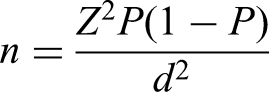Abstract
Background
Low Vitamin D levels are prevalent among people living with HIV (PLWH) on antiretroviral therapy (ART) and are associated with dyslipidemia and cardiovascular risk. This study examined the association between atherogenic indices and low vitamin D levels among PLWH in Southwestern Uganda.
Methods
We conducted a cross-sectional study among 218 randomly selected PLWH who had been on ART for ≥6 months at Mbarara City Health Centre IV, Southwestern Uganda from August 13, 2024 to August 23, 2024. Low serum Vitamin D level was defined as serum 25-hydroxyvitamin D concentration <30 ng/mL.
Results
Atherogenic combined index (ACI), high-sensitivity C-reactive protein (hs-CRP)/HDL-C ratio, and total cholesterol to high density lipoprotein cholesterol – body mass index (TC/HDL-C-BMI) index were independently associated with low vitamin D. Compared to the first tertile, ACI second and third tertiles had higher likelihood of low vitamin D (aPR = 2.23, 95% confidence interval (CI): 1.28-3.89, P = 0.005; aPR = 2.53, 95% CI: 1.05-6.05, P = 0.023). The high-sensitivity C-reactive protein to high-density lipoprotein cholesterol (hs-CRP/HDL-C) ratio third tertile was also significant (aPR = 1.44, 95% CI: 1.05-1.96, P = 0.024). TC/HDL-C-BMI index third tertile showed nearly twice the prevalence (aPR = 1.95, 95% CI: 1.10-3.45, P = 0.023).
Conclusion
ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index are potential predictors for Low serum Vitamin D levels among PLWH.
Plain Language Summary Title
Association between Blood Fat Measures and Inflammation with Low Vitamin D in People Living With HIV in Southwestern Uganda
This study looked at vitamin D levels in adults living with HIV who are receiving antiretroviral treatment in Southwestern Uganda. Vitamin D is important for strong bones, immunity, and overall health. We found that more than half of the participants had low vitamin D levels. We also discovered that certain blood measurements especially those showing inflammation and unhealthy cholesterol patterns were linked to a higher chance of having low vitamin D. These included the atherogenic combined index, the ratio of C-reactive protein to HDL cholesterol, and the total cholesterol to HDL cholesterol ratio combined with body mass index. People with higher values of these markers were more likely to have low vitamin D. These findings suggest that simple blood tests already used in HIV care may help identify patients at risk of vitamin D deficiency, allowing earlier screening, counseling, and treatment.
Introduction
Vitamin D deficiency (VDD) is defined as a serum 25-hydroxyvitamin D [25(OH)D] concentration below 20 ng/mL, while vitamin D insufficiency (VDI) refers to levels ranging from 20 to29 ng/mL. 1 Vitamin D is essential for calcium and phosphate homeostasis, immune regulation, and inflammation control. 2 Among people living with HIV (PLWH), both VDD and VDI are increasingly recognized as prevalent and clinically significant micronutrient deficiencies. 3 Several factors contribute to the high risk of VDD in PLWH, including HIV-associated immune dysregulation, persistent inflammation, limited sun exposure, poor nutritional status, 4 and certain antiretroviral therapies (ART), particularly efavirenz and protease inhibitors, which interfere with vitamin D metabolism.5,6 Additionally, HIV infection may compromise renal and hepatic hydroxylation of vitamin D, further reducing its bioavailability. 7 The clinical significance of VDD in this population is evident from its associations with increased susceptibility to secondary infections, reduced bone mineral density, muscle weakness, cardiovascular complications, and immune reconstitution inflammatory syndrome.2,8
The global burden of VDD among PLWH is considerable. Estimates suggest that 25(OH)D deficiency affects between 70.3% and 83.7% of this population. 9 In sub-Saharan Africa (SSA), the region with the highest HIV prevalence, 10 VDD remains paradoxically widespread despite abundant year-round sunlight. 11 A systematic review and meta-analysis involving 23 African countries reported that 18% of individuals had serum 25(OH)D levels below 30 nmol/L, 34% below 50 nmol/L, and nearly 60% below 75 nmol/L, highlighting widespread deficiency or insufficiency across the continent. 10 In Ethiopia, 75.4% of PLWH were found to have hypovitaminosis D, with 6.4% classified as deficient and 69% as insufficient. 12 Similarly, in East Africa, a study from Aga Khan University Hospital in Nairobi reported a 39% prevalence of VDD among PLWH. 13 Alarmingly high rates have also been reported in infants, with prevalence reaching 34.6% in Tanzania and exceeding 70% in Kenya.4,14,15 In Uganda, limited but emerging evidence indicates that VDD is common among PLWH, with one study in Central Uganda identifying deficiency in over 17% of ART-treated patients at the Infectious Diseases Institute. 16 However, data on vitamin D status in PLWH residing in Southwestern Uganda remain scarce, despite the potential influence of unique environmental, dietary, and HIV-related factors in this region.
Despite the recognized clinical relevance of vitamin D, its relationship with metabolic abnormalities particularly atherogenic indices among PLWH remains poorly understood in SSA. Evidence suggests that VDI is linked to decreased arterial compliance 17 and increased carotid intima media thickness, an established surrogate marker for atherosclerosis in this population. 18 Composite lipid ratios and indices such as high-sensitivity C-reactive protein (hs-CRP)/HDL-C, atherogenic combined index (ACI), residual cholesterol (RC), total cholesterol/HDL-C, LDL-C/HDL-C, Non-HDL-C/HDL-C ratio, and logTG/HDL-C have been shown to predict cardiometabolic risk more accurately than individual lipid measures. 19 The rationale for using these indices lies in their ability to integrate lipid, inflammatory, and anthropometric parameters that reflect the underlying pathophysiological mechanisms linking VDD to cardiometabolic risk. The high-sensitivity C-reactive protein to high-density lipoprotein cholesterol (hs-CRP/HDL-C) ratio captures the interaction between systemic inflammation and protective lipid fractions, both of which are dysregulated in VDD and chronic HIV infection. The ACI and total cholesterol to high density lipoprotein cholesterol – body mass index (TC/HDL-C-BMI) index incorporate traditional lipid markers alongside body composition, providing a more comprehensive assessment of atherogenic burden in populations experiencing ART-related metabolic alterations. These indices are particularly valuable in resource-limited settings, as they are derived from routinely measured clinical and laboratory parameters, making them practical and cost-effective surrogate markers for identifying individuals at risk of low vitamin D levels and associated cardiometabolic complications.
However, there is a paucity of research exploring their association with vitamin D status among PLWH in African settings, particularly in Uganda. Most existing studies are based on Western cohorts and do not account for the unique metabolic and epidemiological context of the African HIV population. To address this knowledge gap, our study assessed the potential of specific atherogenic indices as biomarkers for low vitamin D levels among PLWH receiving ART at a primary healthcare facility in Southwestern Uganda.
Material and Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for observational studies. 20
Study Design, Population, and Eligibility Criteria
We conducted a hospital-based descriptive and analytical cross-sectional study from August 13, 2024 to August 23, 2024. This study was carried out among PLWH attending Mbarara City Health Centre IV (MMC H/CIV); a government health facility which is located in Kamukuzi division, Mbarara City. It has a functional AntiRetroviral Therapy clinic, with approximately 5000 active ART patients. The study population was of PLWH who are currently receiving ART. Participants were selected using a simple random sampling technique from patients attending the ART clinic on each clinic day. On each day of data collection, the ART clinic attendance register was used as the sampling frame. Eligible patients scheduled for clinic review on that day were first screened against the inclusion and exclusion criteria. Each eligible patient was then assigned a unique identification number. These identification numbers were entered into a random number generating method, and the required number of participants for that clinic day was randomly selected. Patients whose identification numbers were selected were approached consecutively and invited to participate in the study. PLWH aged 18 years and above, who had been on ART for at least 6 months, who provided informed written consent, and who agreed to provide blood samples for biochemical analysis were included. Critically ill patients and those already on Vitamin D supplementation were excluded. Participant confidentiality was maintained through coded identifiers, and clinical feedback was provided to attending physicians for participants found to have VDD. Participants who met the study inclusion criteria were enrolled on all ART clinic days throughout the data collection period. Each day, the principal investigator and trained research assistants approached eligible patients and explained the objectives of the study. Patients who agreed to participate provided written informed consent prior to being interviewed using a structured questionnaire. Following the interview, body weight and height were taken, and participants were requested to provide a blood sample for laboratory measurement of serum vitamin D, hs-CRP, and lipid profile parameters.
Sample Size Determination
The Kish and Leslie sample size formula of 1965
21
was used, using 17%, a prevalence that was obtained by a study that was carried out among HIV patients at Infectious Disease Institute.
16
Using P of 0.17, a confidence interval (CI) of 95% which gives a Z value of 1.96 and a level of precision of +/−5% (d of 0.05), below is the estimated sample size, n.
Therefore, this study required a minimum of 217 participants.
Study Variables and Data Collection Tools
Dependent Variable
Low vitamin D levels which included both VDI and VDD was considered the primary outcome variable. VDD was defined as serum 25-hydroxyvitamin D concentrations <20 ng/mL while VDI was defined as serum 25-hydroxyvitamin D concentrations 20-30 ng/mL.22,23 About 5 mL of venous blood was collected from each study participant into a red-top vacutainer, centrifuged within an hour of collection at 3000 rpm for 10 min to obtain serum which was stored in two cryovials one at 6 °C (for measurement of serum lipids and hs-CRP at the end of each day of data collection) and the other at −20 °C (for measurement of Serum 25-hydroxyvitamin D concentrations at the end of data collection). The serum samples in the cryovials stored at −20 °C were shipped in ice-cold cool box to Masaka Regional Referral Laboratory where analysis of Vitamin D levels was conducted. Serum 25-hydroxyvitamin D concentrations were measured using an electrochemiluminescence immunoassay method on the well calibrated and quality-controlled Cobas e411 analyzer (Roche Diagnostics, Germany), following strict adherence to manufacturer instructions. Serum analysis for concentrations of hs-CRP and lipid parameters total cholesterol, LDL-cholesterol, HDL-cholesterol, and triglycerides was conducted on the well calibrated and quality-controlled Cobas C III chemistry analyzer (Roche Diagnostics, Germany) following standard protocols at Mbarara City Health CIV Laboratory.
Independent Variables
The major independent variables were CRP to HDL-cholesterol ratio, ACI, lipid combination index (LCI), Atherogenic Index of Plasma (AIP), Castelli's Risk Index (CRI-I), Castelli's Risk Index (CRI-II), Atherogenic Coefficient (AC), RC, non-HDL-c, TC/HDL-BMI index, and TG/HDL-BMI index that were calculated as follows:
hs-CRP/HDL-C ratio = hs-CRP(mg/L)/[HDL-C(mmol/L)]*386.65
24
ACI = log10 [TG (mg/dL) × non-HDL-c (mg/dL)/HDL-c (mg/dL)]
25
Non-HDL-c = TC (mg/dL) – HDL-c (mg/dL)
28
Castelli's Risk Index (CRI-I) = TC/HDL-C
29
Castelli's Risk Index (CRI-II) = LDL-C/HDL-C
29
Atherogenic Coefficient (AC) = Non-HDL-C)/HDL-C
30
Residual cholesterol (RC) = TC (mg/dL) – HDL-c (mg/dL) – LDL-c (mg/dL)
31
LCI = TC (mg/dL) × TG (mg/dL) × LDL-c/HDL-c (mg/dL)
25
body mass index (BMI) = weight(Kg)/[height(m)]^2 TC/HDL-BMI index = TC/HDL ratio * BMI
32
TG/HDL-BMI index = TG/HDL ratio * BMI
The indices were categorized based on their tertile distributions (Table 1). Participants were classified into three groups according to these tertiles, with the lowest tertile serving as the reference category.
Sociodemographic data (age, gender, marital status, education, employment), lifestyle (diet, smoking, alcohol, sun exposure, physical activity), anthropometric data (weight, height for BMI), and HIV-related clinical data (ART duration, ART regimen, viral load) were collected through a researcher-administered structured questionnaire and medical record abstraction. The weight was measured using a calibrated Secca weighing scale while height was taken using a stadiometer. BMI was calculated by dividing weight (kg) by height squared (m2) and classified as classified as <25 kg/m2, 25- 29.9 kg/m2, and ≥30 kg/m2. 33
Statistical Analysis
Data was analyzed using STATA software version 17. Continuous variables were assessed for normality using the Shapiro-Wilk test. Variables were not normally distributed (P-value <0.05), and therefore they were summarized using medians (interquartile range, IQR). The distribution of the continuous variables between participants with and those without low vitamin D levels was compared using Mann-Whitney U test. Categorical variables were summarized as frequencies and percentages and their distribution by serum Vitamin D level status compared using Chi-square or Fisher's exact tests.
To assess associations between independent variables and the binary outcome variable; low vitamin D levels(1 = Low, 0 = Normal), a generalized linear model regression of the Poisson family with a log link (modified Poisson) using robust standard errors was performed since the prevalence of low vitamin D levels was greater than 10%. 34 Low vitamin D levels were defined as serum 25-hydroxyvitamin D concentrations <30 ng/mL and included both VDI (serum 25-hydroxyvitamin D concentrations 20-30 ng/mL) and VDD (serum 25-hydroxyvitamin D concentrations <20 ng/mL). This cut-off was selected in accordance with widely used international clinical and epidemiological guidelines,22,23 which recognize serum 25(OH)D levels below 30 ng/mL as indicative of suboptimal vitamin D status associated with adverse skeletal, immunological, and cardiometabolic outcomes. However, the main independent variables, the atherogenic indices, were categorized into tertiles (Table 1) to allow assessment of dose-response relationships and to minimize the influence of extreme values, given the absence of universally accepted clinical cut-offs for these composite indices in PLWH, particularly in African populations. The independent variables that were statistically significant at P-value ≤0.2 at bivariate analysis and those with biological plausibility were included in the multivariate regression analysis to adjust for possible confounding. Associations were reported as prevalence ratios (PRs) with 95% CIs, and statistical significance was set at P < 0.05. To adjust for accuracy and precision, some variables that were not statistically significant in the multivariate model (marital status, viral load, duration on ART, alcohol consumption) and those that were highly correlated with other variables (Castelli's Risk Index (CRI – I), TG/HDL-cholesterol ratio, TG/HDL-C-BMI index, BMI) were dropped from the model without significant loss in validity of the model. The final chosen multivariate model which included ACI, high-sensitivity CRP/HDL-cholesterol ratio, TC/HDL-C-BMI index, AIP, AC, CRI – II, LCI, RC, age, sex, and education level was tested for presence of severe multicollinearity using the mean variance inflation factor (VIF) and a VIF < 5 was considered acceptable. The final model was also tested for its suitability to predict the outcome variable: VDD using Hosmer-emeshow goodness of fit test, and a P-value >0.05 indicates good goodness of fit.
Results
Study Flow Chart
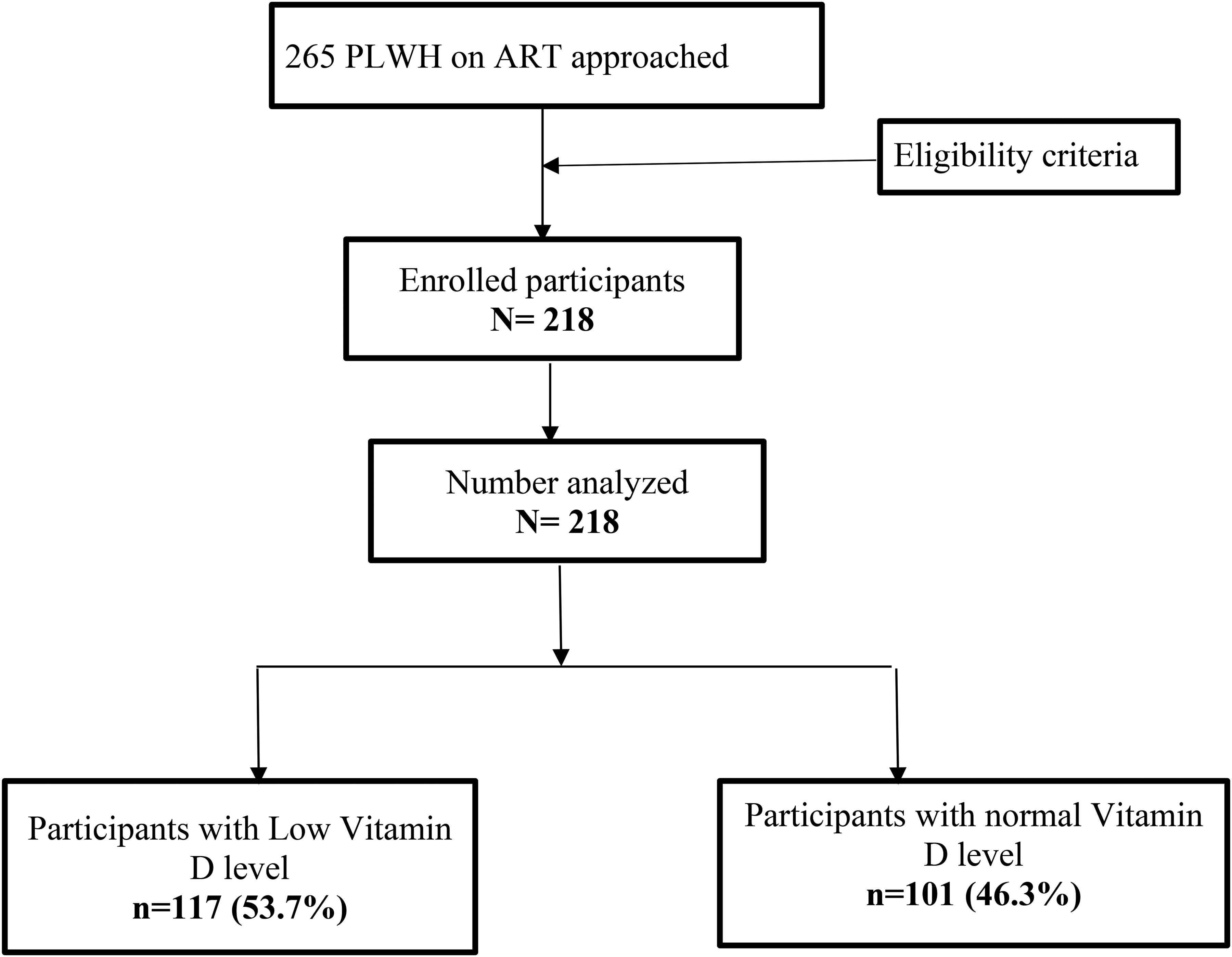
Narrative of the Study Flow Chart
A total of 265 PLWH on ART were initially approached for participation. After applying the eligibility criteria, 218 participants were enrolled into the study. All 218 were analyzed. Among these, 117 participants (53.7%) were found to have low vitamin D levels, while 101 participants (46.3%) had normal vitamin D levels.
Sociodemographic and Clinical Characteristics of the Study Participants
A total of 218 participants were included in this study, with a median age of 38 years (IQR: 30-45). The majority were female (74.8%) and married (64.7%). Female gender was common among participants with low vitamin D levels compared to the group with normal vitamin D levels (82.1% vs 66.3%, P = 0.008). Nearly half (49.1%) had attained primary education, while 35.8% had secondary education or higher, and 15.1% reported no formal education. Most participants (72.0%) were employed, with 52.8% earning less than 100,000 Ugandan shillings per month. A large proportion (95.9%) were non-smokers, and 75.7% reported never consuming alcohol. Median BMI was 24.8 kg/m2 (IQR: 22.0-29.2). Participants with low vitamin D had a significantly higher median BMI compared to those with normal levels (26.4 vs 23.9 kg/m2, P = 0.001). The majority of the participants (92.7%) reported engaging in physical activity. Regarding HIV clinical characteristics, 66.5% had been on ART for more than 5 years, and 93.6% had undetectable viral load. Most participants (73.4%) reported awareness of VDD (Table 2).
Minimum and Maximum Values of Atherogenic Indices in Each Tertile.

LCI, lipid combination index; RC, residual cholesterol.
Sociodemographic and Clinical Characteristics of the Study Participants.

Note: Bold values indicate statistically significant findings.
ART, antiretroviral therapy; IQR, interquartile range.
Biochemical Parameters and Indices of the Study Participants
The median hs-CRP level was 1.37 mg/L, with significantly higher values among participants with low vitamin D compared to those with normal levels (1.66 vs 1.18 mg/L, P = 0.043). Similarly, the hs-CRP/HDL-C ratio was significantly elevated among participants with low vitamin D levels in comparison to those with normal levels (0.004 vs 0.003, P = 0.028). Additionally, TC/HDL-BMI index was significantly higher among participants with low vitamin D compared to those with normal levels (93.25 vs 83.49, P = 0.005) as indicated in Table 3.
Biochemical Parameters and Indices of the Study Participants.
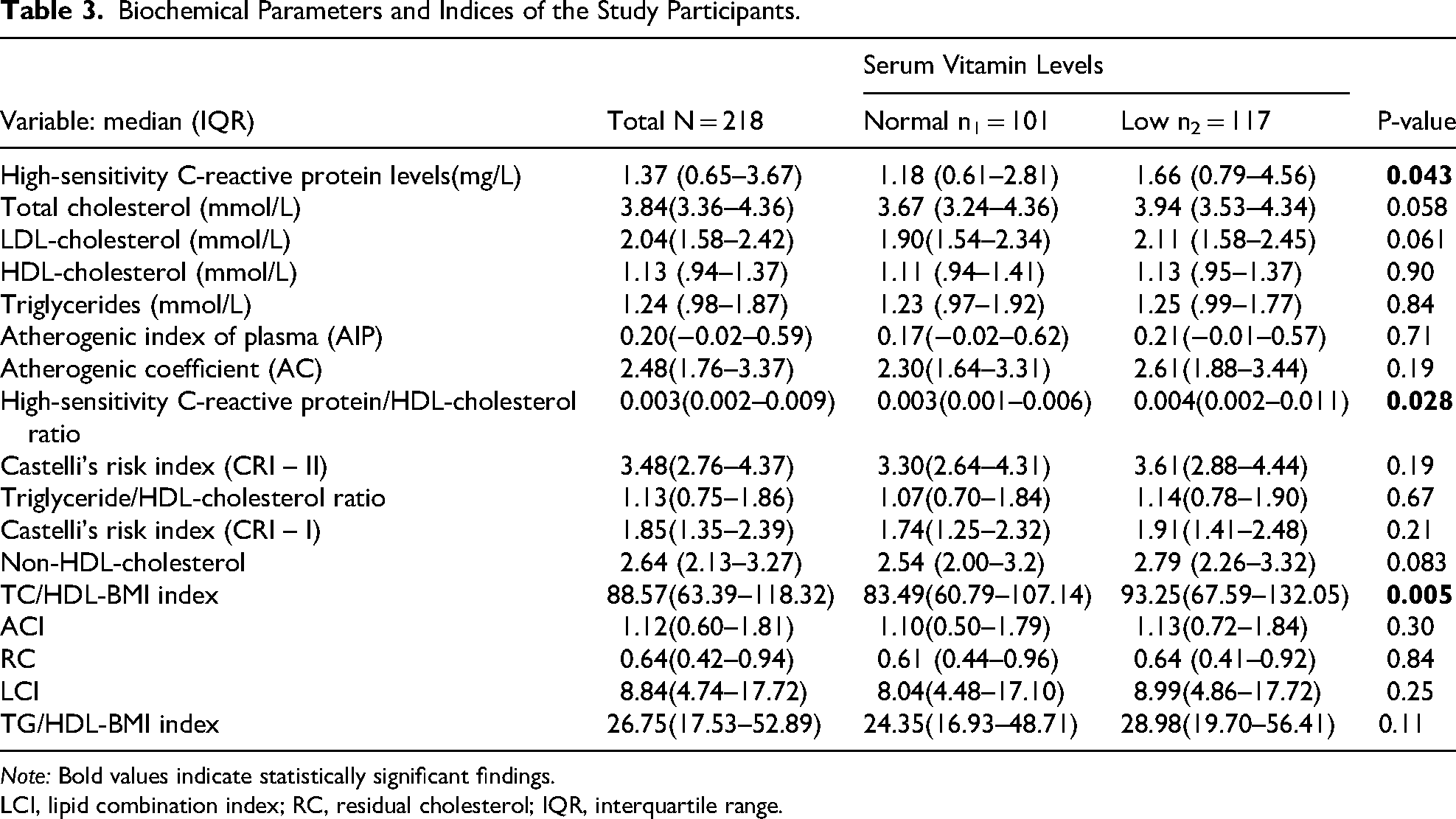
Note: Bold values indicate statistically significant findings.
LCI, lipid combination index; RC, residual cholesterol; IQR, interquartile range.
Prevalence of Low Vitamin D Levels
Out of the 218 participants, 117(53.7%; 95CI:47.0-60.2) had low vitamin D levels. Majority of the participants; 94 (43.1%) had serum 25-hydroxyvitamin D concentrations between 20-29 ng/mL, consistent with VDI while 23 (10.6%) had levels <20 ng/mL, indicating VDD as indicated in Figure 1. The prevalence of low vitamin D levels significantly increased from the lowest to the highest tertile of high-sensitivity CRP/HDL-cholesterol ratio; (45% Vs. 49% Vs.67%, P =

Proportion of study participants with low vitamin D levels.
Distribution of Low Vitamin D Level Prevalence in the Different Tertiles of Atherogenic Indices.

Note: Bold values indicate statistically significant findings.
CRP, C-reactive protein; LCI, lipid combination index; RC, residual cholesterol.
Association of Atherogenic Combined Index, hs-CRP/HDL-C Ratio, and TC/HDL-C-BMI Index with Low Vitamin D Levels
After adjustment for potential confounders in the multivariate analysis (Table 5), ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index were significantly and independently associated with low vitamin D levels. Participants in the second and third tertiles of the ACI had significantly higher likelihood of low vitamin D compared to those in the first tertile (aPR = 2.23, 95% CI: 1.28-3.89, P = 0.005 and aPR = 2.53, 95% CI: 1.05-6.05, P = 0.038, respectively). Similarly, those in the third tertile of the hs-CRP/HDL-C ratio were significantly more likely to have low vitamin D than those in the first tertile (aPR = 1.44, 95% CI: 1.05-1.96, P = 0.024). The TC/HDL-C-BMI index also showed a strong independent association, with individuals in the third tertile having nearly twice the prevalence of low vitamin D compared to the reference group (aPR = 1.95, 95% CI: 1.10-3.45, P = 0.023).
Association of Atherogenic Indices with Low Vitamin D Levels.

Note: Bold values indicate statistically significant findings.
cOR, crude prevalence ratio; aOR, adjusted prevalence ratio; CI, 95% confidence interval; ART, antiretroviral therapy; LCI, lipid combination index; RC, residual cholesterol; CI, confidence interval;TC/HDL-C-BMI, total cholesterol to high density lipoprotein cholesterol - body mass index.
Discussion
In our southwestern Ugandan HIV cohort on ART, 53.7% had serum 25(OH)D < 30 ng/mL (43.1% insufficient, 10.6% deficient), indicating a high burden of suboptimal vitamin D. Vitamin D modulates immunity and deficiency can exacerbate HIV-related inflammation. Our prevalence exceeds Ugandan reports: Conesa-Botella et al found about 15% deficiency (<20 ng/mL) among PLWH 35 and Piloya et al found 13% < 30 ng/mL and 3% < 20 ng/mL in HIV + Ugandan children. 36 It also exceeds healthy African estimates (17.4% deficiency in Kenyan donors 37 ) and approaches continental averages (59.5% < 30 ng/mL, 34.2% < 20 ng/mL 38 ). Global data show about 48% < 20 ng/mL. 39 These discrepancies likely reflect differences in age, ART regimens, sun exposure (equatorial Uganda vs higher latitudes) and urban versus rural lifestyle. 38 For example, Mogire et al found urban Africans had significantly lower 25(OH)D than rural populations. 38 Notably, Mogire also reported about 20% of Africans with <12 ng/mL and about 60% with <30 ng/mL, 38 findings similar to ours. The predominance of insufficiency suggests many are near deficiency thresholds. Clinically, this implies that routine vitamin D monitoring and supplementation might improve bone and immune health in Ugandan PLHIV. Additionally, a meta-analysis confirmed that PLHIV have higher VDD prevalence than uninfected controls, consistent with our data. 40
In our study, higher ACI was strongly associated with VDD: participants in the second/third ACI tertiles had over twice the odds of low vitamin D compared to those in the first tertile. ACI integrates LDL, TG and HDL values and predicts cardiovascular risk, so its correlation with hypovitaminosis D suggests converging metabolic disturbances. Clinically, HIV patients with dyslipidemia should be screened for VDD as part of cardiovascular risk management. Biologically, VDD induces a pro-atherogenic state: low 25(OH)D is linked to higher triglycerides, LDL and inflammatory mediators, whereas vitamin D sufficiency raises HDL and lowers atherogenic lipids. 41 Consistent with our findings, studies in other settings report similar inverse vitamin D-lipid associations: obese youths with low vitamin D had worse lipid profiles and higher AIP, 42 and large US cohorts showed significantly higher AIP in vitamin D-deficient adults. 43 A Taiwanese study also found that 25(OH)D < 30 ng/mL was associated with significantly higher odds of atherogenic dyslipidemia. 44 Conversely, some HIV cohorts report weaker links: one US women's study found 25(OH)D correlated only with triglycerides (not cholesterol) and was suppressed by efavirenz use, 45 and an HIV-infected youth study found no 25(OH)D-lipid correlation. 46 Discrepancies may reflect ART effects or demographic and nutritional differences. Overall, our results align with evidence that VDD and dyslipidemia commonly co-occur, highlighting vitamin D as a preventable cardiovascular risk factor. 42
We also observed that PLWH with low vitamin D levels had significantly higher hs-CRP/HDL-C ratio and in the adjusted analysis, the prevalence of low serum vitamin D significantly rose across increasing hs-CRP/HDL-C ratio tertiles. Clinically, this indicates that individuals with pronounced inflammatory dyslipidemia are more likely to be vitamin D deficient. The hs-CRP/HDL-C index is a composite marker of systemic inflammation and lipid imbalance, 24 suggesting that our vitamin D-deficient subgroup has a pro-inflammatory, pro-atherogenic phenotype. Given that both chronic inflammation and vitamin D status independently impact HIV outcomes,40,47 this association has important clinical significance for managing cardiovascular and immune risk in PLHIV. Vitamin D has well-known immunomodulatory effects: its active form down-regulates nuclear factor-κB and reduces pro-inflammatory cytokine production. 48 VDD therefore increases systemic inflammation as documented by elevated levels of CRP and IL-6.48–51 hs-CRP is a commonly used inflammatory biomarker in clinical practice. 24 Conversely, chronic inflammation (as seen in HIV) and certain ARVs can impair vitamin D metabolism, for example, pro-inflammatory cytokines inhibit 1α-hydroxylation of 25(OH)D.48,52–54 Meanwhile, HDL-cholesterol has anti-inflammatory and antioxidant functions, but inflammation and ART often lower HDL and raise CRP, elevating the hs-CRP/HDL-C ratio.24,51 Population data support these links: higher 25(OH)D correlates with lower hs-CRP.47,48,51,55 Together, these mechanisms form a feed-forward cycle in which VDD and inflammation reinforce each other, making our observed association biologically plausible.
Our findings align with prior observations. Poudel-Tandukar et al reported that HIV patients with 25(OH)D < 20 ng/mL had >3-fold higher odds of elevated CRP, 48 and Currò et al found that VDD in treated PLHIV was associated with upregulated inflammatory cytokines (IL-6, hs-CRP). 47 The graded increase in low vitamin D prevalence across hs-CRP/HDL tertiles resembles findings from NHANES, where higher hs-CRP/HDL quartiles predicted greater metabolic risk. 24 However, some studies note non-linear patterns: Carr et al described a U-shaped vitamin D-inflammation relationship in HIV (elevated CRP at very high 25(OH)D), 56 and Poudel-Tandukar observed the CRP link only in men. 57 These discrepancies may reflect cohort or ART differences. Overall, our results reinforce the prevailing view that vitamin D sufficiency accompanies lower systemic inflammation in HIV.47,51
Our cross-sectional study also found that a composite TC/HDL-C-BMI index was strongly associated with low vitamin D. Participants in the highest index tertile had nearly double the prevalence of VDD (aPR=1.95). Clinically, this suggests that dyslipidemic, overweight PLWH may warrant targeted vitamin D screening since deficiency can worsen bone demineralization, immune recovery and cardiovascular risk. Biologically, adiposity sequesters the fat-soluble vitamin and dyslipidemia reflects a pro-inflammatory metabolic milieu that plausibly lowers 25(OH)D levels. 58 Our finding agrees with prior reports: for example, Souza et al showed that elderly vitamin-D-sufficient adults had significantly lower TC/HDL ratios than those deficient, 59 and Nwosu et al observed inverse 25(OH)D-TC/HDL correlations in children. 60 Likewise, a large cohort of pregnant women found low 25(OH)D predicted higher atherogenic lipid ratios. 61 In contrast, Schwartz et al found no cholesterol-vitamin D association in HIV-infected women, and Conesa-Botella et al noted that lower body weight (not high BMI) was linked to severe VDD in HIV patients. 62 Such discrepancies likely reflect differences in populations, ART regimens (eg, NNRTI vs DTG), nutrition or confounder control. Nonetheless, our results consistent with cardiometabolic literature58,59 are biologically plausible, suggesting the TC/HDL-C-BMI index could help identify PLWH at high risk of VDI.
Overall, our study demonstrates that ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index are significantly associated with low vitamin D levels among PLWH on ART, underscoring the intertwined relationship between chronic inflammation, dyslipidemia, obesity, and micronutrient deficiency. This finding suggests that the ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index, readily measurable composite marker of inflammatory, lipid status and obesity, may serve as potential biomarker for identifying PLWH at risk of low vitamin D. Incorporating these indices into routine clinical assessment could support targeted monitoring and timely interventions, ultimately helping to mitigate the adverse metabolic and inflammatory complications associated with HIV and long-term ART.
Conclusions and Recommendations
VDI and VDD are prevalent among PLWH in Southwestern Uganda. ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index are potential predictors for low serum vitamin D levels in this population. We recommend further longitudinal studies to investigate temporal relationships between abnormal serum atherogenic indices particularly ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index with the risk of VDD and insufficiency among PLWH. These findings could inform targeted screening and intervention strategies in HIV care programs.
Study Limitations
Our study has some limitations that should be considered when interpreting the findings. First, as a cross-sectional study, it provides only a snapshot of the prevalence of low vitamin D levels and its association with high serum levels of ACI, hs-CRP/HDL-C ratio, and TC/HDL-C-BMI index, limiting our ability to establish causal relationships or determine the temporal sequence of events. Additionally, certain variables, such smoking, alcohol consumption status, and knowledge about VDD were self-reported, hence potential for inaccurate reporting. These limitations highlight the need for further research, including longitudinal studies, to better understand the relationship between abnormal serum atherogenic indices with the risk of VDI and deficiency.
Footnotes
Acknowledgements
We would like to thank the participants who provided data for this study.
Ethics Approval and Consent to Participate
This study received ethical approval from the Research Ethics Committee (REC) of Mbarara University of Science and Technology with approval number (MUST-2024-1609). Administrative clearance was also obtained from the Office of the City Town Clerk, Mbarara City before conducting the study. All participants provided written informed written consent prior to enrollment. The consent forms were translated into the local language (Runyankore) to ensure comprehension. Formally educated participants gave consent by signing the written forms. For participants with no formal education, the written informed consent form was read aloud in Runyankore, followed by administration of the comprehension screening tool approved by the MUST REC. Only participants who demonstrated understanding were allowed to consent by placing a thumbprint on the informed consent form. Participants were also explicitly asked to provide consent for the use of their data in the secondary analysis, for which they approved. This study was conducted in accordance with the Declaration of Helsinki (1975) as revised in 2024. Confidentiality of the study participants was observed by giving each participant a study code that was not traceable to them. Participants were also informed that their participation is free and voluntary and that they had a right to withdraw from the study at any time.
Authors’ Contributions
CNB, CN, and JT participated in the conceptualization of the study and data collection. CNB performed data analysis and results interpretation. CNB, JT, CN, AO, DN, MJM, BR, VM, EAJ, FS, IK, LGA, RN, HI, CL, and ROO contributed to writing the first draft of the manuscript with RN, HI, and ROO providing critical revisions. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from Mbarara University of Science and Technology, Faculty of Medicine, Office of the Dean, Faculty Research Support/ Funding
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets used in this study are available from the corresponding author upon reasonable request.