Abstract
Background
Despite global declines in HIV incidence, new infections continue to rise in Latin America. Oral tenofovir-based HIV pre-exposure prophylaxis (PrEP) is effective and can reduce incidence where implemented. PrEP PERU is a prospective cohort study evaluating daily oral PrEP delivery at HIV/AIDS service organizations (ASOs) in Peru.
Methods
We assessed 12-month PrEP retention and adherence among men who have sex with men (MSM) enrolled at 4 ASOs in Lima before the COVID-19 pandemic. The analysis included participants with ≥12 months of follow-up before the March 2020 lockdown. Follow-up visits occurred at weeks 4, 12, and quarterly thereafter. We used robust Poisson regression to evaluate associations between baseline characteristics and 2 outcomes: retention (attending ≥3 follow-up visits within 12 months) and optimal adherence (proportion of days covered ≥80%).
Results
Among 264 MSM who initiated PrEP, median age was 31 years (IQR: 27-37). Retention at 12 months was 71%, and 55% achieved optimal adherence. Retention was associated with age ≥30 and bisexual identity in adjusted models. Optimal adherence was associated with being employed at baseline.
Conclusions
PrEP delivery through ASOs in Lima is feasible and supports sustained engagement among MSM. Targeted strategies are needed to improve outcomes among younger individuals.
Plain Language Summary Title
PrEP can be successfully provided through community HIV organizations in Lima, Peru
Plain Language Summary
HIV is still a major health problem in Latin America, and new infections are rising in some countries. One proven way to prevent HIV is pre-exposure prophylaxis, or PrEP. PrEP is a daily pill that works best when taken regularly.
This study looked at whether PrEP could be given effectively through community-based HIV/AIDS service organizations in Lima, Peru. We followed 264 gay, bisexual, and other men who have sex with men who started PrEP before the COVID-19 pandemic. Participants had follow-up visits after 1 month, 3 months, and then every 3 months. We included only those who had at least 12 months of follow-up before March 2020.
After one year, about 7 in 10 people (71%) kept attending follow-up visits, and about 1 in 2 people (55%) had PrEP available for at least 80% of days. People aged 30 or older and those identifying as bisexual were more likely to stay in care, while those with jobs were more likely to take PrEP regularly. Younger men were less likely to stay on PrEP.
These results show that community HIV organizations can successfully provide PrEP in Lima. To help more people benefit, programs should offer extra support for younger men to make it easier to attend visits and take PrEP every day. Expanding community-based PrEP could help lower HIV infections in Peru and similar countries.
Keywords
Introduction
In Peru, the HIV epidemic is concentrated among men who have sex with men (MSM) and transgender women (TGW). While HIV prevalence remains below 1% in the general population, it is estimated at 10% among MSM and over 30% among TGW.1,2 Since 2010, the annual number of new HIV infections in Peru has increased by an estimated 81%, 2 outpacing regional trends in Latin America. 3 Reversing these trends will require HIV prevention strategies that effectively engage MSM and TGW through human-centered implementation approaches that are responsive to community needs, build trust, and reduce structural and interpersonal barriers to care.
Daily oral pre-exposure prophylaxis (PrEP) with tenofovir disoproxil fumarate plus emtricitabine (TDF/FTC) is a proven HIV prevention modality that is central to the global HIV response,4–7 with demonstrated reductions in HIV incidence where broadly implemented.8–12 In Peru, TDF/FTC was authorized for use as PrEP in 2016 but remained largely inaccessible outside of research studies for several years. In June 2023, the Ministry of Health adopted national HIV prevention guidelines enabling provision of free oral PrEP at public sector clinics. 13 Before this time, fewer than 3000 people had initiated PrEP in Peru, mostly through demonstration studies. 14 ImPrEP, the largest such study in Latin America to date, evaluated daily oral TDF/FTC use among MSM and TGW in Brazil, Mexico, and Peru, including 2293 participants across 10 public clinics in 6 Peruvian cities from 2018 to 2021. 15 Peruvian MSM in ImPrEP were more likely to discontinue PrEP early, exhibited lower adherence, and were less likely to persist in PrEP use compared to their Brazilian counterparts. 15 These findings highlight unique challenges faced by users in the country's public clinic system, including stigma from healthcare providers, limited provider training, lack of trust in health institutions, and systemic inefficiencies.16,17 HIV/AIDS service organizations (ASOs) have a long-standing history of community engagement, research, and service provision with MSM in Peru. Their trusted relationships and person-centered approaches make them well-positioned to complement public sector efforts and reach populations underserved by the public system. Additional data from ASOs are needed to inform PrEP scale-up efforts in Peru and the region.
In 2017, the PrEP PERU demonstration study was launched to evaluate daily oral PrEP implementation at ASOs serving MSM and TGW. This initial report presents 12-month PrEP retention and adherence outcomes prior to the COVID-19 pandemic among MSM enrolled in the PrEP PERU cohort.
Methods
Study Design and Procedures of the PrEP PERU Demonstration Project
PrEP PERU is a longitudinal, prospective, multicenter demonstration project evaluating daily oral HIV PrEP implementation at ASOs in Peru. The study was implemented at 4 ASOs in Lima, Peru, selected for their established clinical infrastructure, experience with HIV prevention research, strong ties to MSM and TGW communities, and complementary distribution across Metropolitan Lima
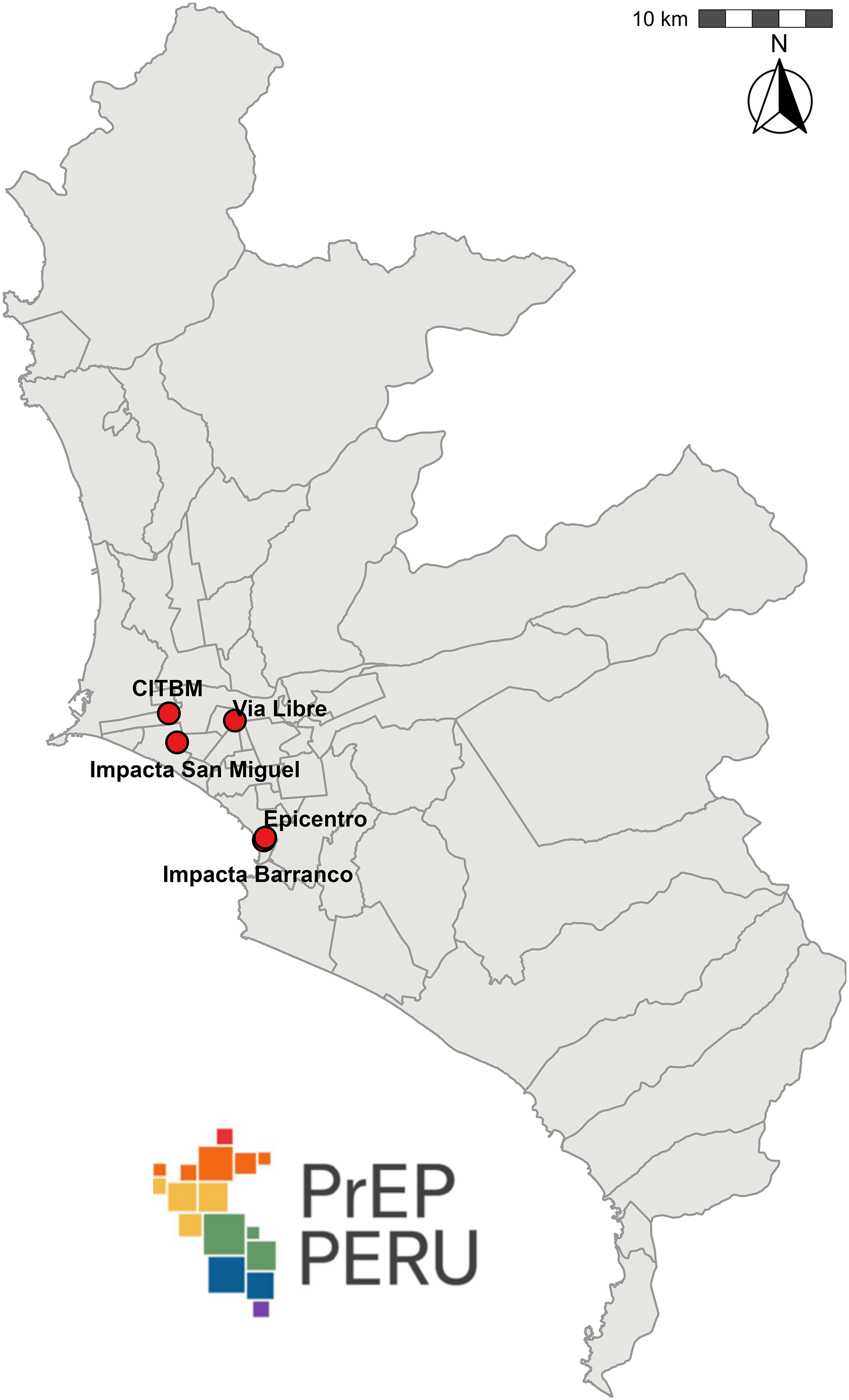
Distribution of HIV/AIDS service organizations participating in the PrEP PERU demonstration project across Metropolitan Lima. Source: CDC Perú (2025). Site coordinates provided by the author.
The study followed a pragmatic implementation model designed to reflect real-world PrEP delivery through ASOs. At enrollment, participants were dispensed a 30-day supply of TDF/FTC. Follow-up visits occurred at week 4, week 12, and then quarterly, with medication refills provided based on the time until the next scheduled visit. In general, participants received a 90-day supply of TDF/FTC at quarterly visits; however, this was ultimately at the discretion of the prescribing clinician, who could reduce the refill duration if deemed medically appropriate. In line with contemporary guidelines, laboratory testing was required at enrollment and follow-up.18,19 At baseline, HIV testing (rapid or laboratory-based), hepatitis B surface antigen, rapid plasma reagin (RPR), and serum creatinine were required, with repeat HIV, RPR, and serum creatinine testing every 3 months thereafter. However, the study did not conduct or cover the cost of laboratory tests. Participants were responsible for obtaining laboratory results independently and presenting them at each quarterly visit, including proof of a recent negative HIV test, in order to receive a new PrEP dispensation. A small clinic fee was also paid by participants at each visit. TDF/FTC was dispensed at no cost to participants, as it was donated by the manufacturer.
Data Collection
Baseline and follow-up data were collected using computer-assisted self-interview (CASI) questionnaires administered in Spanish. Participants received a brief tutorial on the CASI platform at enrolment, and study staff were available at each visit to assist with questionnaire completion as needed. No participant was excluded based on literacy level or prior computer experience. Responses were collected electronically and stored under anonymized participant codes to ensure confidentiality. At enrollment, participants completed a comprehensive baseline questionnaire that assessed sociodemographic characteristics (age, gender identity, sexual orientation, education, employment, income, housing, and health insurance), HIV testing history, prior PrEP use, and concerns about acquiring HIV. Participants reported recent STI symptoms and provided detailed information on sexual behavior over the past 30 days. Sexual partners were categorized as stable, casual, one-night, or transactional, and for each type, participants reported the number of partners, perceived or known HIV status, sexual positioning (insertive, receptive, or both), and condom use.
Alcohol use was measured using items from the alcohol use disorders identification test (AUDIT), supplemented with questions about alcohol use during sex, beliefs about alcohol-related sexual risk, and perceptions of alcohol use in sexual partners. Participants also reported recreational drug use and the venues where they met sexual partners, distinguishing between physical settings (eg, bars, clubs, public spaces) and online platforms.
Follow-up questionnaires were administered quarterly beginning at week 12 and included updates on sociodemographic factors, STI symptoms, substance use, and sexual behavior. Participants were asked about changes in sexual practices, including condom use and number of partners, to assess potential risk compensation. Adherence modules captured self-reported missed doses over the past 30 days, reasons for non-adherence (eg, forgetting, travel, side effects, perceived low risk), and self-efficacy in daily PrEP use. Participants also reported use of adherence strategies such as alarms or pillboxes, experiences with PrEP sharing, and barriers or facilitators to continued use, including structural or psychosocial factors.
Analysis Population and Design
This analysis presents 12-month outcomes and associated baseline factors among MSM enrolled at 4 ASOs in Lima from study inception until the emergence of the COVID-19 pandemic. To ensure complete and uninterrupted follow-up, we restricted the analytic sample to participants who initiated PrEP at least 12 months before the onset of Peru's national lockdown on March 15, 2020—that is, those enrolled before March 15, 2019. This approach minimized the risk of bias due to pandemic-related service disruptions, which included strict mobility restrictions and suspension of study operations at all sites. 20 Due to limited TGW enrollment, we restricted this analysis to cisgender MSM.
Outcomes
We evaluated 2 outcomes at 12 months: (1) retention in care, defined as attending ≥3 follow-up visits within 12 months of enrollment; and (2) optimal adherence, defined as a proportion of days covered (PDC) ≥ 80%. PDC was calculated using pharmacy records to estimate the number of days participants had TDF/FTC in their possession, divided by 366 (the number of days in the 12-month observation period). 21 The use of adherence measures based on pharmacy dispensation data is consistent with other PrEP implementation studies.15,22–24 Although an executed adherence of ≥60% (equivalent to taking at least 4 doses of TDF/FTC per week) has been shown to confer effective HIV prevention among MSM, we selected a more conservative PDC cut-off of 80%. This threshold has been suggested as more appropriate for medication possession metrics such as PDC, which represent only maximal predicted adherence rather than true executed adherence.22,25 Furthermore, recent empirical data from MSM and TGW in Latin America indicate that a higher medication possession threshold is required to adequately discriminate prevention-effective PrEP use based on blood tenofovir levels. 26
Other Variables of Interest
Baseline variables were derived from enrollment questionnaires. Monthly income was dichotomized using a threshold of 1000 Peruvian Soles (PEN), equivalent to approximately 300 USD as of January 23, 2017. During the study period, the national minimum monthly wage ranged from 850 to 930 PEN.27,28 Harmful alcohol use was defined as an AUDIT score ≥8. 29 Sexual behavior variables were dichotomized based on reported behavior or partner type in the past 30 days; total partner number was categorized as ≥5 versus <5. Concurrent participation in a non-PrEP HIV prevention clinical trial was included as a binary variable. Self-reported adherence at week 12 was dichotomized as 100% (no missed doses) versus <100% (any missed doses in the past 30 days).
Statistical Analysis
Baseline characteristics were summarized using descriptive statistics. Associations between baseline variables and 12-month outcomes (retention in care and optimal adherence) were evaluated using robust Poisson regression models to estimate risk ratios (RR) and 95% confidence intervals (CI). Poisson regression was chosen to improve model stability. We performed bivariable analysis to generate crude estimates, then fit separate multivariable models for each outcome. We excluded harmful alcohol use and self-reported adherence from consideration for multivariable analyses due to high missingness. Participants who reported no alcohol use in the past 30 days were automatically skipped out of the AUDIT section in the CASI, explaining missing data for harmful alcohol use. Missingness in self-reported adherence resulted from attrition before the month 3 visit, when this measure was assessed. All other variables met completeness criteria and were eligible for multivariable model selection. Model selection followed a forward stepwise approach that optimized model fit using the Akaike Information Criterion (AIC). 30 In this approach, covariables are added to the model in a sequential and iterative fashion; at each step, the variable that most improves model fit (based on lowest AIC) is selected for inclusion in the model, continuing until no further improvement in AIC is seen with the addition of remaining variables. Analyses were conducted in Stata version 18 (StataCorp LLC, College Station, TX). We used the STROBE cohort checklist when writing this report. 31
Results
Baseline Sociodemographic, Clinical, and Behavioral Characteristics
From January 23, 2017, to March 15, 2019, a total of 269 individuals enrolled in the PrEP PERU study. Five TGW were excluded due to limited enrollment, yielding an analytic sample of 264 MSM. The median age was 31 years (IQR: 27-37). Most participants identified as homosexual/gay (86%) or bisexual (12%), 79% were employed, and 69% reported monthly income ≥1000 PEN. The majority (86%) resided in metropolitan Lima, including Callao. In the 30 days prior to enrollment, the median number of anal sex partners was 5 (IQR: 2-9); 52% reported at least one anonymous partner, 53% reported condomless anal sex, 36% reported a partner living with HIV, and 27% reported drug use before or during sex. A recent STI was self-reported by 16%, 23% screened positive for harmful alcohol use, 91% had previously tested for HIV, and 9% reported prior PrEP use. Baseline characteristics stratified by concurrent clinical-trial participation are shown in Table 1.
Baseline Socio-Demographic, Clinical, and Sexual Behavioral Characteristics of MSM Enrolled in PrEP PERU Cohort Prior to the COVID-19 Pandemic (N = 264).
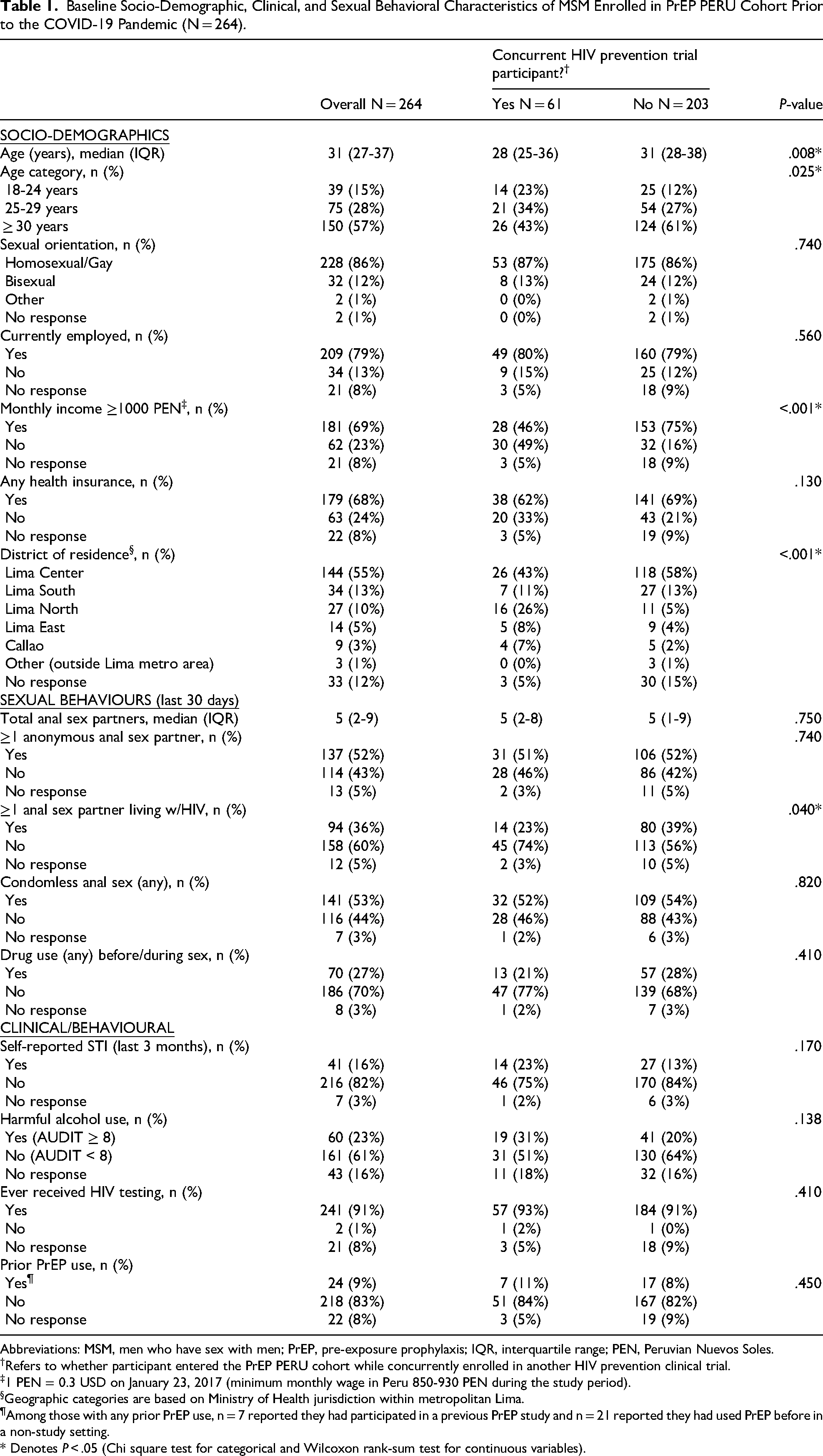
Abbreviations: MSM, men who have sex with men; PrEP, pre-exposure prophylaxis; IQR, interquartile range; PEN, Peruvian Nuevos Soles.
Refers to whether participant entered the PrEP PERU cohort while concurrently enrolled in another HIV prevention clinical trial.
1 PEN = 0.3 USD on January 23, 2017 (minimum monthly wage in Peru 850-930 PEN during the study period).
Geographic categories are based on Ministry of Health jurisdiction within metropolitan Lima.
Among those with any prior PrEP use, n = 7 reported they had participated in a previous PrEP study and n = 21 reported they had used PrEP before in a non-study setting.
* Denotes
Retention in Care
At 12 months, 188 of 264 participants (71%) were retained in care (Table 2). In bivariate analysis, age ≥30 years was the only statistically significant predictor of retention (RR 1.31; 95% CI: 1.11-1.55). In multivariable analysis, retention was positively associated with age ≥30 years (aRR 1.28; 95% CI: 1.06-1.54) and bisexual identity (aRR 1.30; 95% CI: 1.06-1.60).
Factors Associated With 12-Month Retention in Care in the PrEP PERU Demonstration Study, Lima, Peru (2017-2020) (N = 264).
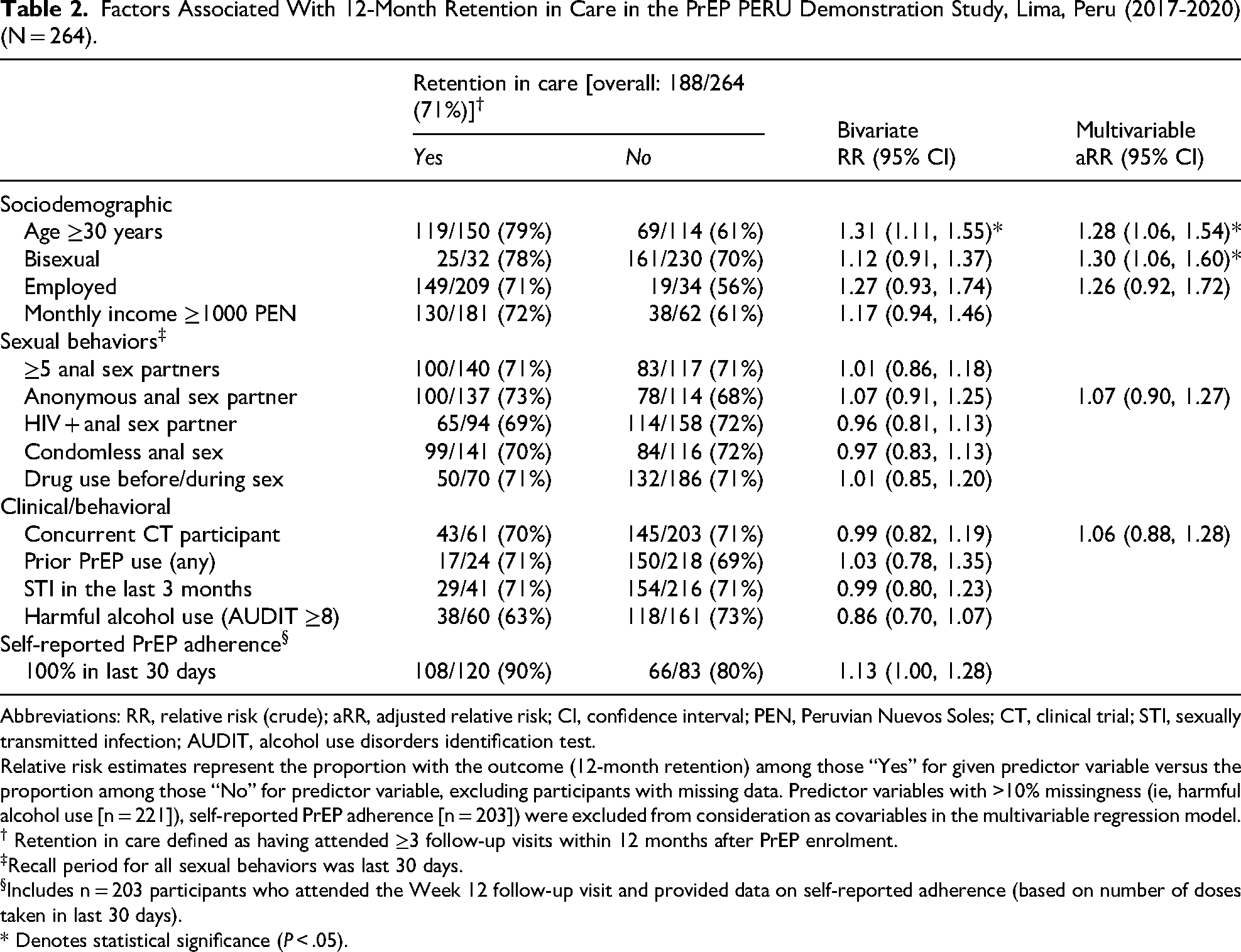
Abbreviations: RR, relative risk (crude); aRR, adjusted relative risk; CI, confidence interval; PEN, Peruvian Nuevos Soles; CT, clinical trial; STI, sexually transmitted infection; AUDIT, alcohol use disorders identification test.
Relative risk estimates represent the proportion with the outcome (12-month retention) among those “Yes” for given predictor variable versus the proportion among those “No” for predictor variable, excluding participants with missing data. Predictor variables with >10% missingness (ie, harmful alcohol use [n = 221]), self-reported PrEP adherence [n = 203]) were excluded from consideration as covariables in the multivariable regression model.
† Retention in care defined as having attended ≥3 follow-up visits within 12 months after PrEP enrolment.
Recall period for all sexual behaviors was last 30 days.
Includes n = 203 participants who attended the Week 12 follow-up visit and provided data on self-reported adherence (based on number of doses taken in last 30 days).
* Denotes statistical significance (
PrEP Adherence
Among 261 participants with available pharmacy data, 144 (55%) achieved optimal adherence, defined as a PDC ≥ 80% during the first 12 months after enrollment (Table 3). The median PDC was 87% (IQR: 48%-98%). In bivariate analyses, optimal adherence was significantly associated with age ≥30 years (RR 1.31; 95% CI: 1.04-1.66), employment at baseline (RR 1.78; 95% CI: 1.08-2.93), and self-reported 100% adherence at week 12 (RR 1.32; 95% CI: 1.06-1.63). In multivariable analysis, only employment remained significantly associated with adherence (aRR 1.68; 95% CI: 1.01-2.77).
Factors Associated With 12-Month Optimal Adherence in the PrEP PERU Demonstration Study, Lima, Peru (2017-2020) (N = 261).
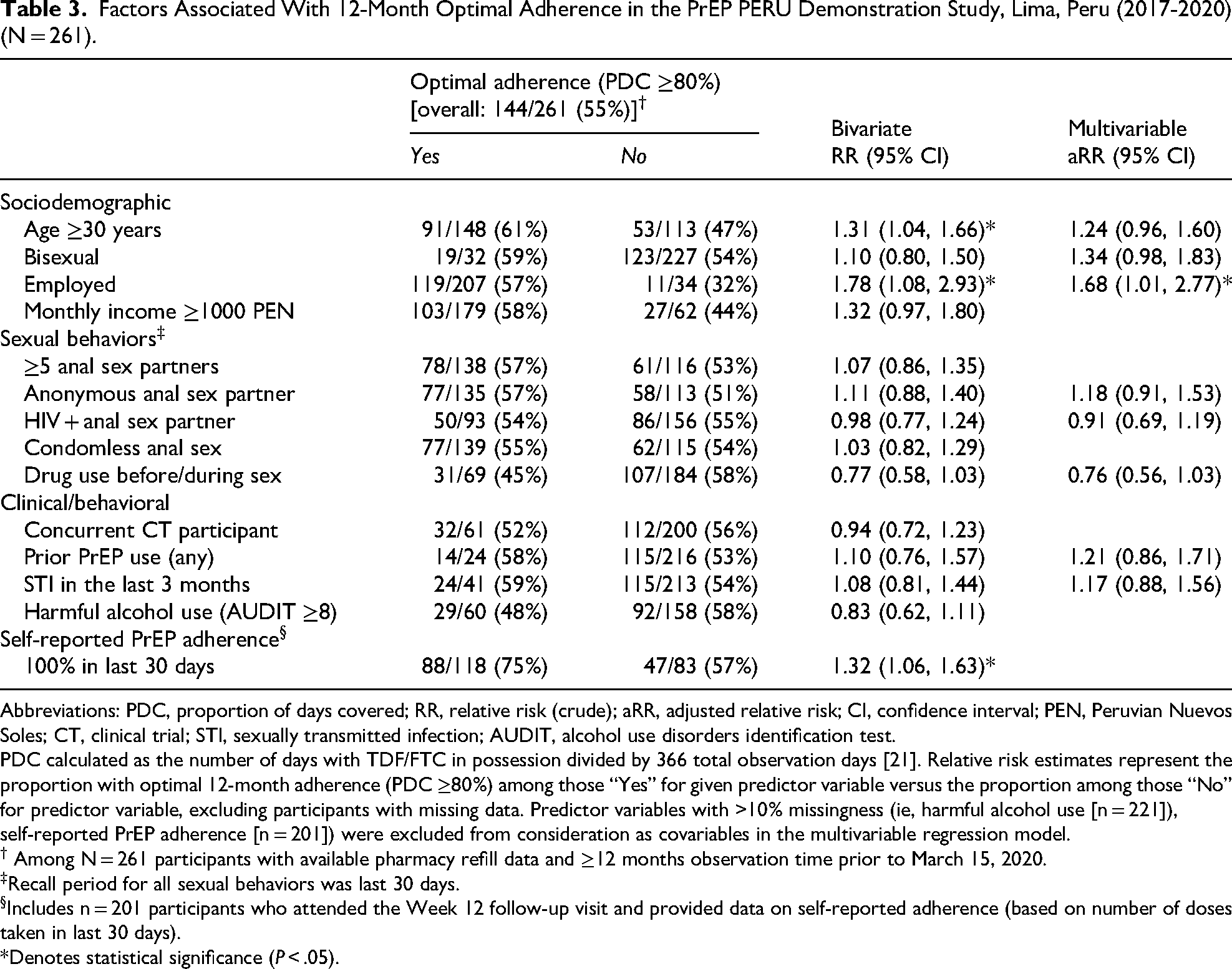
Abbreviations: PDC, proportion of days covered; RR, relative risk (crude); aRR, adjusted relative risk; CI, confidence interval; PEN, Peruvian Nuevos Soles; CT, clinical trial; STI, sexually transmitted infection; AUDIT, alcohol use disorders identification test.
PDC calculated as the number of days with TDF/FTC in possession divided by 366 total observation days [21]. Relative risk estimates represent the proportion with optimal 12-month adherence (PDC ≥80%) among those “Yes” for given predictor variable versus the proportion among those “No” for predictor variable, excluding participants with missing data. Predictor variables with >10% missingness (ie, harmful alcohol use [n = 221]), self-reported PrEP adherence [n = 201]) were excluded from consideration as covariables in the multivariable regression model.
† Among N = 261 participants with available pharmacy refill data and ≥12 months observation time prior to March 15, 2020.
Recall period for all sexual behaviors was last 30 days.
Includes n = 201 participants who attended the Week 12 follow-up visit and provided data on self-reported adherence (based on number of doses taken in last 30 days).
*Denotes statistical significance (
Discussion
This is the first publication from the PrEP PERU study, describing longitudinal outcomes during the pre-pandemic implementation period and providing early evidence on the feasibility of PrEP delivery through ASOs in Lima. In this multicenter study, a high proportion of new PrEP users remained engaged in care during the first year, with modest to high adherence. These results add to prior studies supporting the feasibility of daily oral TDF/FTC as an HIV prevention option for MSM in Peru. Our outcomes are comparable to those reported in other demonstration projects and “real-world” PrEP implementation studies. In a recent meta-analysis including 19 studies of MSM and TGW with up to 12 months of follow-up, the pooled PrEP retention rate was 69.6%, and optimal PrEP adherence was 65.7%. 32 Notably, only 2 studies were conducted in Latin America, a region where PrEP adoption has been slow and the HIV epidemic is growing. 33
Our results provide important evidence of successful PrEP implementation at ASOs in Peru, complementing previous studies in the region. The ImPrEP study, which evaluated PrEP use in Peru during 2018-2022, provides a useful point of comparison. 15 Among Peruvian ImPrEP participants, 12-month retention was 51.8% and 48.4% had high adherence, compared to 71% and 55%, respectively, among PrEP PERU participants, despite our use of a more conservative medication possession threshold to define high adherence (ImPrEP used a medication possession ratio of 60% or greater to define high adherence, compared to the 80% threshold used in our study). Differences in the sociodemographic composition of the 2 study populations may partially explain these differences. ImPrEP was conducted primarily at government health facilities (half of which were located outside of Lima) and, notably, during a period that overlapped with the beginning of the COVID-19 pandemic. The study also included TGW and drew a younger population compared to ours. ImPrEP also recruited individuals who were not actively seeking PrEP, likely resulting in a study population that was less familiar with HIV prevention, on average, than ours. In contrast, PrEP PERU participants were recruited exclusively from sites in metropolitan Lima where HIV prevention counseling and PrEP awareness efforts were already integrated into care, potentially facilitating higher engagement. Nonetheless, consistent with ImPrEP and other recent real-world PrEP studies, 15 34–36 our results highlight younger individuals as particularly vulnerable to PrEP disengagement. These findings contribute to the growing implementation evidence base on PrEP delivery in middle-income settings and highlight the utility of community-based models, such as ASOs, for reaching and engaging key populations.
These results are especially relevant given that oral PrEP remains the most widely available biomedical HIV prevention option in Peru and much of Latin America. While long-acting injectable agents such as cabotegravir and lenacapavir represent promising alternatives, their high cost and complex delivery requirements make them unlikely to be accessible at scale in the near future. 37 In this context, optimizing the use of daily oral PrEP remains a public health priority.
PrEP disengagement among younger MSM reflects broader structural and developmental barriers to care, including stigma, limited autonomy in health decision-making, and inconsistent access to youth-friendly services.16,38 Addressing these disparities will require tailored, youth-centered interventions that are co-designed with affected communities and grounded in local implementation contexts. These results point to the importance of developing approaches that better support PrEP awareness, retention, and adherence among younger MSM in Peru.
This analysis has several limitations. The relatively small sample size may have limited our ability to detect subtle but important differences. Our focus on the pre-pandemic period may reduce the applicability of findings to the current context. While participants were recruited from multiple ASOs, findings may not be generalizable to MSM in other settings, those less engaged with HIV prevention services, or TGW. The fact that participants were responsible for their own laboratory testing may have also selected for a more socioeconomically advantaged population; however, this was probably offset to some extent by the fact that the medication itself was provided free of cost. Importantly, it should be noted that the financial burden borne by participants to access PrEP in this study (namely, the cost of quarterly testing and a small clinic fee, with the medication provided for free) closely resembles the current reality for individuals accessing PrEP at ASOs in Peru. Finally, PDC is only a proxy for true adherence and does not directly measure pill-taking behavior. Notably, self-reported adherence tended to overestimate pill-taking relative to pharmacy refill data, with 57% of participants who had suboptimal PDC reporting 100% adherence. This discrepancy likely reflects recall and social desirability biases, as well as the different timeframes captured by each measure, and is consistent with findings from the regional ImPrEP study, where self-reported and refill-based adherence measures showed only moderate discrimination (AUC ≈ 0.75) of protective drug levels. 26 Nonetheless, metrics based on pharmacy data offer a practical, objective indicator in pragmatic implementation settings. It is also worth acknowledging that the current analysis of factors associated with retention and adherence outcomes focused only on baseline sociodemographic and behavioral characteristics. As a result, our findings may not fully reflect how these barriers and facilitators evolved over time.
As one of the first multicenter studies to evaluate PrEP implementation in Peru prior to national rollout, this analysis also serves as a valuable baseline for assessing program performance over time. As PrEP delivery efforts continue to mature under Peru's national HIV prevention program, 13 these early findings from the PrEP PERU study can inform adaptation and quality improvement strategies to ensure equitable and sustained impact. Our findings also offer valuable guidance for PrEP scale-up efforts in other Latin American countries. Notably, ASOs, given their experience delivering culturally competent, community-based care, can play a key role in expanding access to PrEP and supporting national implementation. To maximize the impact of Peru's PrEP program, future efforts should prioritize reaching young MSM, the population most affected by HIV in Peru, through targeted, innovative communication strategies, including social media, WhatsApp, and mobile apps.39,40 Strengthening the organization and workflow of public clinics, fostering a culture of empathetic, knowledgeable, and affirming care among providers, and investing in sustained community engagement efforts will also be essential to supporting long-term PrEP retention and adherence. Embedding ASO-based models into the national PrEP program could offer a scalable, sustainable approach to improve uptake and continuity of HIV prevention among MSM in Peru and similar Latin American settings.
Footnotes
Acknowledgments
We acknowledge members of the PrEP PERU study group, including the authors of this manuscript and Martín Casapia, Diego Estacio, Gustavo Grandez, José Hidalgo, Juan Hinojosa, Fiorella Huaroto, José Ipanaqué, Narendar Kumar, John MacRae, Milagros Matta, Akemi Matsuno, Luis Mesías, Juan Montenegro, Antonio Narrea, Ivett Pinedo, Joshua Paz, Cesar Sanchez, Milagros Slee, Claudia Tirado, Luis M. Toro, Gabriel Vasquez, and Daniela Zegarra. We thank all study participants for their invaluable contribution. We are also grateful to the community engagement teams and site staff at Asociación Civil Impacta Salud y Educación (Sede Barranco and Sede San Miguel), Asociación Civil Vía Libre, and Epicentro for their dedication and support in the implementation of this study.
ORCID iDs
Ethical Approval
This study was conducted in accordance with the World Medical Association Declaration of Helsinki and was approved by the Institutional Review Boards of Asociación Civil Impacta Salud y Educación (approval no. 0104-2016, approved on April 27, 2016) and the Fred Hutchinson Cancer Center (IR number 10941, approval date: June 19, 2022).
Informed Consent
All participants provided written informed consent prior to enrolment. No incentives were offered for participation.
Author Contributions
PG, JLS, and JRL conceptualized the study. AL led data curation, formal analysis, methodology, and visualization. PR contributed to data curation, formal analysis, and visualization. PG and JGC were responsible for project administration. JGC, JV, PG, JS, HS, and RC contributed to investigation and resource provision. KK and JLS contributed to methodology, with JLS also providing supervision. PR wrote the first draft of the manuscript. AL and JGC led writing—review and editing. All authors contributed to manuscript revision and have read and approved the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
