage specimens sent to Vericel were accessioned; chondrocytes were cultured and cryopreserved until a MACI order was received. Information recorded on the biopsy transmittal notice was entered in a customer relationship management (CRM) database. Each biopsy was assigned a unique lot number. Upon MACI order receipt, MACI was manufactured and release-tested before shipment for surgery. Information from the MACI order form was entered into a CRM database.
MACI Treatment
All surgeons implanting MACI had undergone training in cartilage biopsy procurement, MACI implantation, and post-MACI rehabilitation before shipment of MACI. The cartilage defects were to be prepared and the MACI implanted as described in the MACI prescribing information. Vericel cell therapy specialists or their designees were generally present for MACI surgeries.
MACI Customer Relationship Management Databases
Adult patient demographics, concomitant biopsy procedures, and knee cartilage lesion characteristics (location, dimensions, bone involvement) were collected from the biopsy transmittal memo included with each cartilage biopsy. Patients with joints other than the knee implanted with MACI or pediatric patients (<17 years of age) were not included in this summary. Cartilage lesion information from the MACI order form and biopsy transmittal notice entered into CRM databases was merged, and potential discrepancies were resolved. Concomitant procedures planned at the time of MACI implantation in the CRM database were summarized based on Current Procedural Terminology codes.
Safety Data
Adverse events were coded using the Medical Dictionary for Regulatory Activities.
Data Analysis
Patient demographics, cartilage defect characteristics, concomitant surgical procedures, and adverse events were summarized with descriptive statistics through use of SAS (Version 9.4; SAS Institute). Figures were prepared by use of GraphPad Prism.
Results
Patient Demographics
A total of 1000 adults and 1010 knee joints were treated with MACI by 372 surgeons (Table 1). The male (49.6%)-to-female (50.4%) ratio of treated adult patients was evenly split, and the mean ± SD age was 34.0 ± 9.85 years (range, 17-61 years). The distribution of patient ages is shown in Figure 1; one-third of the treated patients were between 30 and 40 years of age. Of these 1000 patients, 10 had both knees treated with MACI. Further, 4 patients had both knees treated with MACI in 2 different surgeries, and 6 patients underwent MACI implantation in both knees on the same day.
Demographics and Cartilage Defect Characteristics

a N is the number of patients in the analysis. Patients may undergo more than 1 surgery. Age and sex are counted and summarized based on the number of patients.
b N1 is the number of surgeries. Joint to be treated, primary defect bony involvement, number of defects to be treated, primary defect size, and total defect size are counted and summarized based on the number of surgeries.
c Primary defect is defined as the larger/largest defect in the case of multiple defects.

Age distribution of patients treated with MACI (autologous cultured chondrocytes on porcine collagen membrane). Shown are the age categories for 1000 MACI-implanted adult patients age 17 years and older.
Cartilage Defect Characteristics
The majority of patients had a single articular cartilage defect (68.1%); the mean treated single defect size was 4.7 cm2. The distribution of MACI-implanted cartilage defect sizes is shown in Figure 2; approximately two-thirds (65.4%) of the MACI-implanted defects were between 2 and 6 cm2. A total of 43 (4.3%) of the 1010 surgeries were for the treatment of cartilage defects with bone involvement. The mean total treated lesion size, including multiple cartilage defects, was 5.8 cm2. The patella was the most commonly implanted joint surface (32.7%), followed by the medial femoral condyle (31.3%). Mean cartilage defect sizes by location are shown in Figure 3.
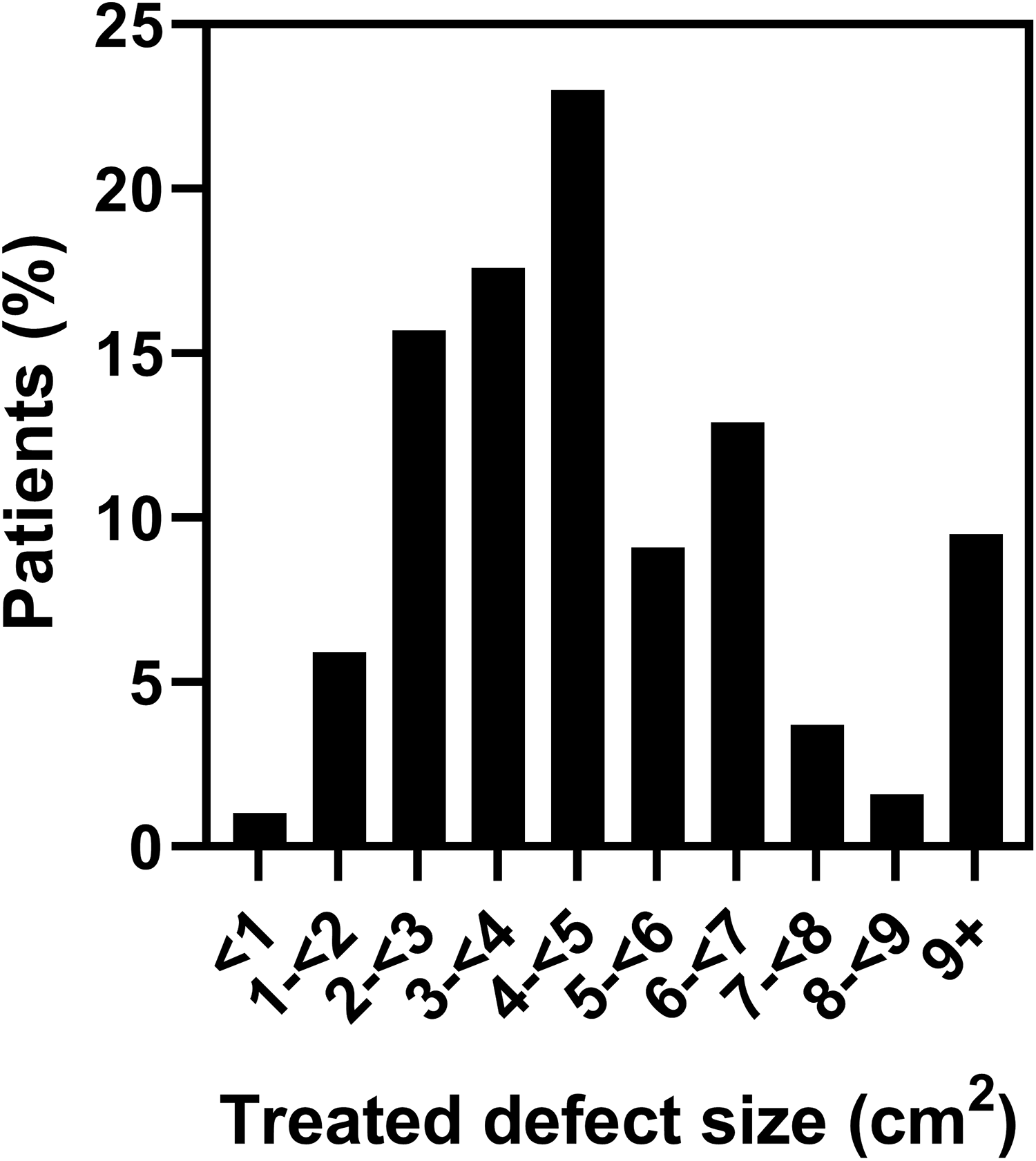
Size of cartilage defect treated with MACI (autologous cultured chondrocytes on porcine collagen membrane). Shown are the size categories for the larger/largest (if >1) MACI-implanted cartilage defect after debridement in 1010 MACI-implanted knees.

Cartilage defect size by defect location. Box and whisker plots of the mean (diamond), median (horizonal line through box), and 25th and 75th quartiles (lower and upper sides of each box) of the cartilage defect size (cm2) by defect location. The lower whisker defines the minimum observation, and the upper whisker defines the maximum defect sizes below the 1.5 interquartile range (IQR) of the 75th percentile. The open circles represent individual observations that are greater than 1.5 IQR above the 75th percentile. LFC, lateral femoral condyle; MFC, medial femoral condyle; Nobs, number of observations; Std, standard deviation.
The anatomic location of MACI-implanted defects for patients with 1 treated defect is summarized in Table 2. Defects were most commonly located in the patellofemoral joint in patients with 1 MACI-treated defect. Of the 326 patients with multiple MACI-implanted cartilage defects, the majority of patients (230/326; 70.6%) were treated in both the patellofemoral and tibiofemoral joints. A total of 73 of the 330 (22.1%) patients who received patellar treatment were treated with MACI on the patella and trochlea.
Defect Location in Surgeries Where a Single Defect Was Treated With MACI a

a Percentages are based on 688 surgeries that entailed a single defect. MACI, autologous cultured chondrocytes on porcine collagen membrane.
Concomitant Procedures
Almost all patients (92.4%) had concomitant surgical procedures at the time of cartilage biopsy acquisition (Table 3). The most common concomitant procedures at the time of biopsy procurement included debridement of the cartilage lesion (83.7%) and meniscal resection (11.3%).
Concomitant Surgeries at the Time of Cartilage Biopsy a

a The total sample was 1000 patients. Some patients had more than 1 procedure.
b Other procedures in >1 patient include “not specified” (n = 51), lateral release (n = 3), osteochondral autograft (n = 3), and tibial tubercle osteotomy (n = 2).
Based on insurance preauthorization information in the CRM database, 132 patients (13.2%) had planned concomitant procedures at the time of MACI implantation (Table 4). The most common planned concomitant surgery was an anterior tibial tubercleplasty (7.8%), followed by reconstruction of dislocating patella (5.5%).
Concomitant Surgeries Planned at the Time of MACI Implantation a

a The total sample was 1000 patients. MACI, autologous cultured chondrocytes on porcine collagen membrane. Some patients had more than 1 procedure.
Safety
Very few patients (26; 2.6%) experienced adverse events (AEs). The most commonly reported AE was arthralgia, reported in 8 (0.8%) patients (Appendix Table A1). No events were reported in more than 1% of patients. A total of 85 AEs were reported in 26 patients with a follow-up period up to 28 months.
MACI treatment failure was reported in 3 patients. A 26-year-old man experienced knee swelling, pain, and graft delamination within 2 months of MACI implantation of a 6-cm2 defect on the right lateral femoral condyle; the patient underwent a concomitant meniscal transplant at the time of MACI implantation. The graft delamination was confirmed arthroscopically. Additional follow-up information was not provided. A 30-year-old woman treated with MACI for 1-cm2 patellar and 0.25-cm2 lateral femoral condyle defects experienced treatment failure 14 months after implantation. At the time of biopsy collection 9 months before MACI implantation, the patient was treated with microfracture/marrow stimulation. Additional follow-up information was not provided. A 36-year-old woman treated with MACI for 3 left knee cartilage defects (total defect area, 10.5 cm2) of the left medial femoral condyle and trochlea experienced treatment failure on an unspecified date after MACI implantation. Prior knee surgeries included osteotomy, microfracture, and bone graft. Concomitant adverse events reported after MACI implantation and at the time of treatment failure included loneliness, depression, mental distress, and knee pain.
Discussion
This study summarizes initial MACI use in the United States and indicates that patient age and articular cartilage defect size are comparable between populations in the United States and elsewhere 4 as well as the pivotal European MACI SUMMIT study. 7 The SUMMIT study required a minimum cartilage defect size of 3 cm2; a majority of US MACI use (77%) was in the treatment of cartilage defects 3 cm2 or larger. For cartilage defects smaller than 2 cm2, autologous chondrocyte implantation is not typically considered a primary treatment option 2 ; however, a small percentage of patients (7%) had a primary cartilage defect smaller than 2 cm2 treated with MACI. This pattern is consistent with use outside the United States, where approximately 7% of treated cartilage defects were 2 cm2 or smaller. 4 Unlike a clinical trial, the current study did not collect patients' sports activity level, medical history, or prior surgeries; we speculate that these patients had failed prior more conservative treatment such as debridement or marrow stimulation.
Additionally, although the population sample sizes are quite different, the incidences of concomitant partial meniscectomy in the United States (11.3%) and the European SUMMIT study (11.1%) are similar. This is likely a result of clinical practice and clinical protocol (and therefore MACI prescribing information) recommending the need to address concomitant pathology in addition to MACI cartilage restoration. As would be expected, the rates of concomitant procedures were much higher (71%) in active US military servicemembers. 9
In contrast to all previously reported MACI use, both male and female patients were treated in equal numbers in the United States, which may be a result of expanded access to recreational sports activities for girls and women in the United States as well as the high percentage of patellar cases. Use of autologous chondrocyte implantation for the treatment of patellar defects was more common in female than male patients in a multicenter US case series. A striking difference in MACI use within versus outside the United States is the higher percentage of patients (31% vs 19%) with more than 1 treated cartilage defect. 4
These data represent our first opportunity to characterize MACI-treated patient and cartilage defect characteristics in the patella. (The summary by Foldager et al 4 from patients outside the United States did not describe defect locations.) As expected, the mean size of the treated defect is smallest for the patella compared with other articular cartilage surfaces and also smaller than the mean defect size reported by Gomoll et al 6 (5.4 cm2). Disease severity in that series 6 was apparently greater than observed in the present cohort, as noted by a higher rate of bipolar lesions (27%) and concomitant osteotomy (69%).
Given the SUMMIT study’s exclusion of malalignment requiring osteotomy to correct tibiofemoral or patellofemoral alignment, this real-world patient population requiring concomitant femoral or tibial osteotomy (n = 43 patients) provides the opportunity to study patient outcomes in this challenging patient population. 5 Many literature reports (42% of clinical studies) describing cartilage restoration procedures excluded patients with malalignment, meniscal injury, and ligamentous instability. 8
This early US experience saw no unexpected safety findings compared with the US prescribing information.
These data must be assessed within the context of the study limitations. This report would have been strengthened if it had included clinical outcomes including patient-reported outcome data. Given that the MACI treatments summarized in this report were conducted by more than 300 surgeons, outcomes data are being collected at only a subset of sites, especially patient-reported outcomes for patients treated with MACI in the patella. Unlike a clinical trial, the current study did not collect patients' sports activity level, medical history, or prior surgeries. Given that this information is derived from the database supporting insurance claims, there is the possibility of underreporting of cartilage defect characteristics that may face challenges for insurance preapproval. Additionally, overestimation of a defect size may have taken place to facilitate insurance preapproval. There is the possibility of underreporting or overreporting of concomitant procedures, because this report summarized the percentage of patients with planned concomitant procedures (13.2%) rather than the actual concomitant procedures that were performed at the time of MACI implantation. In the current study, unlike a clinical study, the postmarketing AE data were spontaneously reported to the manufacturer and may not reflect rates observed in clinical studies or the real world. Also, the safety summary would benefit from a longer safety follow-up period than the posttreatment period of less than 2 years for some patients.
Conclusion
Patient age and mean total MACI-treated articular cartilage defect size in the United States are similar to that of the pivotal European SUMMIT clinical trial and real-world experience outside the United States. This initial experience with MACI suggests that treatment of multiple cartilage defects is more frequent in the United States than elsewhere. It remains to be seen whether the clinical outcomes after MACI treatment in the United States is comparable with those seen outside the United States or in the clinical trial setting.
Footnotes
Acknowledgment
The authors acknowledge Vericel Corporation Company employees Brad Rubin for providing the data sets, Hongbo (Elaine) Li for data cleaning and clinical programming, and Fang Dong for clinical programming quality control.
Final revision submitted February 28, 2020; accepted March 12, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.L.C. has received honoraria and speaking fees from Vericel and hospitality payments from Anika Therapeutics. A.E.R. is an employee of Vericel and has stock/stock options in Vericel. D.C.F. has received research support from Zimmer Biomet; consulting fees from CDC Medical, Ceterix, DePuy/Medical Device Business Services, Linvatec, Smith & Nephew, and Zimmer Biomet; nonconsulting fees from Vericel; and honoraria from Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from The Ohio State University Biomedical Sciences Institutional Review Board (study No. 2019H0313).
APPENDIX
Adverse Events by MedDRA System Organ Class a

| MedDRA System Organ Class | Adverse Events, n (%) |
|---|---|
| Blood and lymphatic system disorders | 1 (0.1) |
| Bone marrow edema | 1 (0.1) |
| Gastrointestinal disorders | 1 (0.1) |
| Abdominal pain, lower | 1 (0.1) |
| General disorders and administration site conditions | 15 (1.5) |
| Asthenia | 1 (0.1) |
| Crepitations | 1 (0.1) |
| Discomfort | 1 (0.1) |
| Fatigue | 1 (0.1) |
| Feeling abnormal | 1 (0.1) |
| Gait inability | 1 (0.1) |
| General physical health deterioration | 1 (0.1) |
| Implant site pain | 1 (0.1) |
| Inflammation | 1 (0.1) |
| Malaise | 1 (0.1) |
| Pain | 4 (0.4) |
| Peripheral swelling | 1 (0.1) |
| Swelling | 1 (0.1) |
| Treatment failure | 3 (0.3) |
| Immune system disorders | 1 (0.1) |
| Autoimmune disorder | 1 (0.1) |
| Infections and infestations | 2 (0.2) |
| Cellulitis | 1 (0.1) |
| Folliculitis | 1 (0.1) |
| Diverticulitis | 1 (0.1) |
| Skin infection | 1 (0.1) |
| Injury, poisoning, and procedural complications | 11 (1.1) |
| Contusion | 1 (0.1) |
| Graft complication | 2 (0.2) |
| Graft delamination | 4 (0.4) |
| Graft overgrowth | 3 (0.3) |
| Joint dislocation | 1 (0.1) |
| Postprocedural swelling | 1 (0.1) |
| Procedural pain | 4 (0.4) |
| Investigations | 1 (0.1) |
| White blood cell count increased | 1 (0.1) |
| Musculoskeletal and connective tissue disorders | 18 (1.3) |
| Arthralgia | 8 (0.8) |
| Arthrofibrosis | 1 (0.1) |
| Arthropathy | 1 (0.1) |
| Cartilage hypertrophy | 1 (0.1) |
| Chondromalacia | 1 (0.1) |
| Joint effusion | 3 (0.3) |
| Joint instability | 1 (0.1) |
| Joint range of motion decreased | 1 (0.1) |
| Joint stiffness | 1 (0.1) |
| Joint swelling | 3 (0.3) |
| Knee deformity | 1 (0.1) |
| Muscle atrophy | 2 (0.2) |
| Nervous system disorders | 1 (0.1) |
| Sleep deficit | 1 (0.1) |
| Psychiatric disorders | 4 (0.4) |
| Depression | 1 (0.1) |
| Emotional distress | 1 (0.1) |
| Frustration tolerance decreased | 1 (0.1) |
| Insomnia | 1 (0.1) |
| Psychiatric symptom | 1 (0.1) |
| Stress | 1 (0.1) |
| Respiratory, thoracic, and mediastinal disorders | 1 (0.1) |
| Dyspnea | 1 (0.1) |
| Pulmonary embolism | 1 (0.1) |
| Skin and subcutaneous tissue disorders | 1 (0.1) |
| Urticaria | 1 (0.1) |
| Social circumstances | 2 (0.2) |
| Immobilization prolonged | 1 (0.1) |
| Loss of personal independence in daily activities | 1 (0.1) |
| Vascular disorders | 2 (0.2) |
| Deep vein thrombosis | 2 (0.2) |
a Number of patients with spontaneously reported adverse events = 26 out of 1000 (2.6%). Number of spontaneously reported adverse events = 85. MedDRA, Medical Dictionary for Regulatory Activities.
