Abstract
Background:
Despite the strong implications for rehabilitation design, the capability of individuals with anterior cruciate ligament reconstruction (ACLR) to adapt and store novel gait patterns have not been well studied.
Purpose:
To investigate how reconstructive surgery may affect the ability to adapt and store novel gait patterns in persons with ACLR while walking on a split-belt treadmill.
Study Design:
Controlled laboratory study.
Methods:
Gait adaptation was compared between 20 participants with ACLR and 20 healthy controls during split-belt treadmill walking. Gait adaptation was assessed in slow- and fast-adapting parameters by (1) the magnitude of symmetry during late adaptation and (2) the amount of the asymmetry during de-adaptation.
Results:
Healthy individuals adapted a new walking pattern and stored the new walking pattern equally in both the dominant and nondominant limbs. Conversely, individuals with ACLR displayed impairments in both slow-adapting and fast-adapting derived gait adaptation and significant differences in behavior between the reconstructed and uninjured limb.
Conclusion:
While surgical reconstruction and physical therapy are aimed at improving mechanical stability to the knee, the study data suggest that fundamental features of motor control remain altered. After ACLR, participants display an altered ability to learn and store functional gait patterns.
Surgical reconstruction followed by comprehensive physical therapy is the standard of care for individuals who want to return to sporting activities after sustaining anterior cruciate ligament (ACL) injuries. 28 However, even after surgical reconstruction, lengthy rehabilitation, and return to sport, alterations in gait and neuromuscular control appear to persist. 5,26 Notably, individuals who have undergone ACL reconstruction (ACLR) exhibit kinetic and kinematic abnormalities at the knee during gait in all planes of motion. These impairments have been shown to persist for up to 17 months after surgery, despite successful completion of rehabilitation programs. 20,26,27 These continued performance decrements and increased risks highlight the need to better understand the locomotor control consequences after ACLR to develop rehabilitation paradigms that better prevent these unwanted consequences.
Locomotor adaptation paradigms using split-belt treadmills (SBTs) have allowed researchers to study intra- and interlimb coordination in various clinical and healthy populations. 8,18,23 –25 During SBT walking, the 2 belts can be decoupled such that 1 leg walks faster than the other, resulting in both rapid and slow adjustments to the asymmetric walking pattern. 22 Fast responses, such as changes in stride length asymmetry and stance time asymmetry, occur as an immediate reaction to restore balance. Slow responses, such as changes in step length asymmetry and double-support time asymmetry, are motor actions that represent the ability of the nervous system to use sensory input to predict the effect of a disturbance and adjust the response accordingly. 3,22 Assessment of locomotor adaptive and storage capabilities may be useful for ascertaining whether individuals with ACLR have the capacity to generate a more normal/symmetric motor pattern. Moreover, despite the strong implications for rehabilitation design, the capability of individuals with ACLR to adapt and store adaptation patterns have not been well studied. 11,12 Thus, it is unknown whether people who have undergone ACLR are able to make adaptations to gait perturbations in a manner similar to healthy individuals.
The aim of this study was to provide insight regarding the ability of individuals with unilateral ACLR to adapt and store gait patterns. We hypothesized that (1) the magnitude of locomotor adaptation (as evinced by asymmetry scores) will differ depending on what belt the injured leg is on and (2) during SBT walking, the individuals who underwent ACLR would demonstrate a reduced amount of locomotor adaptation (lower asymmetry scores during early adaptation, higher asymmetry scores during late adaptation, and lower asymmetry scores during de-adaptation) compared with healthy controls. Specifically, we expected the reconstructed limb to exhibit different values of step length asymmetry, double-support time asymmetry, stride length asymmetry, and stance time asymmetry while walking with the belts decoupled.
Methods
Participants
Twenty participants with ACLR (12 females, 8 males; mean age, 20 ± 1 years; mean height, 1.71 ± 0.11 m; mean weight, 69 ± 14 kg; mean body mass index [BMI], 23 ± 3 kg/m2) and 20 healthy age- and sex-matched controls (12 females, 8 males; mean age, 20 ± 1 years; mean height, 1.70 ± 0.12 m; mean weight, 67 ± 14 kg; mean BMI, 23 ± 3 kg/m2) were recruited. Patients who underwent ACLR were injured during noncontact (n = 15) or contact (n = 5) events and while performing in various activities (7 soccer, 6 basketball, 2 football, 1 rugby, 1 softball, 1 cheerleading, 1 gymnastics, 1 lacrosse). Reconstruction was performed using autograft hamstring (n = 8), patellar tendon (n = 7), Achilles autograft (n = 4), or hybrid allograft/autograft (n = 1). This study was approved by the University of Florida institutional review board, and informed consent was obtained from each individual prior to their participation.
The groups were matched on age (±2 years), sex, and physical activity level. Both groups reported 6 ± 3 hours of physical activity per week, were free from pain, and had not previously walked on an SBT. The ACLR volunteers had sustained at least 1 ACL tear verified by a doctor, followed with surgical reconstruction and rehabilitation, and were cleared to return to sporting activities by their doctor and physical therapist. All participants had returned to activity; 17 of 20 returned to the same sport in which they had incurred the ACL rupture. Only 1 participant had undergone a second ACLR to the same limb. Participants were on average 36 ± 24 months postsurgery. Controls were free from any history of neurological impairment, with no experience of any lower extremity orthopaedic injury for at least 1 year prior. Furthermore, controls were free from any lower extremity orthopaedic injury that could have required surgery or physical therapy.
Experimental Protocol
Retroreflective markers were attached over bony landmarks in accordance with the Vicon Plug-in-Gait marker system. Kinematic data, time-synchronized to the kinetic data, were collected using an 8-camera motion capture system (120 Hz; Vicon). Kinetic data were collected as the participants walked on an instrumented SBT (1200 Hz; Bertec Corp). Participants first walked on the SBT while both belts moved together at a self-selected comfortable speed for 5 minutes to acclimate to walking on the treadmill (Figure 1). All participants were instructed to walk while holding the treadmill hand rails. Participants then walked for 2 minutes at the slow speed (0.75 m/s) and 2 minutes at the fast speed (1.5 m/s). Participants then walked (for a second time) at the slow speed to undo the effects of fast walking and establish a washout period (baseline). 23 Then, the belt under the nondominant leg in the controls (reconstructed leg in the individuals with ACLR) sped up to the fast speed. The belt under the contralateral leg remained at the slow speed. Participants walked under these split conditions for 13 minutes. This was considered the adaptation portion of the experiment (adapt 1). Next, participants walked with both belts at the slow speed for 5 minutes (de-adapt 1). This condition was recorded to assess the amount gait parameters are stored from the adaptation portion of SBT walking (“aftereffects”). The last 30-second period of de-adapt 1 was used for the second baseline for the following adaptation conditions. After the de-adapt condition, participants walked with the belts split but the speeds were opposite of adapt 1. That is, the belt under the dominant leg in controls (uninjured leg in individuals with ACLR) was set to the fast speed while the belt under the contralateral leg remained at the slow speed. Participants walked under these conditions for 13 minutes (adapt 2), and then walked with the belts both set at the slow speed for 5 minutes (de-adapt 2). The adapt 1 and adapt 2 conditions (early and late, respectively) were randomized between all participants.

Experimental protocol. “Fast” (light gray) indicates 1.5 m/s; “Slow” (dark gray) indicates 0.75 m/s walking speed.
Data Processing
All variables calculated for the leg on the fast belt are here referred to as the “fast” leg, and the leg on the slow belt referred to as the “slow” leg. Foot contacts and toe-offs were determined using marker velocity profiles and a 50-N force plate threshold. 19 Marker data were filtered using a fourth-order low-pass Butterworth filter with a cutoff frequency of 10 Hz.
Spatiotemporal Gait Parameters
Stride length was defined as the anterior-posterior distance traveled by the ankle marker from heel-strike to toe-off. 22 Stance time was defined as the percentage of the gait cycle between heel-strike and subsequent toe-off of the same limb. Step length was defined as the anterior-posterior distance between the ankle markers at heel-strike. Fast step length refers to the step length calculated at the heel strike of the fast leg, and slow step length to that calculated at the heel strike of the slow leg. Slow double-limb support refers to the time from fast leg heel-strike to slow leg toe-off, and fast double-limb support refers to the time from slow leg heel-strike to fast leg toe-off.
Asymmetry was defined in each spatiotemporal gait parameter using the following asymmetry index 25 :
Asymmetry data were averaged over the first and last 5 strides of the early and late conditions. “Usual” asymmetry values were determined by averaging data across 30 seconds of slow walking after the acclimation, as well as both baseline conditions. The mean usual asymmetry value was subtracted from all subsequent analysis.
Statistical Analyses
Locomotor Adaptation
Three separate, repeated-measures analyses of variance (ANOVAs) with Bonferroni correction for pairwise comparisons were performed to analyze differences in the spatiotemporal gait parameters for each of 3 pairs of conditions: (1) A 3 × 2 (condition × limb speed) repeated-measures ANOVA was performed to analyze spatiotemporal gait variables (asymmetry scores) among conditions involved in adaptation (baseline, early, late) and between ACLR limbs, when the ACLR limb was on the fast belt compared with when it was on the slow belt (ACLR fast, ACLR slow); (2) a 3 × 2 (condition × group) ANOVA was performed to analyze spatiotemporal gait variables among conditions involved in adaptation (baseline, early, late) and between groups, when the ACLR leg was on the fast belt versus when the control nondominant leg was on the fast belt (ACLR fast, control nondominant fast); (3) a 3 × 2 (condition × group) ANOVA was performed to analyze spatiotemporal gait variables among conditions involved in adaptation (baseline, early, late) and between groups, when the ACLR leg was on the slow belt versus when the control nondominant leg was on the slow belt (ACLR uninjured slow, control dominant slow). Level of significance for all analyses was set at α < .05.
Storage of Adaptation Pattern
Repeated-measures 2 × 2 (condition × limb speed) ANOVAs with Bonferroni correction for pairwise comparisons were performed to analyze differences in the spatiotemporal gait parameters (asymmetry scores) during de-adapt 1 and de-adapt 2 immediately followed by SBT walking for each of 3 pairs of comparisons: (1) between limbs (ACLR fast, ACLR slow), (2) between groups (ACLR fast, control fast), and (3) between groups (ACLR slow, control slow). Level of significance for all analyses was set at α < .05.
Results
Participant demographics were not statistically different. Significant main effects for the repeated-measures comparisons for step length asymmetry, double-support time asymmetry, stride length asymmetry, and stance time asymmetry are displayed in Tables 1 and 2. Both participant groups in this study were able to respond to the SBT stimulus as many gait parameters followed similar trends as those previously observed during SBT walking in healthy young and old adults and also in patient populations such as poststroke and Parkinson disease (Figures 2 –4). 4,18,23 –25 Specific follow-up tests for significant main effects are discussed below. For the sake of clarity, we have only reported limb × condition or group × condition differences.
Summary of Repeated-Measures ANOVA Main Effects for Asymmetry Scores to Assess Adaptation a

a Data are presented as F value (P value). Boldfaced P values indicate statistical significance. ACLR, anterior cruciate ligament reconstruction; ANOVA, analysis of variance.
Summary of Repeated-Measures ANOVA Main Effects for Asymmetry Scores to Assess Storage a

a Data are presented as F value (P value). Boldfaced P values indicate statistical significance. ACLR, anterior cruciate ligament reconstruction; ANOVA, analysis of variance.

Mean asymmetry scores during all walking conditions in individuals who underwent anterior cruciate ligament reconstruction (ACLR). Error bars indicate standard error. *P < .05 for differences between limbs for the early, late, and de-adapt conditions. Indications of significant effects of leg and walking condition are omitted for clarity.

Mean asymmetry scores during all walking conditions between groups on the fast belt. Error bars indicate standard error. *P < .05 for difference between groups for the late and de-adapt conditions. Indications of significant effects of group and walking condition are omitted for clarity. ACLR, anterior cruciate ligament reconstruction; HYA, control group.
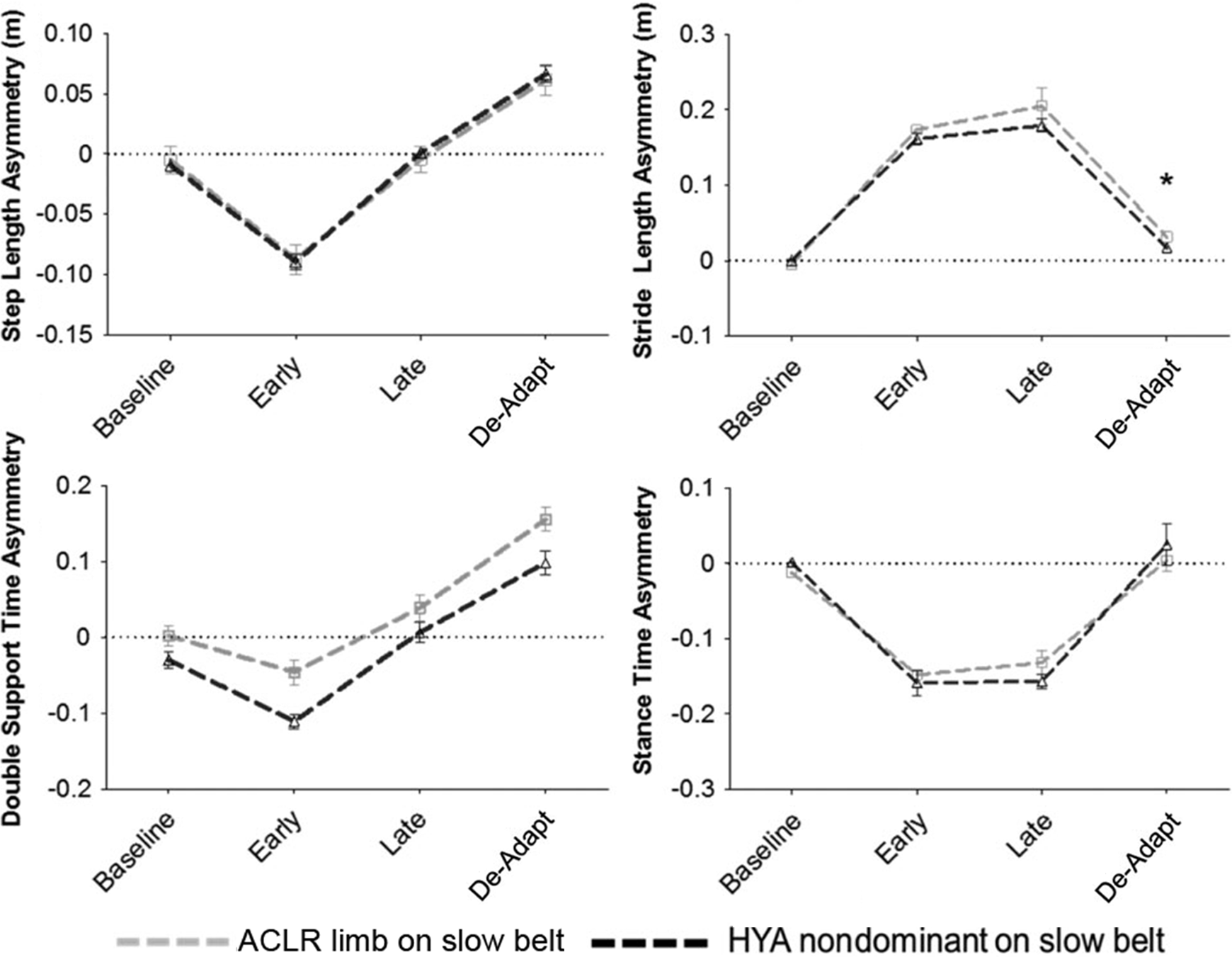
Mean asymmetry scores during all walking conditions between groups on the slow belt. Error bars indicate standard error. *P < .05 difference between groups for the de-adapt condition. Indications of significant effects of group and walking condition are omitted for clarity. ACLR, anterior cruciate ligament reconstruction; HYA, control group.
Locomotor Adaptation
ACLR Limb on Fast Belt Versus ACLR Limb on Slow Belt
Slow-Adapting Parameters
When comparing between limbs of ACLR individuals across conditions, double-support time asymmetry was significantly different between limbs during early (P = .001) and late (P = .011) adaptation (Figure 2).
Fast-Adapting Parameters
Stride length asymmetry between limbs of those with ACLR was greatest during late adaptation compared with baseline and early adaptation (P < .001 and P = .019, respectively).
ACLR Limb on Fast Belt Versus Control Nondominant Limb on Fast Belt
Slow-Adapting Parameters
Step length was significantly more asymmetric in the ACLR leg compared with the control nondominant limb during early adaptation compared with baseline (P < .001) and late adaptation (P < .001) (Figure 3).
Follow-up analyses revealed that double-support time for both the ACLR leg and the control nondominant limb was significantly more asymmetric during early adaptation compared with baseline (P < .001) and late adaptation (P < .001).
Fast-Adapting Parameters
During late adaptation, stride length asymmetry was significantly greater in ACLR compared with control limbs (P = .03) (Figure 3).
ACLR Limb on Slow Belt Versus Control Nondominant Limb on Slow Belt
Slow-Adapting Parameters
No differences between ACLR patients and controls were observed in step length or double-support time asymmetry when the ACLR limb was on the slow belt (Figure 4).
Fast-Adapting Parameters
No differences between ACLR patients and controls were observed in stride length or stance time asymmetry when the ACLR limb was on the slow belt.
Locomotor Storage
ACLR Limb on Fast Belt Versus ACLR Limb on Slow Belt
Slow-Adapting Parameters
For both the reconstructed and uninjured leg, asymmetry scores for double-support time were significantly more positive during the de-adaptation period (both P < .001), suggesting a different walking pattern was observed.
Fast-Adapting Parameters
For both the reconstructed and uninjured leg, stance time asymmetry was not significantly different between de-adaptation and baseline conditions (Figure 2).
ACLR Limb on Fast Belt Versus Control Nondominant Limb on Fast Belt
Slow-Adapting Parameters
Step length asymmetry was significantly more positive in the control group compared with the reconstructed limb of ACLR patients (P = .027). However, no group × condition differences were observed (Figure 3).
The ACLR leg and the control nondominant leg were not statistically different at baseline, but double-support time asymmetry was significantly greater in the control nondominant limb during de-adaptation (P = .009).
Fast-Adapting Parameters
Stride length in the ACLR leg and the control nondominant leg were not statistically different at baseline (P = .054), but stride length asymmetry was significantly greater in the ACLR limb during de-adaptation (P < .001) (Figure 3).
ACLR Limb on Slow Belt Versus Control Nondominant Limb on Slow Belt
Slow-Adapting Parameters
No differences between ACLR patients and controls were observed in step length or double-support time asymmetry when the ACLR limb was on the slow belt (Figure 4).
Fast-Adapting Parameters
Stride length for the ACLR uninjured leg and the control dominant limb were not statistically different at baseline (P = .761), but stride length asymmetry was significantly greater in the ACLR uninjured leg during de-adaptation (P < .001).
Discussion
We investigated the ability of individuals who underwent ACLR to acquire new locomotor behaviors to gain insight into the observed locomotor disturbances seen after surgery and rehabilitation. Healthy individuals adapted and stored the new walking pattern equally in both the dominant and nondominant limbs, regardless of what leg was placed on the fast or slow belt. Individuals who underwent ACLR displayed some impairments in both slow-adapting and fast-adapting measures of locomotor adaptation depending on the limb/belt speed configuration.
By late adaptation, individuals with ACLR walked such that the reconstructed leg spent less time in double-limb support compared with the uninjured leg, regardless of which leg was on the fast belt (see Figure 2). Bilateral differences in timings of gait cycle events have been previously observed in people with ACLR 10 and could reflect a strategy to reduce the duration of the single-limb support on the ACLR leg. ACLR individuals appeared to reach a limit in double-support time that they were capable of employing during the SBT walking task. Indeed, double-support time was not largely perturbed during early adaptation in the uninjured leg when it walked on the fast belt. During the de-adaptation period, the ACLR group was capable of adjusting this parameter such that the ACLR leg spent an increased amount of time in double-support (the ACLR leg spent more time as the trailing leg, more time in terminal stance, and a reduced amount of time during loading when it was the lead leg). In summary, the differences in double-support time are possibly related to the sensory alterations (emerging from the reconstructed knee corrected neither by surgery nor by therapy) that form slow-adapting responses to the repeated perturbations.
ACLR attempts to re-establish the mechanical stability of the knee, and through rehabilitation, the goal is to restore gross movement patterns. A complete restoration of sensory function of the reconstructed ACL would necessitate re-innervation with free nerve endings and mechanoreceptors and re-establishment of the ACLR reflex (hamstring activation after tibial translation). Previous findings from a rat model may indicate that there is potential for this to happen. 2 Also, mechanoreceptors have been identified in reconstructed ACLs removed from human knees approximately 10 years after reconstruction surgery. 6 In spite of these suggestive findings, the disruption of sensory information from the intact ACLs and concomitant reorganization appears to compromise the neuromuscular control of both posture and gait despite reconstruction and completion of physical therapy. 1,7,21 However, while incomplete restoration of the neurosensory feedback is one explanation, it is also possible that other conditions such as persistent muscle weakness, joint laxity, pain, flexion contractures, and early cartilage degeneration may also affect the neuromuscular control of gait adaptation. Below, we discuss potential contributing mechanisms to explain these findings.
Intralimb measures such as stride length and stance time are known to adjust quickly during SBT walking. These changes occur immediately to maintain balance to walk on the decoupled belts and are thought to be controlled at the spinal level. 4,18 This finding supports that spinal structures controlling gait may be affected by the altered sensory feedback associated with the reconstructed knee. Krogsgaard et al 16 reported that re-innervation of the reconstructed ligament is unlikely and that afferent information from the mechanoreceptors in the reconstructed ACL are absent. The role of this afferent input is believed to be reconstructed in the constant update of the motor programs, which are designed to control muscle coordination during motion. Thus, the lack of afferent information or, at best, altered afferent information resulting from compensatory structures likely contributes to the impaired fast-adapting driven locomotor adaptive patterns.
Individuals with ACLR displayed a reduced amount of double-support time adaptation and also storage of double-support time. During de-adaptation, step length was less prominently stored in persons with ACLR compared with the healthy controls. These results support that deficits in individuals with ACLR could impair the ability of the neuromuscular system to update predictions how the body will move in response to changes in the demands of the locomotor task. 3,14 This suggestion is reinforced by findings from Madhavan and Shields, 17 who reported greater overshoot error, knee velocity, and altered muscle activity during a dynamic single-leg weightbearing task in individuals with ACLR even 4 years postsurgery. Moreover, Ferber et al 9 determined that individuals with ACLR display an altered movement strategy on experiencing an unexpected forward perturbation at heel strike. The authors suggested that the knee injury and subsequent reconstruction lead to neuromuscular reprogramming of coordinative movements. Motor learning studies have indicated that temporary inactivation of the primary motor cortex can disrupt consolidation of a learned motor pattern. 15 Indeed, faulty sensory feedback could also be a factor influencing the excitability of this structure. Altered activation of the motor cortex after an ACL injury that persists even after reconstruction could impair the storage of the adaptation pattern (aftereffects). Taken together, these results seem important given the role of sensory information in predictive control regulated by higher centers such as the cerebellum 3 and motor cortex. 15 Though the current study did not investigate the neurophysiological and behavioral relationship of adaptation, future research should investigate the neurophysiological mechanisms that may drive such consolidation of motor patterns in persons with ACLR.
The scope of this study was limited by the lack of information on adaptive learning (which refers to whether individuals can make use of previously acquired motor memory to predict new demands), as this study did not investigate a second exposure to the perturbation after washout of the first, where the belts move in the same configuration as they did during the first exposure. Although measurement of the de-adaptation portion during the first 5 strides of washout provides information of storage of aftereffects, we are unable to comment further on storage of the walking pattern savings for adaptive learning, as mechanisms and structures reconstructed in aftereffect storage could be independent from the savings storage. 15 More information on the ability to store savings and re-adapt to a practiced locomotor pattern would help to establish a greater understanding in persons with ACLR. However, investigating the initial exposure to a novel perturbation provides valuable insight into the neural control of movement and the ability of an individual to alter motor behavior in response to changing environmental constraints. Additionally, the current investigation did not set strict “time after reconstruction” or “graft type” limitations. Yet, an investigation of individual results suggests neither of these criterion appear to influence locomotor adaptation (unpublished observation). Furthermore, previous research has observed that the choice of graft type has minimal influence on the prevalence of osteoarthritis up to 10 years after surgery. 13 These findings may also be somewhat limited by the variety of grafts, diversity of surgeons and rehabilitation programs, lack of functional testing and laxity examinations, as well as unknown meniscal and cartilage problems and unknown presence of early cartilage degeneration in ACLR participants. Further studies that consider these factors will need to be executed. Studying patients with ACL deficiency before and after reconstruction as well as before and after rehabilitation could be valuable to understand the effects of surgery and rehabilitation on locomotor adaptation. In addition, studies that investigate SBT adaptation and how these results correlate with functional tests such as the single-leg hop, triple hop, and so on, would be important to shed light on physical therapy outcomes for return-to-sport criteria.
Conclusion
Both fast-adapting mediated and slow-adapting changes of gait parameters are altered in persons with ACLR during SBT walking. This investigation supports that even after surgical reconstruction and rehabilitation, fundamental features of motor control remain altered. Future studies are needed to investigate whether these changes in locomotor adaptation are affected by the initial injury to the ACL and whether there are additive effects from surgery and physical therapy.
Footnotes
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution.

