Abstract
Background
Children with perinatal HIV exposure are at increased risk for neurodevelopmental (ND) delays, yet little is known about ND screening implementation for this population.
Methods
This longitudinal study evaluated ND screening implementation at a health clinic in Kenya, from 9/2021 to 8/2023. Children aged 18-36 months with perinatal HIV exposure were screened using a 12-item general ND tool. Implementation outcomes—acceptability, feasibility, fidelity, and sustainability—were assessed through time-motion observations, clinic records, and semi-structured interviews with caregivers and staff.
Results
Of 507 eligible children, 405 (80%) were screened. Screening rates were consistent over 24 months, with average time reduced to under 5 min. Facilitators included staff collaboration and caregiver support; barriers included time constraints and child temperament.
Conclusions
ND screening was acceptable, feasible, and sustainable. Policymakers should embed ND screening within national child health programs, invest in workforce training and task-sharing models, strengthen referral and follow-up systems, and ensure affordable access to services.
Keywords
Introduction
Nearly 16 million children globally have been born to mothers living with HIV. 1 Even when HIV transmission is successfully prevented, children who are HIV-exposed, but uninfected face higher risks of adverse health outcomes, including neurodevelopmental (ND) delays, compared to their HIV-unexposed peers.2–4 Evidence suggests that early language and motor development may be particularly affected,2,4,5 while older children with perinatal HIV exposure may experience difficulties in executive functioning and school achievement.6,7 These challenges are often compounded by the broader social and structural drivers of health impacting children living in settings where HIV is most prevalent, where high rates of malnutrition, poverty, and chronic disease independently threaten optimal ND. 8 If a generation of children exposed to perinatal HIV fails to reach its full ND potential, the consequences can be far-reaching—limiting their quality of life, academic achievement, and long-term economic potential.9–14 Recognizing this concern, African governments and policymakers are beginning to explore strategies to protect and enhance the ND trajectories of young children as part of broader efforts to invest in national human capital.15,16
Despite growing global recognition of its importance, early ND screening remains rare in many low-resourced settings, particularly in regions with high HIV prevalence. 17 In many cases, routine child health visits are limited to immunizations and weight monitoring, with little additional preventative care offered or accessed beyond the first 12 months of life.18,19 One notable exception is children born to mothers living with HIV, who typically remain engaged in healthcare systems until 18-24 months of age for confirmatory HIV testing. This provides a unique and time-sensitive opportunity to conduct ND screening during a critical window of brain growth when early intervention can have the greatest impact. 20
To leverage this opportunity, we evaluated the implementation of a ND screening program delivered by paraprofessionals within a public maternal-child health (MCH) clinic in western Kenya. We evaluated key implementation outcomes including acceptability, feasibility, fidelity, and sustainability. Ultimately, an integrated and sustainable screening program offers the opportunity to identify children at risk for ND delays and connect them to services—ensuring that children with perinatal HIV exposure not only survive, but thrive.
Methods
Study Design and Period
We conducted a facility-based, longitudinal, mixed-methods implementation study in western Kenya, completed in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology checklist (Supplemental Table 1). The implementation plan was developed in early 2021, and the ND screening pilot program was conducted from September 2021 to August 2023.
Study Setting
The study was conducted at the MCH clinic of Moi Teaching and Referral Hospital (MTRH) in Eldoret, Kenya. The program was implemented within the infrastructure of the Academic Model Providing Access to Healthcare (AMPATH) program, a nearly 35-year partnership consisting of Moi University, MTRH, and a consortium of North American institutions led by Indiana University.21,22 AMPATH supports comprehensive HIV care services across 65 clinics and collaborates with Kenya's Ministry of Health to innovate healthcare delivery within the MCH clinics, including integration of HIV care for pregnant women and their infants.23,24
Study Activities
Development of a Contextualized Implementation Plan
Using purposeful sampling to target clinic staff and community members who were aware of or involved in ND screening, we conducted semi-structure interviews and focus group discussions to inform the design of a locally appropriate ND screening program and understand perspectives on child ND within the community. Participants included community leaders (ie, village elders and chiefs), MCH clinical staff, rehabilitation therapists, child development teachers, and caregivers of young children with perinatal HIV exposure. In total, 20 healthcare workers and community members participated in interviews (n = 10 male, n = 10 female) and 41 caregivers (n = 37 female, n = 4 male) participated in focus group discussions. Mentor mothers—trained peer supporters living with HIV who work in the MCH clinic supporting other mothers—were identified as the most suitable cadre to administer the ND screening. Stakeholders emphasized the importance of a brief screening tool, ideally requiring no more than 5 min to complete. Additional qualitative findings will be reported separately.
Selection of Neurodevelopmental Screening Tools
As part of the broader study (K23MH116808), we previously developed a 12-item ND screening tool suitable for this setting, with input from Kenyan pediatricians and the only pediatric neurologist available within this region at the time of the study. Detailed tool characteristics and its psychometric properties, including reliability and validity, are detailed in a separate manuscript. 25 In brief, it was determined that 2 age-specific tools were needed: one for children ≤24 months of age and another for those >24 months. Some items were administered directly to the child (eg, asking the child to stack blocks to evaluate fine motor skills and receptive language) while others relied on caregiver responses. Additionally, simplicity of items and their scoring was a priority, as the ND screening tool would be administered by paraprofessionals. This study focuses on the process of implementing ND screening, rather than the specific screening tool used, recognizing that tools will continue to evolve and can be replaced as new, evidence-based options become available.
Training of Staff Performing Neurodevelopmental Screening
In August 2021, 6 mentor mothers were trained to administer the ND screening tools. Screening was conducted as part of routine HIV care for children attending their final confirmatory HIV test visit (at ≥18 months or 3 months post-breastfeeding, per WHO guidelines). 26 Refresher training was held every 6 months during the ND screening program pilot during the mentor mothers’ lunch hour. Food was provided during trainings, as is the custom for trainings within this setting.
Eligibility Criteria for Study Participants
Children were eligible for the ND screening program if they met the following criteria: (1) aged 18-36 months; (2) caregiver aged ≥18 years; (3) perinatally exposed to HIV; (4) attended the MTRH MCH clinic; and (5) lived in a household that spoke English or Kiswahili.
Additionally, we performed baseline, 6-month, 12-month, 18-month, and 24-month interviews with the MTRH MCH clinic staff involved with the ND screening program, as well as caregivers who had their child screened in clinic. Eligibility criteria were: (1) > 18 years of age and (2) worked at the MTRH MCH clinic or provided care to a young child who was screened within the MTRH MCH ND screening program.
Pilot of Neurodevelopmental Screening Program
Children included in this study received routine preventative care at the MCH clinic. The MTRH MCH is set up with partitioned stations, where all children initially flow through the weight and nutrition station, then the immunization station, each with a designated paraprofessional. Children with perinatal HIV exposure then transition to the HIV side of clinic, which includes maternal HIV adherence counselling. Nurses would notify mentor mothers of any eligible children prior to reaching the HIV station. Age-appropriate ND screening was then performed by the mentor mothers prior to transitioning to the laboratory for confirmatory HIV testing. Some toys, such as cars, balls, and plush animals, were available around the time of ND screening to help increase child's comfort with screening process.
During the first 12 months of the pilot, all screened children were invited for additional evaluation for a separate study of psychometric properties of the screening tool. In the second 12 months of the pilot, only children who screened positive for possible ND delay were referred for further assessment by 2 trained members of the research team, consisting of a clinical officer (mid-level provider) and a psychologist. Over the course of the 24-pilot period, any child with suspected ND delay were referred to the pediatric neurology clinic at MTRH for evaluation and, if warranted, treatment.
Implementation Outcomes
We adapted Proctor et al's implementation framework to assess 4 key outcome within the ND screening programs: acceptability, feasibility, fidelity, and sustainability. 27 Items from 2 standardized tools, Evidence-Based Practice Attitude Scale (EBPAS) 28 and the Program Sustainability Assessment Tool (PSAT), 29 were repeatedly administered to clinical staff at 6, 12, 18, and 24 months post-implementation to evaluate acceptability and sustainability, respectively. Feasibility, fidelity, and sustainability were evaluated through monthly clinic record reviews and weekly time-motion studies. For time-motion analysis, trained research assistants would randomly select one day each week to observe and record all child encounters within the clinic and the timing required for each station, including ND screening uptake and duration. Timing was rounded to the nearest minute. Clinic records were reviewed for the number of eligible children, screenings completed, and referrals made.
All implementation outcomes were further explored through semi-structured interviews with clinic staff at 5 timepoints (ie, baseline, which occurred within 2 weeks of initiation, and every 6 months after), each lasting approximately 30-45 min. The 6 mentor mothers performing the ND screening were interviewed at each timepoint, and other key clinical staff working with mentor mothers were interviewed at least once. Interview guides are unique for each timepoint and clinic position (clinical provider, mentor mother, or other clinical staff), but main topics involved the interviewee's awareness of the ND screening program and its initial implementation, as well as perceptions of current outcomes, supports, challenges, barriers, and needed improvements for the program. Items from the EBPAS and PSAT were incorporated into the interview guides for ease of completion, such as “to what extent does the program have strong public support?” and “how likely would you be to adopt an intervention that was new to you if it fit with your clinic approach?.” Caregivers with children eligible for ND screening were also recruited for semi-structured interviews during these time-points, but their involvement was cross-sectional. Interview guides for caregivers included questions such as “how knowledgeable or comfortable did you feel answering questions about your child's development?,” “what did you have to do to make sure you and your child were able to return for full evaluation?,” and “how important is child development for you when you think of your own child?” Those interviewed were compensated between 500 and 1000ksh ($4-8USD) for their time and travel required for the interview. Examples of the interview guides used are included in Appendix A in the Supplemental Files.
Statistical Analysis
Quantitative data from standardized tools, chart review, and time-motion analysis were entered and managed using REDCap.30,31 Data categorizing, recategorizing, and cleaning were performed, and the descriptive statistics were calculated, with frequencies and percentages for categorical data, and means and ranges for continuous data. Basic characteristics about the caregivers, including relation to child, level of education, and time and mode of transportation to clinic, were collected during interviews at 12, 18, and 24 months after ND program initiation. With the PSAT and EBPAS were administered to all clinic staff who were interviewed, only the 6 mentor mothers were interviewed at every time point. Thus, to minimize variability and prioritize data from those most involved with ND screening, we report only the PSAT and EBPAS data from the mentor mothers.
Qualitative interviews and focus group discussions were performed in either English or Swahili, audio-recorded, and transcribed into English. The transcripts were analyzed via thematic line-by-line coding by 2 trained research team members to identify emergent themes and triangulated with notes taken by a research assistant during the interview. Saturation was reached within the interviews. Illustrative quotes are reported both within the text and in Table 1 to support findings within both qualitative and quantitative data from the participants’ point of view.
Additional Illustrative Quotes by Time Point.

Ethical Approval and Informed Consent Statements
Ethical approvals were obtained by the Institutional Review Board of the U.S. institution (1807413887), the Institutional Research and Ethics Committee of the Kenyan institution (Reference: IREC/2018/173, Approval Number: 0003108), and the National Commission for Science, Technology and Innovation (NACOSTI) prior to commencement of this study. Written consent was obtained from all adults enrolled in the study. Because ND screening was integrated into clinical care of the MCH clinic and those data were not collected for this study, no consent was obtained on behalf of the children.
Results
During the 24-month study period, 19,861 children attended the MTRH MCH clinic, of whom 1919 were 18-36 months of age, and 506 were born to mothers living with HIV and eligible for ND screening (Figure 1). One-hundred and ninety-nine time-motion observations were performed, of which 50 were children with perinatal HIV exposure and 16 were eligible for ND screening. Forty-two caregivers of children eligible for ND screening participated in interviews over the 24-month pilot period; all had also completed ND screening. Most respondents were mothers of the child (n = 39), with 2 fathers and 1 grandmother included (Table 2). Additionally, 6 mentor mothers, the nurse and assistant nurse in charge (similar to a clinic manager), 2 nutritionists, 2 nurses, and 1 clinical officer participated in qualitative interviews. Representative quotes across time points are presented in Table 1.

Flow diagram.
Caregiver Characteristics.
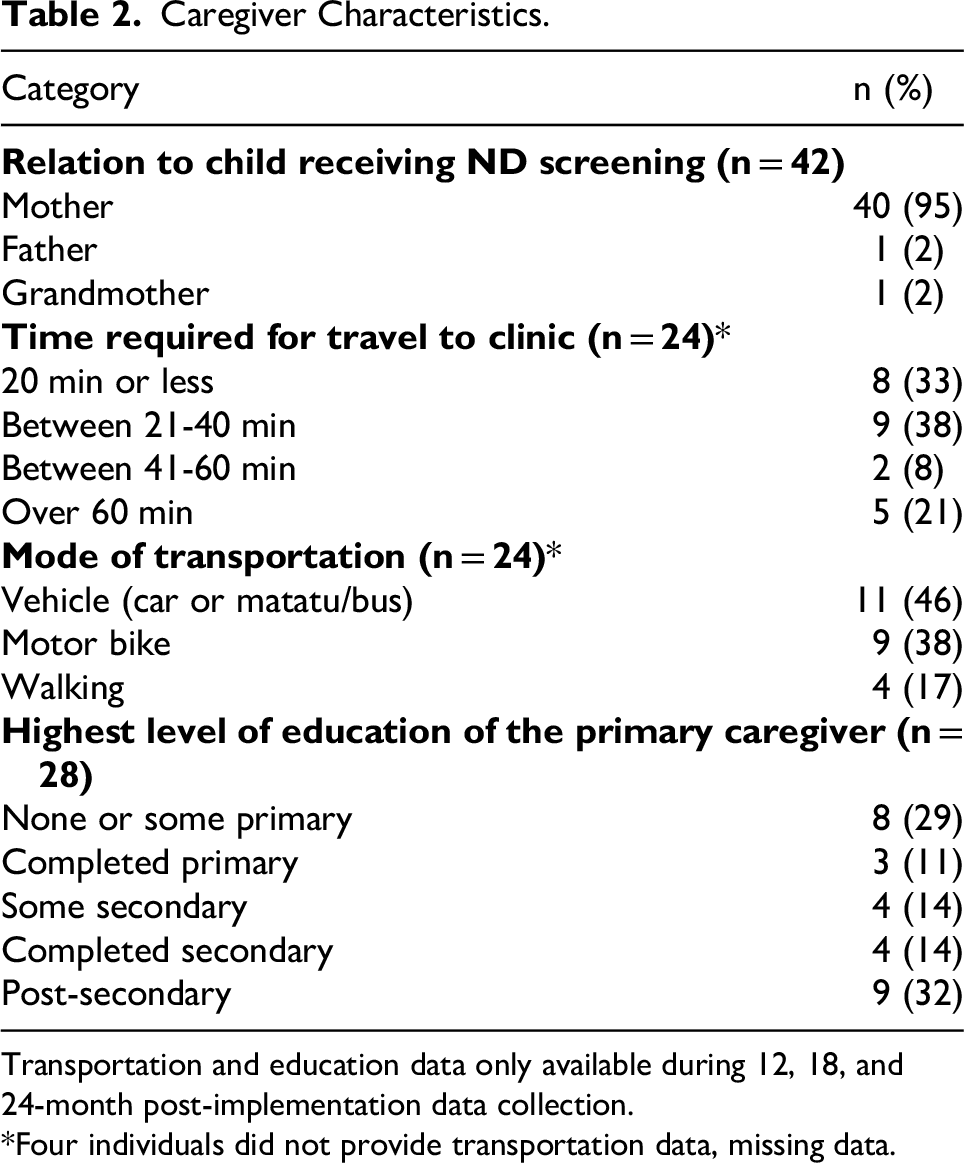
Transportation and education data only available during 12, 18, and 24-month post-implementation data collection.
*Four individuals did not provide transportation data, missing data.
Acceptability
ND screening was consistently described as acceptable to both caregivers and clinic staff. Caregivers and clinical staff expressed that child ND screening was an important and valued part of their clinic experience. Mentor mothers also expressed that performing ND screening helped their clients feel more connected to them and the clinic. Similarly, the caregivers consistently noted they enjoyed the clinic staff showing interest in their child beyond routine care. As one mother noted, “I was so happy because of [the mentor mothers] showing interest to know how my child is developing… I appreciate the clinic is putting more effort to ensure the child is developing well compared to last time I brought my [older] child during testing at 18 months and they didn’t bother to test development then.” One mother, who walked for 90 min to her child's MCH visit, stated: “I care for my child so much, I will ask for permission from work and ensure my child comes… even if it is just for developmental screening, I will still bring her.”
Additionally, ND screening in clinics appeared to encourage ND engagement at home. One mentor mother noted: “We appreciate because the children that we screened, we have opened their parent's eyes. They say when their children were asked the screening questions, they went back home and started playing with the child the way we have been doing.” Caregivers also described learning about ND during the screening process, with one mother noting: “They told me to keep on teaching him to hold things, to pronounce words, to know how to play with other children and to run.”
Six months into implementation of the pilot, both clinic staff and caregivers strongly advocated for expanding ND screening to all children, regardless of HIV status. Some also expressed interest in broadening the eligible age range—from as young as 6 months to as old as 10 years. Clinic staff reported multiple instances in which caregivers of ineligible children actively requested screening, highlighting demand for more inclusive services. One mentor mother noted, “Caregivers are happy about it… Most of the caregivers who visit MCH but are not of the targeted age show interest in developmental screening by asking why their children are not offered the same services.” Additionally, caregivers noted that signaling out their children for ND screening had the potential to increase stigmatization related to their HIV exposure. As one states, “If one is [HIV] positive, there is no need to expose them. [ND screening] should be done in general, whether you are positive or negative.”
While the ND screening program overall was viewed positively, there were a few concerns noted. Two caregivers expressed discomfort immediately after program initiation, as they perceived their children were the only ones receiving ND screening and worried that this implied a pre-existing ND concern. This concern was not repeated in subsequent interviews, possibly related to efforts by the MCH clinic staff to ensure eligible families were informed of ND screening well in advance and reducing surprise at the time of screening. As one mentor mother noted, “Initially we didn’t screen every child who was [eligible] because we didn’t inform them prior to the screening, but for now, we inform them before [coming to our station], which makes it a bit easier to participate.” Additionally, as the program progressed, one mother noted that her prior child had been screened for ND as part of the pilot, so she expected the ND screening to be performed with her subsequent child. Despite this initial concern, caregivers reported trusting mentor mothers and appreciating their respectful approach to performing the ND screening. They found the questions understandable and felt comfortable asking for clarification.
A remaining concern involved affordability of follow-up care. Some caregivers expressed hesitation about receiving a referral without being able to afford therapy. One mentor mother articulated this well: “They say ‘if I went to [the local children's hospital], it would still be the same issue because at the end I will be referred and told to go for physio[therapy]. I cannot pay for that physio myself so it is like there is no help that she got.”
Overall, EBPAS scores indicated the mentor mothers’ willingness to adopt evidence-based practices, and no meaningful trends appeared within the scoring over time. Scores over time are reported in Supplemental Table 2.
Feasibility
Mentor mothers reported that integrating ND screening into clinic flow became increasingly feasible over time, aided by strong clinic-wide support. As one mentor mother explained, “The MCH staff work together for the screening. Every staff in MCH knows our target group and also they recognize the developmental screening is now one of the sections/areas that a child should pass by.” Some expressed that ND screening would ideally occur prior to immunizations, if and when all children receive ND screening, to minimize the timing required to calm the child prior to screening.
Time motion analyses showed that average screening time decreased from approximately 5.2 min in the first 12 months to 4.5 min in the second. One mentor mother noted, “Initially, we thought it could not work because we thought it will take much time but for now, we realized it is working well. It takes only a few minutes. It is easy to perform.” Despite the relatively brief screening time, fewer than half of the caregivers and mentor mothers perceived the screening to take less than 10 min. Caregivers, on average, estimated ND screening to take around 20 min and clinic staff estimated 12 min.
Clinic staff cited long wait times, child temperament, and competing caregiver priorities as barriers to screening. One nurse in charge noted: “Some of them will ask you ‘is it really necessary?’ especially the ones who are in a hurry. This is because they don’t see the need… They get their vaccination; they get their [antiretroviral therapy] and go.” Because the screening was performed after the weight check and immunizations, some children were upset, irritable, or sleeping during the screening time. Clinic staff also noted that the mothers often had to return to jobs or other children, limiting the time they could spend in clinic. A mentor mother mentioned, “The other reason that most mothers tell us is that when they queue for long, time for going back to make lunch catches them up and so they refuse to be screened. You will find a mother saying, ‘I am late to cook lunch for other children, they refuse.’”
Challenges related to time limitations were compounded by language barriers. Some of the children only understood their families’ local tribal language, also known as mother tongue, which required additional time to work with the parents on translation. One mentor mother noted, “We translate to them because most parents in this environment are from the rural areas, so the parent must translate. I mean you tell the parent so that the parent translates to her child. If it is a cup, the parent must tell the child in their language because most parents have taught their children in their mother tongue. When she speaks in mother tongue, the child will understand it quickly.” Another concern identified by the clinic staff, and especially mentor mothers, was the limited privacy available for ND screening. Privacy concerns were largely logistical—relating to child distraction—rather than sensitivity of questions. Of note, none of the caregivers expressed concerns related to lack of privacy.
Fidelity
Among the 506 eligible, 405 (80%) received ND screening. As mentioned, time limitations, child temperament, and competing caregiver priorities were cited as the primary reasons to decline ND screening. The weekly number of screenings remained steady over the 24-month period, ranging from 0 to 5 children/week. Of the 16 time-motion observations performed on children eligible for ND screening, all captured ND screenings that were administered appropriately and completely.
Mentor mothers reported that semi-annual refresher trainings were generally adequate for high-quality administrations of the ND tool. However, some advocated for more frequent meetings to share updates on child referrals, outcomes, and implementation feedback, with one recommending every 2 weeks and another noting every 3 months. The implementation feedback centered on adjusting the timing of ND screening within an MCH visit, the population to target, and the item order within the ND screening tool—all of which were documented for post-pilot consideration.
Sustainability
Clinic staff and mentor mothers expressed strong support for program continuation. At the 24-month interviews, the nurse-in-charge remarked, “What I would say today is that this study has been good to us. It was like an eye opener. You know some things you even don’t think they are very important but when you do the study you open up and you feel like we are supposed to be doing this. So it has really opened up our eyes. So if it shall be integrated we shall be very positive because we have seen the importance [of child development].” The mentor mothers agreed, one noting, “It has become something normal, to screen for development. Stopping will be hard/difficult.”
PSAT scores varied over time (Supplemental Table 3). Perceived leadership support from outside the clinic scored lowest at baseline and again at 24 months, reflecting staff uncertainty about long-term directives. While external leadership engagement improved mid-study, its continuation remained unclear by the pilot's end. Additionally, the extent to which the program had strong public support was scored as relatively lower than other PSAT domains. Both caregivers and clinical staff reported that while they are now aware of the benefits of child ND screening, the public and larger community likely were not. One mother noted, “Some believe that as long as a child is okay health wise, they don’t think that learning is also a part of development.”
A key concern for ND screening raised by clinical staff was the transition of referral responsibilities. During the pilot, the study team handled follow-up referrals, a role not yet formally transferred to a clinical staff member within the hospital system. Staff emphasized the importance of designating a responsible person and providing sustained motivation. One nurse concluded, “I think it will continue because caregivers are positive about, but I am not sure who will do it…[they] need to be motivated, like getting paid incentives.”
Discussion
This study demonstrates the acceptability, feasibility, fidelity, and perceived sustainability of a pilot ND screening program for children born to mothers living with HIV at a busy MCH clinic in western Kenya. Over a 24-month period, 80% of eligible children were screened using a locally adapted tool during routine HIV care visits. We found the screening process to be highly acceptable and feasible, with staff and caregivers expressing strong support. Key facilitators for screening included staff collaboration and caregiver engagement, while barriers included limited time and clinic flow. Staff and caregivers expressed a desire to provide universal ND screening to all children, not just HIV-exposed, and to a greater age range of children. Ultimately, the study highlights that with appropriate adaptation and support, ND screening can be successfully integrated into primary care settings serving high-risk populations.
While the primary goal of ND screening programs was to identify children who may benefit from additional services to optimize their ND, this study suggests that ND screening also offered important benefits beyond identifying those at risk for delays or disabilities. By initiating conversations with caregivers about their child's ND—beyond the routine weight checks and immunizations—the ND screening process helped strengthen relationships between caregivers and clinic staff. This relational benefit is particularly meaningful in the context of HIV, where positive client-provider relationships have been associated with improved adherence to antiretroviral therapies. 32 Additionally, in some cases, ND screening appeared to enhance caregiver knowledge of child ND and encouraged more active engagement in their child's learning and play at home. Similar findings have been observed in high-income settings such as the United States, where implementation of ND screening alone has been associated with trends towards parent-physician communication about child ND and related concerns. 33 This is especially relevant in Kenya, where awareness of early child ND is growing but remains limited. Given the well-documented influence of home environments on child ND, 34 these findings highlight a valuable opportunity to use screening not only as a clinical tool, but also as an entry point for community wide-education and promotion of child ND.
Despite time–motion analyses demonstrating that the average screening lasted less than 5 min, both caregivers and clinic staff substantially overestimated its duration. Prior research has shown that individuals tend to perceive novel or cognitively demanding tasks, involving heightened attention, as lasting longer than those that are familiar or automatic.35–37 Often termed the “oddball” effect, 38 in the clinical context, the novel ND screening process may have felt unfamiliar or high-stakes to caregivers, particularly if they were uncertain about their child's performance or the implications of the results. Such conditions can amplify perceived duration even when the objective task is brief, as subjective time perceptions often rely on imprecise recall and bias regarding past or future events. 39 These findings suggest that the perceived burden of ND screening may extend beyond the minutes required to complete it, reflecting broader aspects of the clinic experience—such as anxiety, waiting, or uncertainty—that shape how caregivers and staff experience the screening process overall.
While many of the concerns raised by caregivers and clinic staff were actionable within the clinic and able to be addressed in further scale-up of ND screening, others reflected broader limitations within the public health and clinical infrastructure. A particularly salient concern involved the financial burden associated with referrals and follow-up services for children identified with potential ND delays. In Kenya, over a third of the population lives below the national poverty line. 40 In the absence of a universal health coverage system, even low-cost services remain inaccessible to many families. Furthermore, although MTRH offers basic rehabilitation services—including occupational, physical, and speech therapy—such services are not consistently available across the country or in other settings with high HIV prevalence. Additionally, during the pilot, members of the research team played a central role in coordinating referrals and ensuring linkage to appropriate services. While this care coordination was valued by both families and clinic staff, it is not currently embedded within the public healthcare system. This gap echoes observations from related efforts in low-resourced settings, where identification of ND delays without accessible referral pathways can lead to caregiver disappointment and non-adherence, as well as reduced program credibility.41,42 The integration of dedicated clinical roles, such as a care coordinator or case manager, could help bridge this gap, ensuring that ND screening translates into meaningful follow-up and support. However, this would require investment and policy-level commitment to strengthen the continuum of care for children with ND concerns. This gap in access highlights a critical barrier to implementation and scale-up of ND screening program in Kenya and other resource-limited settings, where identification without access to affordable follow-up care may lead to frustration or disengagement among families. 43
There remains a limited evidence base for validated screening tools administered by non-specialists in low-resourced settings, particularly regarding implementation research of developmental screening.44–46 This study helps address this critical gap by providing a practical, longitudinal demonstration of a ND screening program in a resource-constrained clinical context, moving beyond tool validation to evaluate real-world implementation outcomes, such as fidelity, fidelity, and sustainability. Prior studies have shown that early intervention programs in low-income communities can be effective when delivered by paraprofessionals such as community health workers. 47 Similarly, integrating ND screening into existing care workflows represents an important opportunity to further enhance reach and impact. Consistent with global evidence emphasizing health system embedding and community engagement as keys to sustainability, 48 our findings reinforce that leveraging existing clinical and human resources to conduct developmental screening can promote program sustainability. 49 While integration of provider-administered ND screening into routine care fostered positive interactions between clinical staff and caregivers, it is essential to consider the long-term sustainability of such programs, particularly in the context of shifting international funding landscapes. To ensure maximum impact with limited resources, future research should explore the cost-effectiveness of ND screening interventions. Technological advancements may offer new opportunities to enhance efficiency. For example, there is growing evidence that routinely collected clinical and demographic data could be used to develop risk prediction models that identify children most vulnerable to poor ND outcomes.50,51 Leveraging such models could help target ND screening and follow-up services more strategically. However, even with more targeted approaches, the cost and availability of follow-up services—such as diagnostic evaluations and early intervention—must again be carefully considered to ensure equitable access and program effectiveness.
This study has several limitations that should be considered when interpreting the findings. First, the pilot was conducted in a single MCH clinic, which may limit the generalizability of the results to other settings with different resources, patient volumes, or staffing structures. Second, nearly all caregiver interview participants were women, with only 2 fathers included. This may have limited our ability to capture perspectives of male caregivers or other family members involved in childcare. However, given that mothers are the primary caregivers of young children in most global contexts, including Kenya, our sample likely reflects the typical caregiving demographic within our study setting. Additionally, while the study collected rich feedback from both caregivers and clinic staff for future use, no formal changes to the screening program were made during the pilot period. A future study will examine the impact of iterative program adaptation. Screening fidelity was assessed through a small number of time-motion observations, and we were unable to independently verify that all mentor mother-led screenings were completed as intended. Additionally, while validated tools such as the PSAT and EBAS were used to assess implementation outcomes, they yielded limited actionable insights in this context. In contrast, in-depth interviews generated the most meaningful data and formed the primary basis of our findings. Finally, we were unable to recruit caregivers whose children were eligible but did not receive ND screening—likely due to initial disinterest or time constraints—introducing potential selection bias and limiting our understanding of barriers to uptake among non-participants. These limitations underscore the need for future implementation research across multiple sites, using a broader range of validated tools and more diverse caregiver populations, to inform scalable approaches to ND screening.
In conclusion, this study provides promising evidence that ND screening can be feasibly and acceptably integrated into routine MCH care for children born to mothers living with HIV in resource-limited settings. Beyond identifying children at risk for ND delays, the screening process fostered stronger client-provider relationships and opened new opportunities for community education and caregiver empowerment around child ND. Notably, both caregivers and clinic staff expressed strong interest in expanding ND screening to include all children—regardless of HIV exposure status—and to cover a broader age range. These findings underscore the perceived value and relevance of ND screening across populations. However, to translate identification into meaningful outcomes, investments are needed to ensure access to follow-up services, care coordination, and workforce capacity. Consideration of cost-effectiveness, technological innovations such as risk prediction models, and sustainable delivery models will be essential to meaningfully scale efforts. With appropriate adaptation and system-level support, ND screening holds the potential not only as a clinical intervention, but also as a catalyst for advancing early childhood ND in high-risk populations.
Conclusions
This study shows that ND screening can be feasibly and acceptably integrated into routine MCH care for children born to mothers living with HIV in resource-limited settings. Beyond identifying children at risk for delays, the screening process strengthened client-provider relationships and created opportunities for community education and caregiver empowerment. Caregivers and clinic staff also expressed strong interest in expanding ND screening to all children—regardless of HIV exposure—and across a wider age range, underscoring its perceived value and relevance. However, to ensure that identification leads to meaningful outcomes, investments are needed to improve access to follow-up services, care coordination, and workforce capacity. Attention to cost-effectiveness, technological innovations such as risk prediction models, and sustainable delivery models will also be essential for scale-up. With thoughtful adaptation and system-level support, ND screening holds the potential not only to improve clinical care, but also to advance early childhood ND in high-risk populations.
Supplemental Material
sj-docx-1-jia-10.1177_23259582251407359 - Supplemental material for Longitudinal Implementation of a Neurodevelopmental Screening Program for Children Born to Mothers Living With HIV in Maternal-Child Clinics in Kenya: A Mixed Methods Study
Supplemental material, sj-docx-1-jia-10.1177_23259582251407359 for Longitudinal Implementation of a Neurodevelopmental Screening Program for Children Born to Mothers Living With HIV in Maternal-Child Clinics in Kenya: A Mixed Methods Study by Megan S. McHenry, Eren Oyungu, Amina Abubakar, Ananda Roselyne Ombitsa, Micaela Gaviola, Cleophas Cherop, Beatrice Kaniaru, Carolyne Jerop, Alan McGuire and Rachel C. Vreeman in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251407359 - Supplemental material for Longitudinal Implementation of a Neurodevelopmental Screening Program for Children Born to Mothers Living With HIV in Maternal-Child Clinics in Kenya: A Mixed Methods Study
Supplemental material, sj-docx-2-jia-10.1177_23259582251407359 for Longitudinal Implementation of a Neurodevelopmental Screening Program for Children Born to Mothers Living With HIV in Maternal-Child Clinics in Kenya: A Mixed Methods Study by Megan S. McHenry, Eren Oyungu, Amina Abubakar, Ananda Roselyne Ombitsa, Micaela Gaviola, Cleophas Cherop, Beatrice Kaniaru, Carolyne Jerop, Alan McGuire and Rachel C. Vreeman in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgments
We would like to acknowledge our study participants, the MTRH MCH clinic staff, and the Uasin Gishu County Children Officer for their time and support for this project.
Ethical Approval and Informed Consent
Ethical approvals were obtained by the Institutional Review Board of Indiana University (1807413887), the Institutional Research and Ethics Committee of Moi University (Reference: IREC/2018/173, Approval Number: 0003108), and the National Commission for Science, Technology and Innovation (NACOSTI) prior to commencement of this study. Written informed consent was obtained from all adults enrolled in the study. Because neurodevelopmental screening was integrated into clinical care of the maternal-child health clinic and those data were not collected for this study, no consent was obtained on behalf of the children.
Author Contributions
MSM conceived the study, led conceptualization, obtained IRB approval, oversaw all aspects of implementation, conducted the analysis, and drafted the manuscript. EO, AA, AN, and RCV served as mentors, guiding MSM throughout the project and providing manuscript review, feedback, and approval. ARO coordinated the study and ensured activities were completed according to plan. MG supported data analysis and contributed to manuscript writing. CC, BK, and CJ conducted data collection, assisted with analysis, and reviewed the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Mental Health (K23MH116808).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
