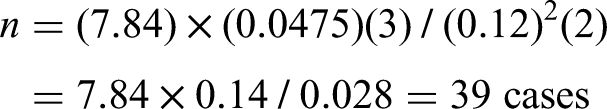Abstract
Background
Dolutegravir (DTG)-based antiretroviral treatment is now the recommended regimen because of its high efficacy and fewer adverse effects. Nonetheless, hyperglycemia as adverse effect of DTG was reported in few clinical observations.
Methods
A case-control study was carried out among DTG-based antiretroviral therapy (ART) users during the study period. EPI Info version 4.8 and SPSS version 26 were used for data entry and analysis, respectively. Binary logistic regression model was used to determine association between risk factors and outcome measures. The associated factors of hyperglycemia were identified using the odds ratio. A P-value <.05 was used to test significance.
Results
This study contained 42 cases (DTG-based ART users who developed hyperglycemia) and 84 controls (DTG-based ART users who didn’t develop hyperglycemia). On bivariable analysis, lower base-line CD4 count, greater body mass index, and lower grade school attendees were significant at P-value <.25. On multivariable analysis, overweight/obesity (body mass index ≥25) plausibly approached statistical significance but did not reach conventional threshold (P-value<.05) as risk factor for hyperglycemia among DTG-based ART users.
Conclusion
Overweight/obesity-driven hyperglycemia secondary to insulin resistance might be the explanation for hyperglycemia among DTG-based ART users. Further studies with larger sample sizes and prospective designs are needed to confirm these findings.
High blood sugar level was found to be a side effect of dolutegravir-based antiretroviral medication. weight gain-driven hyperglycemia might be the reasonable explanation for its cause among DTG users.
Keywords
Introduction
Antiretroviral therapy (ART) has brought a significant impact in reduction of HIV-related morbidity and mortality, and transformed the infection into almost manageable disease. A combination of at least 3 ART drugs from 2 different classes is required to suppress viral replication and restore immune function. ART regimen consisted of 2 NRTIs in combination with a third drug which can be an NNRTI, PI, or INSTI.1,2 INSTIs act by inhibiting the catalytic activity of HIV integrase enzyme, an HIV encoded enzyme required for viral replication. Dolutegravir (DTG)-based ART regimen has some unique characteristics including superior efficacy, un-boosted daily dosing, tolerability, high barrier to drug resistance and treatment durability compared with the existing first line regimens.2–4 WHO and Ethiopian ART guidelines recommend the use of tenofovir/lamivudine/DTG (TDF/3TC/DTG) as the preferred first-line regimen for adults.3,5 Recently, a few case reports and observational studies have raised concerns about potential adverse events, such as hyperglycemia following the initiation of DTG.6–8 Documented risk factors of hyperglycemia in ART-experienced patients include HIV infection itself, high viral load, low CD4 count, prolonged duration of HIV infection, and long-term exposure to ART. Other factors contributing for hyperglycemia include advanced age, male gender, high body mass index (BMI), low socioeconomic class, and cigarette smoking.9–15 Determinants of hyperglycemia among PLHIV taking DTG-based ART regimen were not adequately explored in Ethiopia. The aim of the study was to determine the associated factors of hyperglycemia among DTG-based ART users in the study locality.
Methods
Study Settings
The ART Clinic situated in University of Gondar hospital, carried out an institutional-based case-control study from June 1, 2022, to November 30, 2022. The hospital is located in Northwest Ethiopia, 748 km from Addis Ababa, the country's capital. The ART Clinic provided outpatient medical services for patients with HIV infection. The clinic was staffed with internists, medical residents, medical practitioners, and unit Nurses. The ART Clinic was established in 2003. A total of 6000 clients started ART since its establishment. Free access to ART in the clinic was launched in 2005.
Study Population and Study Subjects
The study population was 18 years and older people living with HIV (PLHIV) adults who were taking DTG-based ART regimen in the ART clinic, University of Gondar hospital from 1 June, 2022 to 30 November, 2022. The study subjects were 18 years and older PLHIV who were taking DTG-based ART regimen for at least 3 months at the ART clinic, University of Gondar hospital, during the study period.
Inclusion Criteria
Eighteen years and older PLHIV adults who were taking DTG-based ART regimen for at least 3 months at the ART clinic, University of Gondar hospital, during the study period.
Exclusion Criteria
PLHIV adults who were pregnant during the study period, and who were diagnosed to have diabetes before commencement of the study were excluded from the study.
Sample Size and Sampling Technique
Sample Size
The sample size was chosen from a study done in Uganda, considering one variable assumed to bring difference in the 2 groups. Assuming 2-sided confidence level (CI) = 95%, Power = 80%, ratio of cases to controls = 1:2, hypertension was found to be the significant risk factor for hyperglycemia (OR of 4.0 and proportion of exposure among controls was 5%) which gave a sample size of 41 cases and 82 controls including 5% contingency for nonresponders.
16
P1 = proportions of exposure in cases; P0 = proportion of exposure in controls
P1 = 0.05(4)/ 1 + 0.05(3) = 0.2/1.15 = 0.17
d = P1 − P0 = 0.17-0.05 = 0.12
Sampling Technique
We used consecutive sampling technique. Cases were selected based on their fasting blood sugar (FBS) level, and controls were recruited for each case in a ratio of 2:1.
Study Variables
Dependent variable: Hyperglycemia among patients on DTG-based ART regimen
Independent variable: Sociodemographic characteristics (age, gender, residence, marital status, occupation, and educational status), clinical and behavioral factors (BMI, comorbidities, family history of diabetes, smoking history, and regular exercise), and HIV-related factors (duration of HIV since diagnosis, baseline CD4 count, WHO clinical stage at diagnosis, initial ART regimen, and duration of DTG-based ART regimen).
Data Collection Instruments and Procedures
Data were gathered using semistructured and pretested questionnaires. The questionnaire was pretested in English and translated into local language (Amharic) for data collection, then retranslated back to English with maintaining its consistency. The questionnaire had been pretested on 5% of PLHIV in a similar setup before the actual data collection was commenced to check for consistency and reliability of the questionnaire. As risk factors of hyperglycemia among DTG-based ART users, the questionnaires were designed with sociodemographic data and information on HIV infection, antiretroviral therapy, and behavioral factors. The medical records to be reviewed were identified by their medical registration/card numbers. Then, data collectors reviewed and extracted data from patient medical records and registries using a check list. Base line FBS level was determined at the time of switch to DTG-based ART regimen. The recent FBS level was determined during data collection procedure with finger-prick morning blood glucose determination with blood glucose meter (Model No. XR-RAK-668; Hangzhou Xinrui Medical Device Co., Ltd, China). A total of 3 health professionals, 2 ART nurses as data collectors and 1 medical resident as supervisor, were recruited for the data collection process.
Statistical Analysis
EPI Info version 4.8 (Epi Info, Atlanta, USA) was used for data entry, and SPSS version 26 (SPSS Inc., Chicago, USA) was used to analyze it. Patient characteristics were presented as median with interquartile range for continuous variables and counts (percentages) for categorical variables. Multicollinearity was checked between independent variables through variance inflation factor for continuous independent variables and spearman's rank correlation for categorical independent variables. The association between independent and dependent variables was determined using a binary logistic regression analysis. Variables associated with hyperglycemia among DTG-based ART users with a P-value < .25 in the bivariable analysis were further analyzed using multivariable analysis to control for potential confounding factors. The model goodness of fit was tested using Hosmer and Lemeshow's test, which yielded a value of 0.61. Crude odds ratio and adjusted odds ratio were reported. A P-value <.05 was used to test significance.
Ethical Considerations
The Institutional Review Board (IRB) of the College of Medicine and Health Sciences, University of Gondar, granted ethical approval for a study protocol that adhered with the Declaration of Helsinki (17/04/2022; IRB No. 1656/2022). Only after obtaining written informed consent were study participants recruited. Every piece of information was handled with confidentiality. Patients who were diagnosed with hyperglycemia during the data collection period were treated in accordance with ADA guidelines. 17
Definition of Terms
Hyperglycemia: FBS level ≥ 100 mg/dL on 2 occasions in 2 consecutive days.
Normal base line glucose level: FBS < 100 mg/dL (17)
Prediabetes: FBS level of ≥ 100 mg/dL and <126 mg/dL (17)
Diabetes mellitus: FBS level of ≥ 126 mg/dL (17)
Cases: PLHIV adults taking first line DTG-based ART regimen, who developed hyperglycemia.
Controls: PLHIV adults taking first line DTG-based ART regimen, who didn’t develop hyperglycemia.
Results
Hundred and fifty individuals living with HIV were approached at the ART clinic, University of Gondar hospital during the study period. Of them, 126 patients (42 cases and 84 controls) met the requirements for inclusion. The remaining 24 individuals were excluded because of incomplete data or missing baseline serum FBS levels (Figure 1).

Recruitment of Study Participants.
Sociodemographic Characteristics
The median (IQR) age of patients enrolled in this study was 44 (28-60) years. Majority of study subjects were females (76%), attended formal education (75%), and were urban dwellers (87%) (Table 1).
Sociodemographic Characteristics of Patients Taking First Line DTG-based ART at University of Gondar Hospital, 2022.

DTG, dolutegravir; ART, antiretroviral therapy.
*Student, daily laborers.
Patient and Behavioral Characteristics
Following the start of a DTG-based ART treatment, 7 patients (17%) developed diabetes (FBS ≥126 mg/dL), and 35 patients (83%) had impaired FBS level = 100-125 mg/dL). One-third (36%) among the cases, and one-fifth (21%) among the controls had BMI of ≥ 25 kg/m2. Almost all study subjects in both groups did not have a history of cigarette smoking or metabolic disorders (Table 2).
Patient Characteristics and Behavioral Factors of Patients Taking First-line DTG-based ART at University of Gondar Hospital, 2022.

DTG, dolutegravir; ART, antiretroviral therapy; DM, diabetes mellitus; BMI, body mass index.
*Hypertension, ** chronic kidney diseases.
HIV-Related Characteristics
In both groups, the majority of study participants (87%) had been diagnosed with HIV infection for at least 5 years (91% of cases and 86% of controls). Prior to starting ART, half of the cases (52%) and one-third of the controls (38%) had advanced HIV infection (baseline CD4 count < 200 cells/mm3). WHO clinical stages III and IV were present in nearly half (42%) of the cases and one-third (35%) of the controls. Before switching to a DTG-based regimen, the majority of patients in both groups were first initiated on a TDF/3TC/EFV regimen (43% of cases and 65% of controls). In both groups, the majority of patients (79% of cases and 81% of controls) were on DTG-based ART regimen for at least 24 months (Table 3).
HIV-Related Factors of Patients Taking First-line DTG-based ART at University of Gondar Hospital, 2022.

DTG, dolutegravir; ART, antiretroviral therapy; TDF, tenofovir; 3TC, lamivudine.
Factors Associated With Hyperglycemia Among Patients on DTG-Based ART Regimen
Binary logistic regression analysis revealed that advanced immunosuppression (baseline CD4 count <200/mm3), greater body mass index (BMI ≥25 kg/m2) and lower grade school attendees were significant at P-value <.25. On multivariable logistic regression analysis, BMI ≥25 (OR: 2.257, CI [0.964-5.281]; P = .061) and lower grade school attendees (OR: 4.265, CI [1.020-17.850], P = .057) approached statistical significance but didn’t reach conventional threshold (P-value<.05) as risk factors for DTG-induced hyperglycemia (Table 4).
Binary Logistic Regression Analysis of Patients Taking First-line DTG-based ART at University of Gondar Hospital, 2022.

COR, crude odds ratio; AOR, adjusted odds ratio; DTG, dolutegravir; ART, antiretroviral therapy; BMI, body mass index.
Discussion
DTG-based ART regimen is approved by the WHO and National guideline for the treatment of HIV infection in combination with NRTIs.2,3 Regrettably, there are few case reports and observational studies suggesting concerns on new onset hyperglycemia after initiation of DTG-based ART regimen. To mention few of the case reports, 4 cases of HIV-infected patients from western world developed new-onset diabetes after 3 weeks to 2 years of DTG-based ART use, who were initially on NNRTIs- or PI-based regimens. In all cases, hyperglycemia was controlled after discontinuation of DTG.8,18,19 Similar case reports were reported from Ethiopia. Six cases of HIV-infected patients developed new-onset diabetes after switch to DTG-based ART regimen. Glycemic control in all of them was achieved with insulin and metformin while on DTG-based regimen.20,21 Likewise, hyperglycemia as a side effect of DTG was reported from clinical trials including VIKING-3, SPRING-2, SAILING and SINGLE trials. In our case-control study, overweight/obesity (BMI ≥25) plausibly approached statistical significance but didn’t reach conventional threshold as risk factor for DTG-induced hyperglycemia. Similarly, ADVANCE and NAMSAL trials reported that raised fasting glucose, high blood pressure and dyslipidemia were associated with DTG use, likely driven by obesity. 14 Also, a cohort study (NA-ACCORD) in United States revealed that INSTIs-based ART regimen conferred greater risk of diabetes mellitus, likely mediated through weight gain. 15 Higher incidence of new onset diabetes mellitus among adults receiving DTG-based ART regimen were reported from various studies.16,22,23 To the contrary, few studies specified that no association between incidence of new onset diabetes and DTG-based ART regimen.24–27 Overweight/obesity-driven hyperglycemia secondary to insulin resistance might be the explanation for hyperglycemia among DTG-based ART users.14,15
Limitation of the Study
It was a single center hospital-based study, and conclusion couldn’t be drawn for the general population. Since the study type was a case-control study, it might call up on to do a large-scale prospective study to determine the actual cause of hyperglycemia.
Conclusion
Overweight/obesity-driven hyperglycemia secondary to insulin resistance might be the explanation for hyperglycemia among DTG-based ART users. Further studies with larger sample sizes and prospective designs are needed to confirm these findings.
Supplemental Material
sj-doc-1-jia-10.1177_23259582251375873 - Supplemental material for Determinants of Hyperglycemia in Adult People Living With HIV Taking Dolutegravir- Based Antiretroviral Therapy at University of Gondar Hospital, North West Ethiopia, 2022: A Case-Control Study
Supplemental material, sj-doc-1-jia-10.1177_23259582251375873 for Determinants of Hyperglycemia in Adult People Living With HIV Taking Dolutegravir- Based Antiretroviral Therapy at University of Gondar Hospital, North West Ethiopia, 2022: A Case-Control Study by Addisu Liknaw, Abilo Tadesse, Workagegnehu Hailu, Tsebaot Tesfaye and Melaku Tadesse in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251375873 - Supplemental material for Determinants of Hyperglycemia in Adult People Living With HIV Taking Dolutegravir- Based Antiretroviral Therapy at University of Gondar Hospital, North West Ethiopia, 2022: A Case-Control Study
Supplemental material, sj-docx-2-jia-10.1177_23259582251375873 for Determinants of Hyperglycemia in Adult People Living With HIV Taking Dolutegravir- Based Antiretroviral Therapy at University of Gondar Hospital, North West Ethiopia, 2022: A Case-Control Study by Addisu Liknaw, Abilo Tadesse, Workagegnehu Hailu, Tsebaot Tesfaye and Melaku Tadesse in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgments
We are grateful to the study participants and their health personnel.
Ethics Approval and Consent to Participate
Ethical approval was obtained from the Institutional Review Board of College of Medicine and Health Sciences, University of Gondar (17/04/2022; IRB No. 1656/2022). Formal letter of permission was obtained from the University of Gondar hospital administrative body. Study subjects were recruited only after written informed consent was obtained. Every piece of information was handled with confidentiality.
Consent for Publication
Written informed consent for publication was obtained from study subjects.
Authors’ Contributions
AL contributed to the conception, design, data collection, analysis, writing, and review of the manuscript. AT contributed to the conception, design, analysis, writing, and review of the manuscript. WH, TT, and MT contributed to conception, design, analysis, and review of the manuscript. All authors read and approved the final manuscript and approved its submission for publication.
Funding
Funding for research was obtained from the “Research and Publication Office” of College of Medicine and Health Sciences, University of Gondar. The funding body had no role in the design of the study, data collection, analysis and interpretation of the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data generated and analyzed were included in this research article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.