Abstract
Background
While advancement in treatment have turned HIV into a manageable chronic condition achieving viral suppression, it has become imperative to focus on overall health and improving health-related quality of life of people living with HIV (PLHIV) beyond viral suppression. This study aimed to describe how PLHIV perceive and manage their overall health, factors influencing patient–provider communication, and the impact of poor patient–provider communication on health outcomes.
Methods
This cross-sectional, online survey recruited HIV-positive adults in the United States between February 22 and June 2, 2022, via the Profiles Panel. Data on sociodemographic and health characteristics, and social determinants of health were collected. The study assessed how PLHIV view and manage their overall health by measuring self-rated overall health, Patient Activation Measure® (PAM®), general and HIV-specific health locus of control (HLOC). Multivariable analyses were used to identify the barriers and unmet needs in the patient–primary HIV clinician communication.
Results
Of the 781 PLHIV who completed the study, most participants were cisgender male (56.2%), non-Hispanic, White (51.5%), and <50 years old (67.0%). Over 90% of participants reported being virally suppressed or having an undetectable viral load. About one-third (31.5%) reported their overall health as either poor or fair. Participants were highly activated in managing their health with 58.0% at PAM level 3 and 33.3% at level 4 and had high scores across the dimensions of both general and HIV-specific HLOC. Overall, 18.2% of the participants reported suboptimal satisfaction and 16.8% reported facing difficulty in verbalizing their HIV-related health concerns with their primary HIV clinician. One-fourth of the participants reported “sometimes” or “never/rarely” discussing their HIV treatment medications with their primary HIV clinician. Furthermore, participants dissatisfied with their primary HIV clinician were twice as likely to self-report poor overall health (OR: 2.2, 95% CI: 1.38, 3.48).
Conclusion
This study underscores the critical role of managing overall health by optimizing patient–provider relationships in influencing individual holistic well-being beyond viral suppression. Healthcare interventions should prioritize strategies to enhance patient communication and satisfaction, recognizing its profound impact on HIV and overall health outcomes.
Plain Language Title:
Impact of Poor Patient–Healthcare Provider Communication on Healthcare Outcomes Among People Living With HIV in the United States
Patient and healthcare provider communication (HCP) is important as it affects the overall well-being of people living with HIV. The study highlights that it is important to implement strategies to enhance patient–HCP communication, patient satisfaction, and empowerment to improve health outcomes for people living with HIV.
Introduction
Over the past three decades, there have been notable advancements in the efficacy, safety, tolerability, and convenience of HIV antiretroviral therapy (ART).1–4 With modern ART, the HIV epidemic has transformed from a fatal disease to a chronic condition5,6 specifically for people living with HIV (PLHIV) who are diagnosed early, initiate ART promptly, and adhere to their ART regimen.5,7 Despite these advancements and successful viral suppression in many cases, PLHIV experience lower health-related quality of life compared to the general population, particularly in the areas of anxiety, depression, and HIV-associated stigma, which could negatively impact access to care, engagement with health care providers, and adherence to treatment.8–12
The National HIV/AIDS Strategy Federal Implementation Plan 2022-2025 expanded its scope of indicators of progress to include quality of life measures to address domains such as physical and mental health, and structural factors. 13 These aspects may require managing overall health beyond viral suppression with positive patient engagement.
Understanding how PLHIV perceive and manage their overall health is a crucial step toward identifying key concerns among PLHIV, including unmet needs and barriers in seeking care and gaps in patient–provider interactions. 14 Poor patient–provider communication, including patient dissatisfaction with information shared by healthcare providers (HCPs) can lead to lower mental health and trust issues. 15 This may also lead to patients not sharing all medications they receive or health behaviors with their primary care providers, which could impact adherence to ART and overall health outcomes among PLHIV.16–18 This underscores the need of a holistic approach for effective HIV management that goes beyond medical treatment to improve health-related quality of life for PLHIV by accounting patient perspective and having a positive patient–provider interaction. 19
Patient–provider interaction plays a pivotal role in managing overall health and improving quality of life. 20 Better communication from the HCP (primary HIV clinician) and improved patient health literacy can have a positive impact on HIV care.21–23 Evidence demonstrates that patient–provider engagement enhances ART adherence.24,25 Previous studies have reported gaps in patient–provider communication.19,22,23 The global Positive-Perspectives study reported that PLHIV with salient treatment-related concerns were uncomfortable initiating important discussions with their providers, while many primary HIV clinicians did not proactively initiate these discussions. 19 Factors contributing to this hesitancy among PLHIV included sociocultural conventions, paternalistic model to care, time limitations, perceived indifference from the provider, and expectations of being stigmatized.19,23 While several studies over the past two decades have described the association between poor patient–primary HIV clinician communication, mistrust, and health-related quality of life on a global level,19,22,26–28 these findings were based on aggregated data and did not specifically assess the nuances within individual countries such as the United States (US).
This study describes how PLHIV perceive and manage their overall health and identify unmet needs and gaps in patient–provider communication. It also examined factors influencing patient–provider communication, and the impact of poor communication on health outcomes among PLHIV in the US.
Methods
Study Design, Data Source, and Procedures
This cross-sectional, online survey included HIV-positive adults in the US. The study participants were recruited between February 22, 2022, and June 2, 2022, via the Kantar Profiles Panel and its panel partners. The Kantar Profiles Panel consists of highly validated, permission-based respondents recruited through a pre-existing general-purpose web-based consumer panel via opt-in emails, co-registration with panel partners, e-newsletter campaigns, and online banner placements. All panelists explicitly agree to be a panel member, register with the panel through a unique email address, and completed an in-depth demographic registration profile.
Potential participants were sourced from both a general population panel (maintained by Kantar Profiles) and several targeted panels (ie, panel consists of those panel members known to have HIV). Email invitations were sent to unique email addresses of potential respondents in optimized batches (meaning the invitations did not go out to all eligible respondents in a single day). Panel companies have procedures in place to ensure no duplicate respondents can take survey by tracking the IP address. All data was collected using a programmed web survey. Based on the programming of the survey, out-of-range or implausible responses were not possible. Prior to initiating the survey, appropriate edit programming was conducted to ensure the final dataset required minimal cleaning of invalid responses. These programming procedures for the web-based survey data entry tool included response ranges, consistency checks, skip patterns, and other special edit procedures. Prior to a full launch of the quantitative survey, we issued a soft launch of data collection and paused fielding to check the responses. This served as an additional quality control check to ensure the online survey was operating as intended.
In addition, we implemented several methods to minimize different sources of bias. To minimize selection bias, we used quotas to recruit a demographically diverse sample. To aid in the reduction of non-response bias, we send email reminders to panel members invited to participate in this survey. We informed participants that their survey responses would be kept confidential, which helps to reduce false reporting. Further, the order of response options and statements that were rated on the same scale within the same question were randomized to minimize any order effects. The reporting of this study conforms to the STROBE cross-sectional reporting guidelines (Supplemental Material: STROBE checklist). 29
The recruitment process initially allowed for natural fall-out, with no quotas put in place. However, a quota sample was introduced at the end of March 2022, where only participants who were either <25 years or ≥65 years were permitted to complete the study survey. The study protocol was reviewed by the Institutional Review Board, Pearl IRB, Indianapolis, IN (IRB #21-KANT-269) and received an exemption under FDA 21 CFR 56.104 and DHHS 45 CFR 46.104(b)(2):(2) Tests, Surveys, Interviews regulations. All participants provided written informed consent electronically.
Prior to locking the study database for analysis, quality control checks were conducted to verify the integrity of the data. This included checking for issues such as survey speeding (ie, completing the survey in less than half of the median length of survey completion time) straight-lining (ie, selecting the same response/rating option for every question that uses the same response/rating scale), and illogical responses (eg, duration of HIV is shorter than duration on ART).
Patient Sample
Participants were included in the study if they were ≥18 years of age, capable of reading and writing in English, diagnosed with HIV for ≥6 months and were prescribed medication for HIV treatment, and provided informed consent. Participants were excluded from the analyses if they did not give informed consent or complete the survey. Participants who completed the full survey received compensation for their time in the form of rewards points that are redeemable for gift cards and other rewards.
Study Measures and Outcomes
The questionnaire was developed through review of prior literature (including ATLIS, 30 Millennials and HIV, 31 and Positive Perspectives Study 32 ), multiple work sessions with key stakeholders, review of secondary material for relevance to our intended outcomes, and input from a key thought leader. The questionnaire was further validated through pre-tests with 3 PLHIV via cognitive interviewing techniques. Feedback from these pre-tests was used to further refine survey questions as needed to ensure that all questions, language, and format were clear to the intended patient audience.
The study assessed self-reported sociodemographic characteristics, including age, sex assigned at birth, gender identity, sexual orientation, race/ethnicity, educational level, employment status, insurance status, region, location of residence; social determinants of health (SDOH), including housing instability (ratings of “somewhat” to “extremely worried” about stable housing), food insecurity (ratings of “sometimes” to “always worried” food would run out before having the chance to buy more), and transportation (frequency of putting off or avoiding healthcare visits due to distance and/or transportation issues) (Supplementary Table S1); and health characteristics, including body mass index, smoking status, duration of HIV diagnosis, history of HIV being undetectable or virally suppressed, viral load (VL) status, length of time virally suppressed, non-HIV comorbidity burden, self-reported conditions, history of sexually transmitted infections (STIs), polypharmacy, and primary clinician who manages HIV of PLHIV.
Various measures were used to assess how PLHIV view and manage their overall health (Supplementary Table S1). Self-rated overall health status was categorized into three groups: poor or fair, good, and very good or excellent. Patient activation, which assesses individual's knowledge, skill and confidence for managing health and healthcare, was evaluated using the mean Patient Activation Measure® (PAM®) 33 score (range: 0-100) as well as PAM® levels where participants exhibited varying levels of engagement (level 1 (range: 0-47.0): disengaged and overwhelmed, level 2 (range: 47.1-55.1): becoming aware but still struggling, level 3 (range: 55.2-72.4): taking action and gaining control, and level 4 (range: 72.5-100): maintaining behaviors and pushing further). Additionally, participants’ general health locus of control (HLOC) was assessed using the Multidimensional Health Locus of Control (MHLC) Form B, 34 which consists of three subscales: internal HLOC, the belief that one's health is influenced by their own choices and behaviors; external—powerful others HLOC, the belief that one's health is determined by powerful others; and external—chance HLOC, the belief that one's health is determined by luck, fate, or chance. HIV-specific HLOC was assessed using MHLC Form C, 35 which consists of four subscales: internal HLOC, the belief that one's HIV-related health is influenced by their own choices and behaviors; external—doctor HLOC, the belief that one's HIV-related health is determined by their doctors; external—other people HLOC, the belief that one's HIV-related health is determined by other people; and external—chance HLOC, the belief that one's health is determined by luck, fate, or chance. Participants’ agreement with statements about health and HIV diagnosis (including psychological, social, economic, and empowering impacts) were also measured (Supplementary Table S1).
Barriers and unmet needs in patient–provider communication with the primary clinician who manages HIV (hereafter referred to as “primary HIV clinician”) were assessed by the following measures: (a) difficulty verbalizing health concerns with primary HIV clinician—participants rated the ease or difficulty of discussing four types of health concerns with their primary HIV clinician (5-point rating scale, 1 = extremely easy to 5 = extremely difficult); from these items, a composite difficulty verbalizing health concerns variable was computed (no = rating of extremely easy, somewhat easy, or neutral to all four of the health concern types; yes = rating of extremely or somewhat difficult to at least one health concern type); (b) Satisfaction with primary HIV clinician (5-point rating scale; 1 = not at all satisfied to 5 = extremely satisfied); (c) Rating of agreement about feeling comfortable discussing health options and/or concerns with their clinician (5-point rating scale, 1 = strongly disagree to 5 = strongly agree); (d) Ratings of agreement about perceived judgment of sexual practices and orientation (5-point rating scale, 1 = strongly disagree to 5 = strongly agree); (e) Frequency with which primary HIV clinician monitors weight and weight management activities including diet/nutrition, exercise, and alcohol consumption (5-point rating scale; 1 = never to 5 = always); and (f) Frequency of discussing medications with primary HIV clinician, including prescription medications for treating HIV or all other conditions, and vitamins, supplements, or over-the-counter (OTC) medications (5-point rating scale, 1 = never discuss to 5 = always discuss). For the frequency of discussing medications items, we chose to group “sometimes” with “never/rarely”. Past research has shown that the strongest factor associated with PLHIV perceiving their personal needs were met was the frequency in which their HCP asked them about side effects from their HIV medication coupled with research showing about one-third of PLHIV are uncomfortable discussing treatment-related issues with their HCPs, including concerns about side effects and long-term impacts of ART. 19 Therefore, only sometimes discussing medications PLHIV are taking could indicate a potential gap in care, even among PLHIV who have undetectable VL. See Supplementary Table S1 for more detail on these measures.
Statistical Analysis
Sociodemographic and health characteristics, how PLHIV view and manage overall health, and patient–provider communication gap were summarized using descriptive statistics. Mean and standard deviation (SD) were reported for continuous variables, whereas frequencies and percentages were reported for categorical variables. No imputation strategy was employed for missing data. For variables where there is missing data, the base N is reported and a footnote includes the number of respondents with missing data, along with the reason for exclusion.
Bivariate analyses were conducted to identify factors associated with difficulty in verbalizing health concerns, dissatisfaction with the primary HIV clinician (yes = rating of not at all, slightly, or moderately satisfied vs no = rating of very or extremely satisfied), and poor self-rated overall health (yes = fair or poor vs no = excellent, very good, or good). Independent variables or predictors were selected based on statistical significance and theoretical relevance. Power analyses, specifying a two-tailed t-test for mean differences in two independent samples, α = .05, 80% power, and achieved sample sizes for these outcomes were conducted to understand the detectable effect sizes. With achieved sample sizes of 520 categorized as no and 261 categorized as yes for difficulty in verbalizing health concerns, 649 categorized as no and 142 as yes for poor satisfaction with the primary HIV clinician, and 525 categorized as no and 246 as yes for poor self-rated overall health, there was sufficient sample to detect a small effect size (Cohen's d = .21, .26, and .22, respectively). Multivariable analyses were conducted using logistic regression models (generalized linear models) to identify factors associated with difficulty in verbalizing health concerns and factors associated with poor self-rated overall health. Covariates were selected based on clinical importance and statistically significant differences in the bivariate analyses. Odds ratios (OR) with 95% confidence intervals (CIs) were reported, and P-values < .05, two-tailed, were considered statistically significant. All statistical analyses were conducted using R version 4.3.1 and IBM® SPSS Statistics version 28.
Results
Sociodemographic and Health Characteristics
A total of 2081 participants accessed the survey and 1943 screened for eligibility, of which 781 PLHIV met the eligibility criteria and completed the survey (Supplementary Figure S1). As no responses were flagged for quality control issues, all 781 participants were included in analyses. Over half of the participants were cisgender male (56.2%) and non-Hispanic White (51.5%); mean age was 43.7 years. Most were employed (69.1%), while 41.7% had some college or associate degree.
Overall, 37.1% were diagnosed with HIV ≥10 years ago prior to completing the survey. Nearly 59.0% of the participants reported ≥2 non-HIV comorbidities. Approximately 40.0% of the participants reported taking ≥5 pills/day or currently taking medicines for ≥5 conditions, indicating a high prevalence of polypharmacy (Table 1).
Characteristics of the Study Participants.

N = 1 respondent who reported being insured but did not know which type of insurance was included in the uninsured category.
Excludes n = 129 respondents who did not select “Currently undetectable or virally suppressed” to the question asking about VL status at time of survey completion, including the n = 96 who selected “I don’t know” and the n = 33 who reported their actual viral load, which was <200 copies/mL.
Respondent did not select any comorbidity listed in the survey but could have another condition that was not listed.
Defined as taking ≥5 pills/day or currently taking medicines for ≥5 conditions.
BMI, body mass index; HIV, human immunodeficiency virus; HPV, human papillomavirus; HS, high school; PLHIV, people living with HIV; SDOH, social determinants of health; STIs, sexually transmitted infections.
Patients’ View and Management of Overall Health
About one-third of the participants reported their overall health as either poor or fair. In general, the participants were highly activated in managing their health (mean ± SD PAM score: 69.86 ± 13.25), with 58.0% of the participants at PAM level 3 (taking action and gaining control) and 33.3% at level 4 (maintaining behavior and pushing further) (Table 2).
How PLHIV View and Manage Their Overall Health.

HIV, human immunodeficiency virus; MHLC, Multidimensional Health Locus of Control; PAM, Patient Activation Measure; PLHIV, people living with HIV; SD, standard deviation.
Most participants believed that since their HIV diagnosis, they paid more attention to their health (80.4%), were better able to engage in self-care (67.1%) and gained control of their lives (66.1%). Nevertheless, 57.5% indicated that their mental health has been negatively impacted by their HIV diagnosis, and 55.6% indicated that they kept their HIV diagnosis a secret due to concerns about how others might view them (Table 2).
Barriers and Unmet Needs in Patient–Provider Communication
Overall, 18.2% of the participants reported suboptimal satisfaction (rating of not at all, slightly, or moderately) with their primary HIV clinician (Supplementary Table S2). Nearly a fifth (17%) of the participants reported (somewhat or extreme) difficulty in verbalizing their HIV-related health concerns with their primary HIV clinician (Figure 1A), and 11.1% did not agree that they were comfortable discussing their health options and/or concerns with their physicians (Figure 1B). Moreover, 36.5% perceived that their primary HIV clinician judges their sex practices (Figure 1C). Participants also reported that their primary HIV clinician never or rarely monitored the lifestyle aspects which can contribute to overall health, including weight (11.3%) and weight management activities (diet/nutrition [20.6%], exercise [24.7%], and alcohol consumption [41.0%]) (Figure 1D).
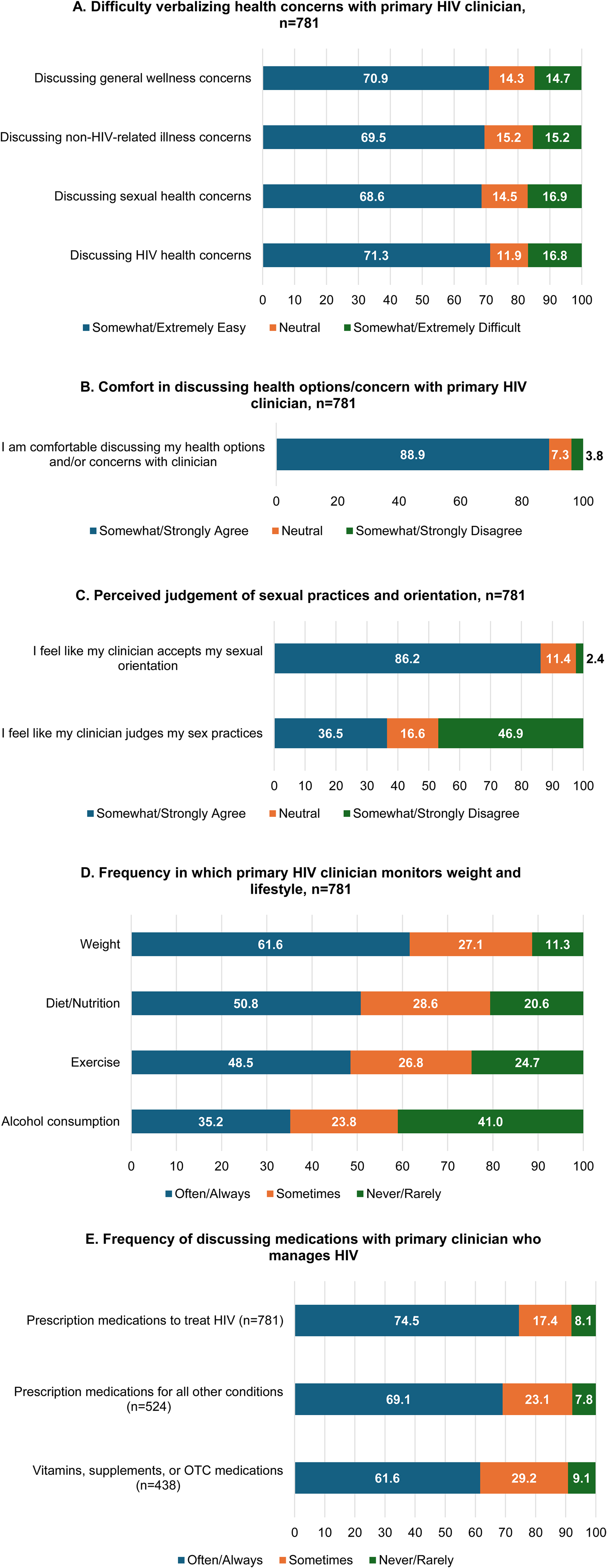
Barriers and unmet needs in communication between PLHIV and primary HIV clinicians. Note: Frequency of discussing prescription medications for all other conditions excludes 257 participants who were not taking a prescription medication for any other health condition, and frequency of discussing vitamins, supplements, or OTC medications excludes 343 participants who were not taking a vitamin, supplement, or OTC medication. Abbreviations: HIV, human immunodeficiency virus; OTC, over-the-counter; PLHIV, people living with HIV.
In addition, one-fourth of the participants (25.4%) reported sometimes or never/rarely discussing their HIV treatment medications with their primary HIV clinician. About 31.0% and 38.3% of the participants reported that they sometimes or never/rarely discussed their medications for non-HIV health conditions and vitamins, supplements, or OTC medications, respectively, with their clinicians (Figure 1E).
Factors Associated With Difficulty Verbalizing Health Concerns With Primary HIV Clinician
In the bivariate analyses, participants who had difficulty verbalizing their health concerns with their primary HIV clinician were more likely to be younger (age <50 years; 83.5% vs 58.7%, P < .001), commercially insured (52.9% vs 37.7%, P < .001), reporting housing instability (73.9% vs 47.3%, P < .001), and food insecurity (70.1% vs 50.8%, P < .001), recently diagnosed with HIV (1 to 5 years ago; 41.4% vs 26.3%, P < .001), and reported suboptimal ART adherence (64.8% vs 30.4%, P < .001) as compared to participants who did not report difficulty in verbalizing health concerns (Table 3).
Factors Associated With Difficulty of Verbalizing Health Concerns With Primary Clinician Who Manages HIV, N = 781.

No = rating of extremely easy, somewhat easy or neutral to all 4 of the health concern areas (general wellness, non-HIV-related illness, sexual health, HIV health); Yes = rating of extremely difficult or somewhat difficult to at least 1 of the health concerns areas.
n = 1 respondent who reported being insured but did not know which type of insurance was included in the uninsured category.
Defined as ≥1 non-HIV comorbidities.
Defined as taking ≥5 pills/day or currently taking medicines for ≥5 conditions.
Somewhat agree + strongly agree.
Neutral + somewhat disagree + strongly disagree.
Indicates statistical significance at P < .05.
BMI, body mass index; EHE, ending the HIV epidemic in the US; HIV, human immunodeficiency virus; MHLC, multidimensional health locus of control; PAM, Patient Activation Measure; SDOH, social determinants of health, SD, standard deviation.
Additionally, PLHIV who had difficulty verbalizing health concerns had higher mean scores on the general and HIV-specific HLOC powerful others/other people (general: 26.9 vs 25.9, P = .008; HIV-specific: 12.7 vs 10.9, P < .001) and chance subscales (general: 25 vs 20.8, P < .001; HIV-specific: 24.8 vs 19.7, P < .001), indicating a higher external locus of control (Table 3).
In the adjusted multivariable analysis, the odds of having difficulty verbalizing health concerns were greater among PLHIV who had worries about stable housing (OR:1.78, 95% Cl: 1.16, 2.74) compared to those who did not, and greater for those diagnosed with HIV between 6 and 12 months ago (OR: 2.17, 95% CI: 1.10, 4.35) and 5 to 10 years ago (OR: 1.83, 95% CI: 1.02, 3.29), compared to those diagnosed >10 years ago (Figure 2).

Multivariable model showing factors associated with difficulty verbalizing health concerns with primary clinician who manages HIV. Note: ORs and 95% CIs from a binary logistic generalized linear model examining factors that are associated with difficulty verbalizing health concerns with the primary clinician who manages HIV. Only factors that were statistically significant (P < .05) are shown in the figure. Age, sex at birth, sexual orientation, race/ethnicity, education, insurance, BMI, smoking status, undetectable viral load, polypharmacy, PAM score, general HLOC (MHLC-B) (internal and powerful others subscales), HIV-specific HLOC (MHLC-C) internal subscale, perceived judgment of sexual practices (I feel my clinician judges my sexual practices), psychological impact (I feel stressed trying to meet all my responsibilities such as my physical health, family and/or work), and empowering impacts of HIV diagnosis (I pay more attention to my health since my HIV diagnosis) were other variables included in the model. Respondents who selected “not listed” for sex at birth were collapsed with the reference group. Respondents who selected “another race” or “prefer not to say” were collapsed with the reference group. Abbreviations: BMI, body mass index; CI, confidence interval; LCL, lower confidence level; HIV, human immunodeficiency virus; HLOC, health locus of control; OR, odds ratio; UCL, lower confidence level.
Factors Associated With Dissatisfaction With Primary HIV Clinician
Participants who reported dissatisfaction with their primary HIV clinician (compared to those who were satisfied) were more likely to be younger (78.9% vs 64.3%, P < .001), less likely to have undetectable VL (78.9% vs 87.0%, P = .024), more likely to have suboptimal HIV medication adherence (52.1% vs 39.6%, P = .008), and had lower scores on the PAM (64.1 vs 71.1, P < .001) (Supplementary Table S3). The participants who reported dissatisfaction with their primary HIV clinician (vs those who were satisfied) were more likely to report having discomfort in discussing health concerns with their primary care clinicians (12.0% vs 2.0%, P < .001) (Supplementary Table S3).
Factors Associated With Poor Overall Health
In the bivariate analysis, participants who self-rated their overall health poorly (vs those who did not self-rate their health as poor) were more likely to report difficulty in verbalizing their health concerns with their primary HIV clinician (38.6% vs 31.1%, P = .037), were dissatisfied with their primary HIV clinician (30.1% vs 12.7%, P < .001), and reported never, rarely, or sometimes discussing medications to treat their HIV (37.0% vs 20.2%, P < .001), or for all other conditions (30.5% vs 16.3%, P < .001), and vitamins, supplements, or other OTC medications (28.9% vs 18.1%, P = .003) with their primary HIV clinician (Supplementary Table S2). Additionally, those reporting poor overall health tended to have lower mean scores for PAM (64.9 vs 72.2, P < .001), general HLOC subscales (internal, 25.8 vs 28.1, P < .001; powerful others, 25.6 vs 26.5, P = .024) and HIV-specific HLOC subscales (internal, 26.1 vs 27.4, P < .001; doctor, 14.72 vs 15.55, P < .001) than those who did not report poor self-rated health (Supplementary Table S2).
In the adjusted multivariable analysis, the odds of reporting poor self-rated overall health were about two times higher (OR: 2.19, 95% CI: 1.38, 3.48, P < .001) among participants who were dissatisfied with their primary HIV clinician, and 1.6 and 2.2 times higher for those with self-reported anxiety (OR: 1.59, 95% CI 1.06, 2.38, P = .025) and hypercholesterolemia (OR: 2.17, 95% CI: 1.31, 3.60, P = .003) as a comorbidity, respectively. Additionally, higher odds of poor self-rated overall health were reported among PLHIV worried about stable housing (OR: 1.81, 95% CI: 1.17, 2.82, P = .009), in those with lower PAM score (OR: 0.98, 95% CI:0.96, 0.99, P = .004), and lower score in general HLOC internal subscale (OR: 0.88, 95% CI: 0.83, 0.93, P < .001) and higher score in general HLOC powerful others subscales (OR: 1.08, 95% CI: 1.03, 1.13, P = .002) (Figure 3).

Multivariable model showing factors associated with poor overall health status (n = 781). Note: ORs and 95% CIs from a binary logistic generalized linear model examining barriers and unmet needs in patient–provider communication and other factors that are associated with poor self-rated overall health. Only factors that were statistically significant (P < .05) are shown in the figure. Difficulty of verbalizing health concerns, never to sometimes discusses HIV prescription medications with primary clinician who manages HIV, never to sometimes discusses other prescription manages with primary clinicians who manages HIV, never to sometimes discusses vitamins, supplements, or OTC medications with primary clinician who manages HIV, individual comorbidities (including cardiovascular disease, depression, gastroesophageal reflux disease (GERD), high blood pressure (hypertension)), polypharmacy, suboptimal adherence, age, sex at birth, sexual orientation, race/ethnicity, worried food would run out, undetectable viral load, and HIV-specific HLOC (MHLC-C) internal and doctors subscales were other variables included in the model. Respondents who selected “not listed” for sex at birth were collapsed with the reference group. Respondents who selected “another race” or “prefer not to say” were collapsed with the reference group. Abbreviations: CI, confidence interval; HIV, human immunodeficiency virus; HLOC, health locus of control; LCL, lower confidence level; OR, odds ratio; PAM, Patient Activation Measure; UCL, upper confidence level.
Discussion
Considering the advancements in HIV ART, which have turned HIV into a manageable chronic condition for PLHIV 36 achieving viral suppression, it becomes imperative to focus on improving their health-related quality of life beyond viral suppression. Factors that impact health-related quality of life in PLHIV could be influenced by interactions with primary HIV clinicians, who should be playing a critical role in identifying and addressing both clinical and health-related quality of life issues beyond HIV treatment. This study assessed how PLHIV view and manage their overall health, factors that influence patient–provider communication, gaps and unmet needs associated with interactions between PLHIV and their primary HIV clinician, and their impact on poor health outcomes.
In this study, despite over 90% of the participants having suppressed or undetectable HIV VL and a majority being highly active in managing their health, about one-third of them reported their current overall health as poor or fair. This finding is notable given that previous studies have demonstrated higher PAM scores in PLHIV are associated with better health outcomes.16,37 The discrepancy between high PAM score and self-reported poor health indicates a gap in addressing broader health concerns beyond viral suppression, underscoring the need for a more comprehensive approach to improving health-related quality of life in this population. Furthermore, over a third of the participants reported having multimorbidity and polypharmacy, highlighting the complexity of managing other aspects of PLHIV that include quality of life beyond the ART care. This also emphasizes the careful choice of ART medications that are less burdensome and have fewer drug-disease interactions. Consistently, more than half of the respondents reported stress, negative impact of their HIV diagnosis on mental health and not disclosing their HIV status due to fear of stigma. Nevertheless, most PLHIV were engaged in their health and self-care due to their HIV diagnosis.
HLOC is a multidimensional tool that improves understanding of PLHIV's perspective on their health. 35 Consistent with previous studies from Iran and the US, participants in this study had high HLOC scores. Specifically, they scored high on internal and powerful other dimensions, indicating that PLHIV believe that their own behavior and that of the powerful others, such as clinicians, influence their health.18,38 Furthermore, the high scores in the powerful others dimension of HLOC could be linked to a positive relationship with trust in physicians, which may in turn enhance patient–provider communication. 38 For HIV-specific HLOC, PLHIV scored highest on the doctors and internal HLOC dimensions, and lowest on the other people (belief that one's HIV is determined by other people) and chance HLOC, aligning with findings from the Italian version of the MHLC-Form C. 35 These observations indicate that PLHIV believed in themselves and their primary HIV clinician in improving their health and well-being.
In the present study, most PLHIV reported positive relationships and satisfaction with their primary HIV clinician. However, nearly 20% reported suboptimal satisfaction, and one-third reported difficulty verbalizing health concerns, including general wellness, non-HIV–related illness, sexual health, and/or HIV health. This finding aligns with a prior study where one-third of PLHIV were uncomfortable in verbalizing health issues with their primary HIV clinician. 19 Additionally, PLHIV who had difficulty verbalizing health concerns were more likely to perceive that their primary HIV clinician judges their sex practices and report a psychological impact of their HIV diagnosis, compared to those without difficulty in verbalizing health concerns. These findings highlight the importance of addressing and treating psychological issues arising from an HIV diagnosis and implies the need for clinicians to ensure they are supportive and accepting of their patients, regardless of their sexual practices. It is possible that these perceptions of feeling judged by clinicians are misperceptions, and thus, it is even more imperative that clinicians be able to break down barriers and establish trust with these individuals.
About one-fourth of PLHIV reported never, rarely, or sometimes discussing their HIV medications with their primary HIV clinicians. Even more PLHIV reported infrequent (never, rarely, or only sometimes) discussions about their prescription medications for other conditions as well as vitamins, supplements, and other OTC medications. Among those who reported never to sometimes discussing their medications, the majority rated these discussions as very to extremely important, highlighting a significant PLHIV-provider communication gap. A similar finding was reported in the 2019 Positive Perspectives study, where at least one-third of PLHIV were uncomfortable discussing the medications, they take with their primary HIV clinicians. 19 These findings highlight the barriers/gaps in patient–provider communication that could impact overall health management. Clinicians may not have a comprehensive understanding of the patient's full treatment regimen, including non-HIV medications and OCT supplements, which could interfere with effective ART care. Hence, improving communication strategies through regular medication reviews and fostering an environment of trust between patients and providers is crucial for effective health management.
Verbalizing health concerns and satisfaction with primary HIV clinicians are crucial for patient-centered care and improving quality of life. 19 In the current study, difficulty verbalizing health concerns with primary HIV clinicians was strongly associated with SDOH factors, including having unstable housing, highlighting the impact of SDOH on the patient–provider relationship and patient engagement in HIV care. 39 Difficulty verbalizing health concerns with their primary HIV clinicians was also strongly associated with being diagnosed with HIV either 6 months to <1 year or between 5 to 10 years, compared to those diagnosed over 10 years prior. Newly diagnosed PLHIV might struggle with care complexities, while those diagnosed 5 to 10 years ago might face ongoing challenges that PLHIV diagnosed >10 years ago do not face as frequently. Of note, lower scores on the doctors’ subscale (belief that doctors do not control their health) of the HIV-specific HLOC indicated greater difficulty in expressing health concerns. This implies that when PLHIV perceive less influence or support from their HIV clinician in managing their condition, they may find it challenging to effectively communicate their health issues.
In this study, younger PLHIV, specifically those under 50 years, had higher odds of reporting dissatisfaction with their primary HIV clinicians, potentially due to generation gap in expectations or communication styles. Research indicates that different generations have distinct preferences and expectations regarding healthcare interactions, which can influence their satisfaction levels. 39 Paradoxically, those with polypharmacy had lower odds of reporting dissatisfaction with their HIV clinicians. Lower PAM scores, reflecting less knowledge and confidence in managing health, were linked to higher dissatisfaction, highlighting the critical role of patient empowerment in the patient–provider relationship. Furthermore, lower scores in the powerful others’ subscale of the general HLOC were associated with greater dissatisfaction with the primary HIV clinician, suggesting that PLHIV who perceive less influence from providers may be more dissatisfied with their HIV management.
In this study, dissatisfaction with a primary HIV clinician, anxiety and hypercholesterolemia, worries about stable housing, and lower college education significantly predicted poor self-rated overall health after adjusting for covariates. Previous reports have discussed the association of several factors such as stress, social isolation, time constraint, and challenges in transportation to clinic, use of treatment and services, and cultural challenges with poor self-rated overall health, a critical indicator of quality of life in PLHIV.19,40 Lower patient activation scores and internal general HLOC (belief that one's health is determined by own actions) scores were also associated with poor self-rated health. Conversely, higher powerful others HLOC scores, indicating greater belief that one's health is determined by other people (eg, HCPs), were associated with poor self-rated health. In other words, believing that one's health is determined by powerful others may have a positive influence on the patient–provider relationship but may also be detrimental to one's overall health; it is possible that those having high powerful others HLOC may have a lower tendency to take responsibility for one's health and instead blame others for their poor health.
Overall, these findings emphasize the importance of addressing both clinical and psychosocial aspects of HIV care to mitigate communication barriers and improve patient satisfaction with their care as well as the care provider. Interventions aimed at enhancing patient–provider communication, and promoting patient empowerment can also lead to better health outcomes for PLHIV.
This study has some limitations to be considered. Selection bias cannot be ruled out as this was an online survey and may have excluded patients who were institutionalized or did not have internet access, as well as those who were reluctant to declare their HIV status online. Furthermore, individuals who agreed to participate may be characteristically different from those who decided not to participate. The cross-sectional design restricts the study's ability to establish causality. Also, the participant-reported nature of the study may have introduced bias due to inaccurate reporting or recall bias as HIV diagnosis, diagnosis of comorbidities, and testing for STIs were not confirmed by a healthcare professional. Additionally, the study's findings may lack generalizability as the study included participants with high patient activation levels, potentially limiting the representation of those with lower patient activation levels. Finally, as our study was exploratory in nature, we acknowledge that our study was not grounded in or guided by a theoretical model. Future research may benefit from using a theoretical model to guide the research objectives and develop hypotheses to be tested.
Conclusion
This study underscores the critical role of managing overall health by optimizing patient–provider relationships in influencing individual holistic well-being beyond viral suppression. Healthcare interventions should prioritize strategies to enhance patient communication and satisfaction, recognizing its profound impact on HIV and overall health outcomes.
Supplemental Material
sj-pdf-1-jia-10.1177_23259582251359440 - Supplemental material for Moving Beyond Viral Suppression: Poor Patient–Provider Communication and Its Impact on Healthcare Outcomes Among People Living With HIV in the United States
Supplemental material, sj-pdf-1-jia-10.1177_23259582251359440 for Moving Beyond Viral Suppression: Poor Patient–Provider Communication and Its Impact on Healthcare Outcomes Among People Living With HIV in the United States by Bekana K. Tadese, M. Janelle Cambron-Mellott, Jean Marie Arduino, Bridget L. Balkaran, Shakiba Eslamimehr and José M. Zuniga in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251359440 - Supplemental material for Moving Beyond Viral Suppression: Poor Patient–Provider Communication and Its Impact on Healthcare Outcomes Among People Living With HIV in the United States
Supplemental material, sj-docx-2-jia-10.1177_23259582251359440 for Moving Beyond Viral Suppression: Poor Patient–Provider Communication and Its Impact on Healthcare Outcomes Among People Living With HIV in the United States by Bekana K. Tadese, M. Janelle Cambron-Mellott, Jean Marie Arduino, Bridget L. Balkaran, Shakiba Eslamimehr and José M. Zuniga in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgements
The authors acknowledge Bryan Ricker and Kristin Falbo, MS, formerly of Oracle Life Sciences, Oracle Corporation for their contributions to study design, execution and project management, and Silki, PhD, and Aiman Lakdawala, MSc CMPP™, of Indegene Pvt. Ltd, India, for their assistance with medical writing.
Ethical Considerations
The study protocol and amendments were reviewed by an Institutional Review Board (Pearl IRB, Indianapolis, IN) and received an exemption under FDA 21 CFR 56.104 and DHHS 45 CFR 46.104(b)(2):(2) Tests, Surveys, Interviews regulations (IRB #21-KANT-269).
Author Contributions
MJCM, JMA, BLB, JMZ did the conception of the study, study design, development/finalization of protocol and survey.
BKT, MJCM, JMA, BLB, SE, JMZ did development of protocol amendments, reviewing and interpreting study results.
MJCM, BLB, SE conducted statistical analyses.
BKT, MJCM, JMA, BLB, SE, JMZ were involved with either drafting and/or providing critical revisions to finalize the manuscript.
All authors have reviewed and approved the final manuscript and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
Declaration of Conflicting Interest
M.J. Cambron-Mellott is an employee of, and B.L. Balkaran, and S. Eslamimehr are former employees of Oracle Life Sciences, Oracle Corporation, which received funding from Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA to conduct and report on the study. M.J. Cambron-Mellott and B.L. Balkaran own stock in Oracle Corporation. José M. Zuniga received consulting fee from Gilead Sciences (Advisory Board) and Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA (research study). Bekana K. Tadese and Jean Marie Arduino are employees of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA, and Jean Marie Arduino owns stock in Merck & Co., Inc., Rahway, NJ, USA.
Data Availability Statement
The data presented, including the methodology, are the intellectual property of Oracle Life Sciences and cannot be shared without an appropriate access request. Results in part were presented at the Continuum 2024 conference June 9-12, 2024, Fajardo, Puerto Rico.
Patient Consent Statement
All patients who participated in this study provided written informed consent electronically prior to accessing the survey.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
