Abstract
Background
The use of dolutegravir (DTG)-based antiretroviral therapy (ART) has been associated with the development of hyperglycemia. This study determined the prevalence and factors associated with hyperglycemia among people living with HIV (PLWH) on DTG-based ART in Uganda.
Methods
We conducted a cross-sectional study among 219 systematically recruited PLWH on DTG-based ART for ≥1 year at a Health Centre IV in Central Uganda. Data were collected using a structured questionnaire, anthropometric measurements, and laboratory investigations. Logistic regression was performed to identify factors associated with hyperglycemia.
Results
Out of 219 participants, 45 had fasting glucose ≥110 mg/dL, giving a hyperglycemia prevalence of 20.55% (95% confidence interval [CI]: 15.68%-26.45%). Overweight (aOR 8.11, 95% CI: 2.76-23.85, P < .001) and abnormal waist-to-hip ratio (aOR 4.36, 95% CI: 1.23-15.37, P = .022) were significantly associated with hyperglycemia.
Conclusions
Hyperglycemia is prevalent among PLWH on DTG-based ART and is potentially associated with overweight and abnormal waist-to-hip ratio.
Plain Language Summary
People living with HIV now live longer because of effective antiretroviral therapy. However, some HIV medicines can cause long-term health problems, including high blood sugar (hyperglycemia), which increases the risk of diabetes and heart disease. Dolutegravir is the most commonly used HIV medicine in Uganda, but concerns have been raised about its link to high blood sugar. This study was carried out at Ntwetwe Health Centre IV in Central Uganda to find out how common high blood sugar is among adults living with HIV who are taking dolutegravir-based treatment, and to identify factors associated with it. A total of 219 adults who had been on dolutegravir for at least one year were included. Information was collected through interviews, body measurements (such as weight and waist size), and laboratory blood tests after overnight fasting. The study found that about 1 in every 5 patients (20.6%) had high blood sugar. This means that high blood sugar is fairly common among people living with HIV on dolutegravir. Men were more likely to have high blood sugar than women. Patients who were overweight and those with excess fat around the waist and hips were also at much higher risk. These findings highlight the importance of regularly checking blood sugar levels in people living with HIV who are taking dolutegravir, especially men and those who are overweight or have increased abdominal fat. Early screening can help detect high blood sugar before it progresses to diabetes and its complications. The study recommends that routine blood sugar testing should be included during the start of treatment and follow-up care for people living with HIV on dolutegravir-based therapy in Uganda.
Introduction
Human immunodeficiency virus (HIV) infection remains a major global health challenge, 1 with approximately 38 million people living with the virus in 2022. 2 Uganda continues to bear a substantial burden of HIV, with national estimates indicating an adult HIV prevalence of 5.1% to 5.5%.3–5 According to the Uganda Population-based HIV Impact Assessment, 80.8% of people living with HIV (PLWH) aged 15 years and older were aware of their HIV status, 96.0% of those diagnosed had been initiated on antiretroviral therapy (ART), and 92.0% had achieved viral load suppression in 2021. 6
The scale-up of ART across sub-Saharan Africa, including Uganda, has markedly improved survival and life expectancy among PLWH. 7 However, the long-term use of ART has been accompanied by emerging metabolic abnormalities and cardiometabolic comorbidities such as diabetes mellitus, hypertension, and dyslipidemia that complicate sustained HIV care. People living with HIV receiving ART are at increased risk of these conditions3,8 which contribute substantially to non-AIDS morbidity and mortality. 9
Following a recommendation from World Health Organization in 2018, dolutegravir (DTG)-based ART regimens were adopted as first-line therapy for all PLWH in Uganda.10,11 Dolutegravir is a potent integrase strand transfer inhibitor with a high genetic barrier to resistance, favorable tolerability, and minimal drug–drug interactions, commonly administered with tenofovir and lamivudine.11,12 Despite these advantages, accumulating evidence from clinical studies and case reports indicates that DTG use is associated with metabolic disturbances, particularly hyperglycemia.12–15 In a qualitative study by Zakumumpa et al 16 among 49 ART clinic managers and clinicians, hyperglycemia was the most frequently reported adverse effect of DTG-based regimens. Additionally, DTG has been linked to significant weight gain, with subsequent obesity and insulin resistance, both of which are strongly associated with hyperglycemia.8,17,18 Proposed mechanisms include impaired insulin secretion and signaling, potentially mediated through magnesium chelation.18,19
In Uganda, a number of studies have documented the association between DTG-based ART and hyperglycema.13,20,21 A recent prospective study by Collins Ankunda et al reported a higher incidence of hyperglycemia among PLWH exposed to DTG (25.2 per 100 person-years) than among those not exposed (20.8 per 100 person-years). 22 Hospital-based studies conducted in urban referral centers have reported hyperglycemia prevalences ranging from 7.3% at Mbarara Regional Referral Hospital (RRH) 23 to 12.8% at Kiruddu National Referral Hospital, 24 with a similar prevalence of 12.5% reported in Eastern Uganda, 25 where over 90% of participants were receiving DTG-based ART.
Notably, existing evidence on DTG-associated hyperglycemia in Uganda is predominantly derived from urban and tertiary care settings, where access to diagnostic and monitoring facilities is relatively better. Yet, a substantial proportion of PLWH reside in rural and periurban areas, where routine metabolic screening is limited and hyperglycemia may remain undiagnosed. Consequently, the true burden of hyperglycemia among PLWH on DTG-based ART in these settings is likely underestimated. This study therefore aimed to determine the prevalence of hyperglycemia and identify associated factors among PLWH receiving DTG-based ART in a lower-level health facility in Central Uganda.
Methods and Materials
Study Location and Study Period
The study was conducted at Ntwetwe Health Centre IV ART Clinic; a public Health facility which serves over 2000 PLWH receiving DTG-based ART. Majority of the patients who receive ART services from the study site come from Kyankwanzi, Kiboga, Mityana, Mubende, and Kibaale districts. This study site is located in Ntwetwe constituency in Ntwetwe Town Council along with Masodde-Kibaale road, Kyankwanzi district, Central Uganda. The study was conducted between from September 22, 2023, and October 27, 2023. Baseline random blood glucose testing is not routinely performed for ART-naïve patients initiating DTG-based ART at Ntwetwe Health Centre IV ART clinic. Clinical decisions at treatment initiation are guided primarily by patients’ medical history and clinical presentation rather than glycemic assessment. Blood glucose testing is typically requested only when patients present with symptoms suggestive of diabetes mellitus. Furthermore, routine monitoring of blood glucose levels during follow-up of PLWH receiving DTG-based ART is not standard practice at this facility
Study Design and Populations
We employed a cross-sectional study to determine prevalence and factors associated with hyperglycemia among patients on DTG-based ART.
The source population of this study were all adult PLWH on DTG-based ART, while those systematically selected PLWH attending Ntwetwe Health Centre IV ART Clinic during the data collection period were the study population.
Sample Size Determination and Sampling Technique
The Keish Leslie formula of 1965
26
was used to calculate the minimum required sample size using the following assumptions; 17.1% proportion of PLWH in Ethiopia on DTG-based ART who developed hyperglycemia after follow up,
27
5% precision and a 95% confidence interval (CI) and the Z-statistic of 1.96:
Therefore, the minimum sample size of 218 study participants was required for this study.
Adult PLWH aged 18 years and above who had been receiving DTG-based ART for at least 12 months and who provided written informed consent were enrolled using systematic sampling. Based on an estimated clinic attendance of approximately 660 eligible PLWH within duration of 1.5 months, a systematic sampling interval of 3 was applied. During the study period from September 22, 2023, to October 27, 2023, eligible participants attending the ART clinic were listed in order of arrival, and every third eligible patient was consecutively selected and recruited until the required sample size was achieved.
Eligibility Criteria
We included all male and female adult PLWH who were aged 18 years and above on DTG-based ART for a period of at least 12 months using a systematic selection procedure. However, those adult PLWH on DTG-based ART who were pregnant, unable to communicate, breastfeeding, missing medical records, and those visiting clinic due to critical illness were excluded from the study. Participants with a known diagnosis of hyperglycemia; preexisting diabetes mellitus or prediabetes and thus receiving hypoglycemic agents were also excluded from the study. Additionally, we also excluded those on lipid-lowering medication, corticosteroids, or oral contraceptives pills from the study.
Variables
The outcome variable of this study was hyperglycemia ascertained based on fasting blood glucose measurement done under standard methods. The independent variables were categorized as social-demographic factors, behavioral factors, clinical factors, and HIV and/or ART-related factors, and biochemical parameters. The social-demographic factors included age, sex, and education level, religion, employed status, and marital status. The behavioral factors included smoking status, alcohol consumption, and physical activity. Clinical factors included hypertension, family history of hypertension, family history of diabetes mellitus, overweight, obesity, waist circumference (WC), waist-to-height ratio, and waist-to-hip ratio. HIV and/or ART-related factors include duration on ART, and duration on DTG-based ART. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters (kg/m2). Waist circumference was measured as the smallest circumference around a person's waist just above the hips. Waist-to-hip ratio was obtained by dividing WC by hip circumference the biochemical parameters included total cholesterol (TC), serum triglycerides (TG), and serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST).
Definition of Operational Terms
Hyperglycemia: Fasting plasma glucose (FPG) ≥110 mg/dL.28–30
Prediabetes: FPG 110-125 mg/dL. 31
Diabetes mellitus: FPG ≥126 mg/dL. 31
Body mass index: Categorized as <25 (normal), 25 to 29.9 (overweight), and ≥30 (obesity). 32
Waist circumference: Considered abnormal if WC > 94 cm for men and >80 cm for women. 33
Waist-to-hip ratio: Considered abnormal if ≥0.90 for males or ≥0.85 for females. 33
Waist-to-height ratio: Considered abnormal if ≥0.5 34
Total cholesterol: Classified as elevated if TC ≥ 200 mg/dL, 30
Triglycerides: Considered elevated if TG ≥ 150 mg/dL. 30
Hypertension: Defined as systolic blood pressure ≥140 mm Hg and/or diastolic blood pressure ≥90 mm Hg. 35
Methods of Data Collection
The information on social-demographic, family background for chronic diseases (diabetes mellitus, hypertension, cardiovascular diseases), and behavioral factors was collected by 2 trained research assistants; one an enrolled nurse and the other a final-year student of a Clinical Medicine who directly interviewed study participants in Luganda (local language) using a semistructured questionnaire. The 2 trained research assistants also reviewed medical records of the consented study participants to obtain data regarding duration on ART, and duration on DTG-based ART, and recent viral load results and Medications.
The measurement of height and weight that were required in the calculation of BMI, waist/hip circumferences needed to calculate waist-to-hip ratio, and blood pressure were also performed by the 2 research assistants. A digital sphygmomanometer was used to take blood pressure measurements. Two blood pressure readings were recorded at a 5-min interval, and their mean value was taken as the blood pressure. Weight and height of the participant were measured using a portable weight and height scale. The waist and hip circumferences were measured using a nonelastic tape meter. Waist circumference was measured midway between the lower rib and the iliac crest in a horizontal plane at around the umbilicus while the respondents were instructed to breathe gently out. In order to prevent pressure and bias in measurement, the tape meter was not fastened securely. Two circumference readings were taken, and third measurement was only done if the difference between first 2 was greater than 3 cm. Waist and hip circumferences was taken as the mean of 2 or 3 measurements. Hip circumference at the point yielding the maximum circumference over the buttocks was measured with a tape meter in a standing position at the end of a gentle expiration. Measurements were also made at least twice, and the average of the 2 measurements was recorded.
About 4 mL of venous blood sample was collected from a study participant who had fasted overnight using fluoride-oxalate (gray top) and red top vacutainer vacuum bottles by the principal investigator and a laboratory assistant attached to the ART clinic mini-laboratory. Plasma and serum were separated by centrifugation at 3000 rpm for 4 min within a period not more than 1 h after blood collection. The separated serum and plasma samples were stored in cryovials in the main laboratory freezer at 2 °C to 8 °C before analysis in the evening of each day of blood sample collection. The plasma glucose, serum TC and TG concentration, ALT, and AST activities were measured using the HumaStar 80 clinical chemistry analyzer (Human Diagnostic) at Ntwetwe Health Centre IV (Clinical Chemistry department) laboratory Hub, Kyankwanzi district.
Data Quality Assurance
Two health personnel, one an enrolled nurse and the other a final-year student of a diploma in Clinical Medicine, were deployed as research assistants to collect the data from study subjects. Before beginning the actual data collection process, a medical officer at Ntwetwe Health Centre IV trained the 2 research assistants on proper interview techniques and anthropometric measurements. Throughout the data collection period, we employed standard measuring procedures and instruments. The questionnaire was validated through checking for its clarity, correctness, completeness from the supervisor and consistence of the items in it, coherence of the tool with connection to the conceptual frame work, objectives of the study, and statement of the problem. It was also pretested on among 10 PLWH who met our inclusion criteria at Kiyuni Health Center III ART clinic.
We used a well calibrated HumaStar 80 chemistry analyzer for biochemical investigations. The analyzer calibration was performed using AutoCal. Normal quality control samples, that is, HumaTrol N and pathological quality control samples, that is, HumaTrol P were run on the analyzer before research samples were run. Additionally, 10% of the collected samples collected were shipped and tested for study biochemical parameters at the Mbarara University of Science and Technology Clinical and Research Laboratory to guarantee the accuracy of the results obtained from the onsite testing laboratory. The results from the 2 laboratories did not significantly differ from each other. The data from the questionnaires and laboratory results were also carefully doubled entered by the Principal researcher into Microsoft Excel. The data were examined for correctness and consistency with the data on the questionnaires and laboratory results before being exported to STATA software for statistical analysis.
Methods of Data Analysis
Collected research data in the Microsoft Excel was exported to STATA Software version 17 Software for analysis. All studied variables were compared by gender of the participants at a univariate level to test for statistical difference between the 2 groups. Categorical variables were summarized using frequencies and proportions, and then a χ2 test or Fisher exact test was used to compare them by gender. A P-value <.05 was considered to be statistically significant.
Mean with standard deviation was used to summarize continuous variables that were normally distributed while median with interquartile range (IQR) was used to summarize those that were not normally distributed. A Student t test was used to compare means between the gender categories for normally distributed continuous variables and the medians for the variables that are not normally distributed were compared using the Mann-Whitney U test. A P-value was obtained, and a P-value <.05 was considered statistically significant.
The proportion of participants with hyperglycemia together with its 95% CI was determined by dividing the number of patients with hyperglycemia by the total number of study participants and was expressed as percentage. The prevalence of hyperglycemia in the different groups of the categories variables was compared using a χ2 test or Fisher exact test to test for any statistical difference in its distribution. A P-value <.05 was statistically significant.
The factors associated with hyperglycemia were assessed using logistic regression analysis. All independent variables, at bivariate level, were compared with hyperglycemia (glucose levels that was dichotomized into high and normal levels). The associations were measured using odds ratios together with their 95% CIs, and the statistically significant odds ratios were indicated by a P-value <.2 at the bivariate level.
The variables that were clinically and/or statistically significant at this level were also included in the multivariate model to adjust for confounding effects in order to obtain factors independently associated with hyperglycemia. In the final multivariate model, associations were considered significant at a P-value <.05. We used Hosmer-Lemeshow test to test for the suitability of the final selected model in predicting our outcome variable (hyperglycemia), and the obtained P-value of .4189 indicated good goodness of fit for our final selected model.
Results
Study Flowchart

Narrative of Study Flowchart
A total of 271 PLWH on ART were initially approached for study participation. After applying the eligibility criteria, 219 participants were enrolled. All 219 participants were included in the final analysis. Among these, 45 participants (20.55%) had hyperglycemia, while 174 participants (79.45%) did not have hyperglycemia.
Characteristics of the Study Participants
A total number of 219 PLWH on DTG-based ART fully participated in this study with majority being females 116/219 (52.97%). The median age of the participants was 45 years (IQR: 34-54) with males; 46 years being older than females; 44 years; P = .049. Majority of the study participants were married/cohabiting; 117 (53.42%), attained primary level of education; 146 (66.67%) were employed; 130 (59.36%) and were catholic by religion; 98 (44.75%) (Table 1).
Characteristics of Study Participants Stratified by Gender.
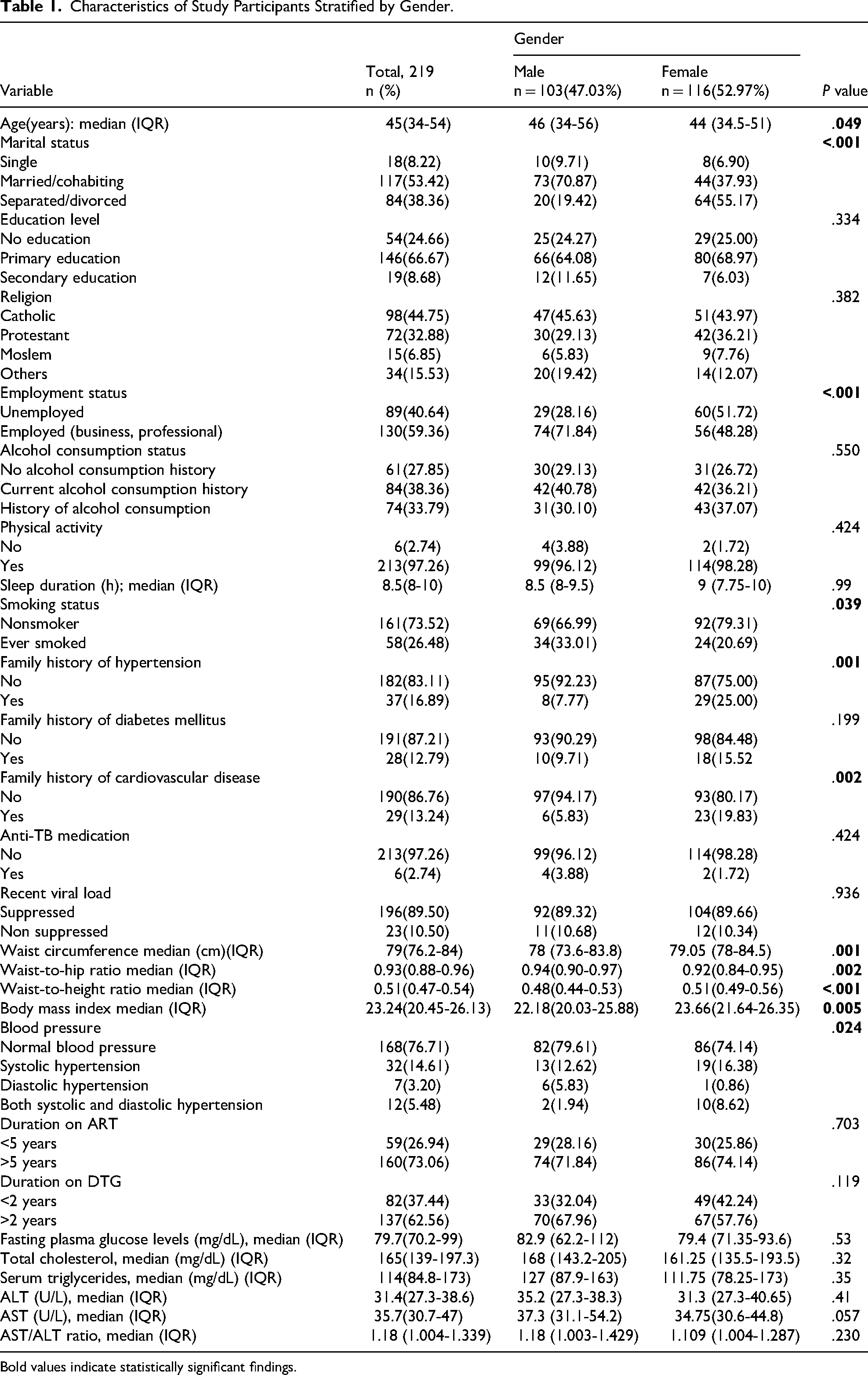
Bold values indicate statistically significant findings.
Significant differences in the distribution of variables such as smoking status, marital status, blood pressure, employment status, and family history of hypertension and cardiovascular diseases were observed between male and female study participants, P-value <.05. Additionally, the median values of obesity indices were significantly higher in female study participants than in males; P-value <.05 (Table 1).
Prevalence of Hyperglycemia Among PLWH on DTG-Based ART
Out of the 219 study participants, 45 had FPG ≥ 110 mg/dL, giving an overall prevalence of hyperglycemia of 20.55% (95%CI: 5.68%-26.45%). The prevalence of prediabetes was 15.53% (34/219; 95% CI: 11.29%-20.98%), while the prevalence of diabetes mellitus was 5.02% (11/219; 95% CI: 2.79%-8.87%).
The prevalence of hyperglycemia was significantly higher in males (21.13%) than in the females (12.93%), P = .003(Table 2). Additionally, a significant difference in the distribution of hyperglycemia was observed by duration on DTG-based ART: ≥2 years (26.28%) versus <2 years (10.98%) P = .007, TC; ≥200 mg/dL (33.33%) versus <200 mg/dL (16.67%) P = .010, waist-to-height ratio; abnormal (25.78%) versus normal (13.19%) P = .023, BMI; 25-29.9 kg/m2 (51.85%) versus >30 kg/m2 (15.38%) versus <25 kg/m2 (9.87%), P ≤ .001, blood pressure; diastolic hypertension (71.43%) versus systolic hypertension (12.50%) versus both systolic and diastolic hypertension (25.00%) versus normal (19.64%) P = .012, and employment status; employed (25.38%) versus unemployed (13.48%) P = .032 (Table 2).
Distribution of Hyperglycemia Prevalence in the Different Categorical Variables.

Bold values indicate statistically significant findings.
Factors Associated With Hyperglycemia Among PLWH on DTG-Based ART
After adjustment for confounders, male gender; aOR: 4.90 (95%CI: 1.45-16.59, P = .011), having a BMI of 25 to 29.9 kg/m2; aOR: 8.11 (95%CI: 2.76-23.85, P < .001), and abnormal waist-to-hip ratio; aOR: 4.36 (95%CI: 1.23-15.37, P = .022) were significantly associated with hyperglycemia (Table 3).
Factors Associated With Hyperglycemia.

Abbreviations: aOR, adjusted odds ratio; cOR, crude odds ratio.
P-value <.05 indicates statistical significance. Bold values indicate statistically significant findings.
Discussion
In this rural Ugandan population receiving DTG-based ART, approximately one-fifth of PLWH had hyperglycemia, comprising 15.5% with prediabetes and 5.0% with diabetes mellitus. Clinically, this substantial burden of dysglycemia is concerning, as sustained elevations in blood glucose are known to increase the risk of cardiovascular and microvascular complications and may reflect DTG-associated metabolic effects reported in other settings.24,25 This finding suggest that undiagnosed hyperglycemia may be common among PLWH on DTG-based ART in the rural setting of Central Uganda. Dolutegravir has been linked to significant weight gain, with subsequent obesity and insulin resistance, both of which are strongly associated with hyperglycemia.8,17,18 Proposed mechanisms include impaired insulin secretion and signaling, potentially mediated through magnesium chelation.18,19 Therefore, treatment guidelines should emphasize blood glucose monitoring for PLWH on DTG regimens as early identification of prediabetes and diabetes can guide interventions to mitigate long-term complications.24,25 Importantly, HIV infection itself has been recognized as an independent risk factor for dysglycemia.36,37 As our study did not include an HIV-negative comparison group, we were unable to disentangle the relative contributions of HIV infection and ART to hyperglycemia. HIV-related metabolic effects should therefore be considered a potential underlying contributor and confounder when interpreting our findings.
In Uganda and the region, reported hyperglycemia in this population varies widely. Our 20.6% hyperglycemia prevalence is comparable to 19.8% found among DTG-treated adults at Jinja RRH. 38 However, it exceeds the prevalence from central Uganda Kiruddu Hospital (12.8%) 24 and a rural eastern Ugandan study (12.5%) 25 and is much higher than 7.3% reported at Mbarara RRH. 23 These lower estimates may reflect methodological differences. For example, the Kiruddu study and the Eastern Uganda study both used glycated hemoglobin (HbA1c) (5.7% cutoff) to define hyperglycemia. While HbA1c is generally more reliable for identifying established hyperglycemia than a single fasting blood glucose measurement, it may miss early or acute hyperglycemia. In contrast, we used FPG ≥ 110 mg/dL and required ≥1 year on DTG-based ART, while Kiruddu included participants who had been on DTG-based ART for at least 3 months and the study at Mbarara RRH was retrospective screening for hyperglycemia among PLWH on DTG-based ART who had at least an adverse drug event recorded on their file. Urban-rural differences and sample size may also matter. In our rural cohort, traditional diets or undetected risk factors could raise prevalence compared to urban cohorts. Thus, inclusion criteria and setting likely explain much of the Ugandan heterogeneity.
Across East Africa, our observed hyperglycemia prevalence of 20.6% lies between the estimates reported from Ethiopia and Kenya. In Ethiopia, a cross-sectional study conducted at Debre Markos Referral Hospital among PLWH who had received DTG for at least 6 months reported a lower hyperglycemia prevalence of 12.1%, 39 which is notably below our estimate. This discrepancy is likely explained by methodological and population differences. Although both studies used FPG to assess glycemic status, the Ethiopian study included participants with a shorter minimum duration on DTG-based ART (≥6 months), whereas our study restricted inclusion to individuals on DTG-based ART for at least one year, allowing greater cumulative metabolic exposure. Additionally, the Ethiopian study was conducted in an urban referral setting, where earlier diagnosis and routine metabolic screening may have reduced the proportion of undetected dysglycemia compared with our rural cohort.
In contrast, a Kenyan referral-center study reported a markedly higher hyperglycemia prevalence of 55.9%, 40 which substantially exceeds our findings. This difference is plausibly attributable to variations in diagnostic approaches and inclusion criteria. Unlike our study, which defined hyperglycemia strictly using FPG ≥ 110 mg/dL, the Kenyan study employed a combination of random blood glucose and HbA1c measurements, which are more sensitive to both acute and chronic dysglycemia and therefore likely to identify a larger number of cases.
In broader SSA contexts, studies also report higher rates. In Zimbabwe, a recent study of PLWH including TB coinfection on standard or double-dose DTG found 31% hyperglycemia. 41 Here, hyperglycemia was defined by HbA1c. Similarly, a multicenter survey in Niger reported 36.4% prevalence using FPG ≥ 126 mg/dL. 42 Both are well above our 20.6%. These SSA studies’ higher rates may stem from unique factors: the Zimbabwe analysis included double-dose DTG which may elevate glucose. In Niger, only 39% had baseline glucose data (potential selection bias), and the population may differ in demographics or background disease.
Our finding that male sex was associated with nearly 5-fold higher odds of hyperglycemia may reflect sex-specific metabolic differences. Men tend to accumulate more visceral fat and lack estrogen's insulin-sensitizing protection, predisposing them to insulin resistance and impaired glucose control.43,44 These biological factors, together with possible behavioral and genetic differences, may potentiate DTG-related metabolic disturbances. Our results are consistent with a case–control study conducted in Mbale, Uganda, which also reported a significant association between male sex and hyperglycemia among PLWH receiving DTG-based ART. 45 However, our findings contrast with those from another Ugandan prospective study that found no significant sex differences in DTG-associated hyperglycemia, 46 as well as a Kenyan study in which age and BMI but not sex were independent predictors of hyperglycemia. 40 Conversely, an Ethiopian HIV study reported men had about 4.3-fold higher odds of diabetes than women. 47 These discrepancies likely reflect variations in study design, sample size, glycemic definitions, and population characteristics. Nonetheless, our adjusted analysis suggests that male PLWH on DTG-based ART may represent a higher-risk group for hyperglycemia.
The observed association between overweight and hyperglycemia in our study is biologically plausible. Excess adiposity promotes insulin resistance via inflammatory adipokines and dysregulated glucose metabolism.48,49 This mirrors some PLWH data; for example, a large Asia–Pacific cohort found BMI > 25 kg/m2 significantly raised incident high fasting glucose. 49 Our finding is also in agreement with the findings of related studies in Central and Eastern Uganda24,25 and Kenya 50 that found significantly higher odds of hyperglycemia in overweight participants in comparison to normal weight participants. However, not all studies agree. In a Ugandan study among PLWH on DTG-based ART by Ankunda et al, BMI was not an independent predictor of new hyperglycemia after multivariable adjustment. 22 Notably that study found long-term DTG use (≥2 years) sharply increased hyperglycemia risk, 22 suggesting a direct drug effect. Discrepant findings likely reflect differences in design and outcomes: our study defined hyperglycemia FPG ≥110 mg/dL, whereas the study by Ankunda et al defined hyperglycemia by random glucose (RBS ≥ 7.0 mmol/L). 22 Clinically, our results support vigilant glucose monitoring and weight management in overweight PLWH on DTG-based ART, consistent with evidence linking adiposity to diabetes risk48,49 and recent recommendations for routine glycemic screening in this setting. 51
Abnormal WHR was also independently associated to hyperglycemia in our study. This is biologically plausible: elevated WHR reflects visceral fat accumulation, which promotes insulin resistance via inflammatory cytokines and ectopic lipid spillover. 52 Indeed, integrase inhibitors like DTG are known to induce pronounced weight gain often centrally compared with older ART, 52 potentially worsening insulin resistance. Clinically, DTG-treated PLWH with abdominal obesity warrant vigilant glycemic monitoring. These findings echo other studies. For example, Agete et al found high WHR strongly predicted hyperglycemia (aOR 2.6) and diabetes (aOR 4.1) in Ethiopian PLWH 53 ; notably, they saw no independent effect of DTG itself on glucose levels, 53 implying adiposity was the culprit. In contrast, Mengistu et al reported that only older age, higher BMI and TGs but not WHR predicted hyperglycemia in DTG-treated PLWH. 39 Such discrepancies may reflect differences in cohort or methods: for instance, Mengistu et al defined hyperglycemia as fasting glucose ≥110 mg/dL54 capturing milder dysglycemia. Similarly, Duguma et al found obesity by BMI strongly predicted diabetes in HIV patients (aOR 9.6), 55 underscoring that overall adiposity drives dysglycemia. Differences between studies likely stem from variation in body-fat patterns, ART exposure and glycemia definitions, but collectively support a mechanistic link between visceral fat and hyperglycemia in PLWH. Although hypercholesterolemia (TC ≥ 200 mg/dL) and hypertriglyceridemia (TG ≥ 150 mg/dL) were observed to be positively associated with hyperglycemia among PLWH on DTG-based ART, consistent with previous studies,56,57 these associations were not statistically significant after multivariable adjustment, suggesting potential confounding by other metabolic factors or limited statistical power to detect independent effects.
Conclusion
The prevalence of hyperglycemia among PLWH on DTG-based ART is relatively high. Male gender, overweight, and waist-to-hip ratio were significantly associated with hyperglycemia among PLWH on DTG-based ART.
Recommendation
We recommend for routine screening for hyperglycemia during initiation and follow up of PLWH on DTG-based ART especially among males, those with abnormal weight to hip ratio, and BMI above 25 kg/m2.
Limitations of the Study
The study had a few limitations that should be considered in the interpretation of results. A single FPG level was taken to measure glycemic status. This measurement may not be representative of long-term glycemic states of the patients. This was a cross-sectional study and therefore whether participants became hyperglycemic after acquisition of the observed significant risk factors was not clear. Therefore, our study was limited by the inability to conclude on the causal relationship between the significant factors identified and the subsequent development of hyperglycemia. The study may also have been limited by a possibility of recall bias during interviews.
Footnotes
Acknowledgments
The authors extend our profound gratitude to the Almighty God, whose guidance and strength enabled us to undertake and successfully complete this research. Our sincere appreciation goes to our esteemed mentors and research supervisors: Mr Simon Peter Rugera, Ms Ritah Kiconco, and Dr Edward Kikabi. Their unwavering guidance and mentorship provided invaluable support from the inception to the culmination of our research project. The authors would like to express our gratitude to the dedicated lecturers in the Department of Medical Laboratory Science, whose expertise and guidance played a pivotal role in shaping our professional journey. Special thanks are due to the District Health Officer of Kyankwanzi District, the In-charge of Ntwentwe Health Centre IV, the In-charge of the ART clinic, and the entire laboratory and ART clinic staff at Ntwetwe Health Centre IV. Their technical support was instrumental throughout the data collection period. The authors would like to express gratitude to Mbarara University of Science and Technology for the opportunity to conduct this research, which forms a substantial component of the Master's dissertation in Medical Laboratory Science. The insights gained from this work have significantly contributed to the academic journey and the attainment of advanced qualifications.
Ethical Approval and Informed Consent
Ethical clearance was obtained from the Research Ethics Committee (REC) of Mbarara University of Science and Technology (MUST) (REC number: MUST-2023-943, Date: 13/9/2023). District administrative permission was sought from the Kyankwanzi District Health office. In addition, the PI also had to seek site administrative permission from the In-charge of Ntwetwe HC IV. All participants provided written informed consent prior to enrollment. To ensure comprehension, the consent forms were translated into the local language, Luganda. Participants with formal education provided consent by signing the forms, whereas for those without formal education, the consent form was read aloud in Luganda, followed by administration of a comprehension screening tool approved by MUST-REC. Only participants who demonstrated understanding were allowed to provide consent via thumbprint. Participants were also explicitly asked for consent to allow their data to be used for secondary analysis, which they approved. This study was conducted in accordance with the Declaration of Helsinki (1975). Participant confidentiality was maintained by assigning study codes that were not traceable to individuals, and all personal identifiers were removed to protect privacy. All laboratory results, including fasting plasma glucose, total cholesterol, triglycerides, alanine aminotransferase, and aspartate aminotransferase, were documented in the participants’ medical records at the ART clinic. Participants identified with elevated fasting plasma glucose (≥110 mg/dL) were promptly communicated to the attending clinicians for appropriate clinical action.
Author Contributions
Conceptualization, methodology and investigations, formal analysis, resources, data analysis, and result interpretation: Charles Nkubi Bagenda; Translation of study tools: Elastus Ssemwanga; Writing—original draft preparation: Charles Nkubi Bagenda, Carol Nantongo, Sylivia Achieng Lumumba, Michael Junior Mugisa, and Elastus Ssemwanga; Writing—review and editing: Charles Nkubi Bagenda, Carol Nantongo, Elastus Ssemwanga, Sylivia Achieng Lumumba, Michael Junior Mugisa, Edward Kikabi, Ritah Kiconco, and Simon Peter Rugera; Supervision and project administration: Ritah Kiconco, Edward Kikabi, and Simon Peter Rugera. All authors have read and agreed to the publication of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data used in this study are available from the corresponding author upon request.


