Abstract
In August 2012, a 47-year-old male patient was diagnosed with human immunodeficiency virus (HIV) infection, with no other underlying disease or opportunistic infection. Baseline measurements revealed an HIV RNA count of 79 400 copies/mL and a CD4+ T-lymphocyte count of 8 cells/μL. Despite successful viral suppression with HIV RNA levels remaining below the detectable threshold for 9 consecutive years following the initiation of antiretroviral therapy, the patient's CD4+ T lymphocyte count persistently remained below 200 cells/μL. Resistance testing of the viral reservoir (HIV DNA) was conducted, which revealed proviral resistance. Based on these results, the antiviral treatment regimen was adjusted. One year later, the patient's immune function had significantly improved, with the CD4+ T lymphocyte count having increased to 319 cells/μL.
Introduction
Human immunodeficiency virus (HIV) primarily targets CD4+ T lymphocytes, leading to impaired cellular immune function. When CD4+ T lymphocyte counts below 200 cells/μL, patients become highly vulnerable to opportunistic infections and cancers, which can ultimately lead to death. Early initiation of antiretroviral therapy (ART) is effective in suppressing HIV replication and restoring immune function. However, some patients, known as immunological nonresponders (INRs), fail to achieve adequate immune recovery despite effective viral suppression. Currently, no definitive or highly effective treatment exists for INRs. This study presents a case of an INR whose immune function improved following an adjustment of antiviral therapy based on HIV DNA resistance testing.
Case Description
A 47-year-old male of Han ethnicity was confirmed to be HIV-positive by the CDC in August 2012. He had no prior history of syphilis, hepatitis B, hepatitis C, diabetes, or hypertension. Baseline measurements revealed an HIV RNA count of 79 400 copies/mL and a CD4+ T lymphocyte count of 8 cells/μL. No resistance testing was conducted before the initiation of treatment. Routine blood tests revealed a hemoglobin (HGB) level of 82 g/L and a platelet count (PLT) of 33 × 10^9/L, with other indices within normal ranges. Liver function tests revealed an alanine aminotransferase (ALT) level of 55 U/L, an aspartate aminotransferase (AST) level of 105 U/L, an albumin level of 27.3 g/L, a total bilirubin level of 34.4 μmol/L, and a direct bilirubin level of 18.6 μmol/L. Kidney function tests revealed an estimated glomerular filtration rate (eGFR) of 87 mL/(min·1.73 m2). ECG and chest X-ray results were unremarkable. Abdominal color Doppler ultrasound revealed right hepatic enlargement, increased and coarse hepatic parenchymal echogenicity, increased and enhanced intrahepatic ductal wall echoes, slight widening of the portal vein, normal upper limits for the common bile duct, a rough gallbladder wall, splenomegaly, and no significant abnormalities in the pancreas or kidneys. The patient was diagnosed with AIDS, liver dysfunction, hypoproteinemia, thrombocytopenia, and moderate anemia. Considering that liver dysfunction is associated with anemia, ART was initiated in August 2012 with a regimen of tenofovir (TDF, 0.3 g, Qd), lamivudine (3TC, 0.3 g, Qd), and lopinavir/ritonavir (LPV/r, 500 mg per dose, Q12h). The patient demonstrated good adherence to the medication regimen. Despite successful viral suppression, there was no significant increase in the CD4+ T lymphocyte count. The outpatient follow-up results are shown in Table 1. Over the course of nine years of receiving ART, the patient's HBV and HCV serological tests were both negative, the patient's CD4+ T lymphocyte count remained persistently below 200 cells/μL, with a peak value of only 110 cells/μL. The patient was clinically diagnosed as being a severe INR.
Patient Immune Function Follow-up.
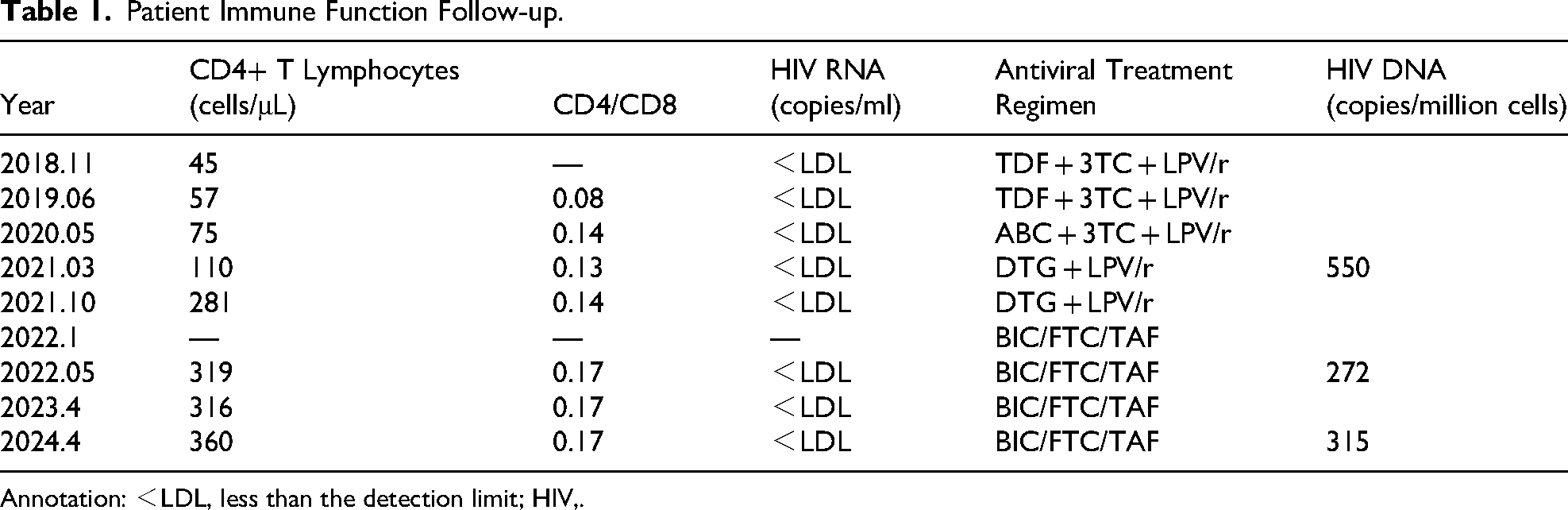
Annotation: <LDL, less than the detection limit; HIV,.
In March 2021, a comprehensive assessment of the HIV viral reservoir (HIV DNA) was conducted. The HIV gene subtype was identified as CRF01-AE, with a viral load of 550 copies per million cells. HIV DNA resistance testing revealed no resistance mutations in the protease or integrase regions. However, nucleoside reverse transcriptase inhibitor (NRTI)-related mutations were identified, including M41ML, D67DN, K70KR, V75VIM, M184V, L210LW, T215FSY, and K219KE. Nonnucleoside reverse transcriptase inhibitor (NNRTI)-related mutations were also identified, including K101KE and G190GA. This indicated resistance to abacavir (ABC), zidovudine (AZT), stavudine (d4T), didanosine (ddI), emtricitabine (FTC), lamivudine (3TC), tenofovir disoproxil fumarate (TDF), doravirine (DOR), efavirenz (EFV), etravirine (ETR), nevirapine (NVP), and rilpivirine (RPV) but sensitivity to integrase inhibitors and protease inhibitors. In March 2021, the HIV treatment regimen was adjusted to lopinavir/ritonavir (LPV/r, 500 mg per dose, Q12h) and dolutegravir (DTG, 50 mg, QD). Six months after starting this new regimen, the patient had a significantly increased CD4+ T lymphocyte count of 281 cells/μL. In January 2022, the patient requested a switch to a more convenient single-tablet regimen. Although the patient had developed resistance to FTC/TAF, sustained virological suppression was achieved after more than 10 years of ART. After the patient provided his informed consent, the HIV treatment regimen was changed to bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF). The outpatient follow-up results for the CD4+ T lymphocyte count and HIV RNA are presented in Table 1.
Ethical Approval and Informed Consent
This study was conducted in accordance with the Declaration of Helsinki, and prior approval was obtained from the institutional review board. Written informed consent was obtained from the patient.
Discussion
In addition to the evaluation of HIV RNA, immune reconstitution is a critical indicator, of the success of ART in HIV-infected individuals. Clinically, immune levels are primarily assessed through CD4+ T-cell counts and the CD4/CD8 ratio. Numerous studies have shown that HIV-infected individuals who are INRs are prone to various non-AIDS-defining events (NEDs), malignancies, and an increased risk of death.1-3 Therefore, achieving robust immune reconstitution is a primary goal of ART. While the majority of HIV-infected individuals experience immune recovery after initiating ART, 10% to 40% of HIV/AIDS patients fail to achieve complete immune reconstitution despite sustained virological suppression, leading to partial immune reconstitution or immunological nonresponse. 4 Factors influencing incomplete immune reconstitution include low baseline CD4+ T-cell counts, older age, low body mass index, comorbidities such as malignancies, and the ART regimen itself.5-7 In addition, there are also reasons related to CD4+ T-cell themselves, such as the imbalance between production, destruction, and migration of CD4+ T cells, the irreversible damage that the CD4+ T-cell pool might have been exposed to, which resulted in the inability to fully reconstitute, the potential effect of defective proviruses expressing viral proteins, which are thought to have pathogenic effects on cells, deficiencies in the regeneration of central memory CD4+ T-cells and excessive apoptosis, increased CD4 and CD8T-cell activation at baseline, and hyperactivated and exhausted CD4+ T-cells in combination with lymphocyte functional impairment.8,9 In this case, the patient was a young male who initiated ART at the age of 35, with no chronic underlying disease. The initial ART regimen included lopinavir/ritonavir (LPV/r), a second-line antiretroviral drug known to be beneficial for CD4+ T-cell recovery compared to NNRTIs. Despite the absence of many factors leading to poor immune reconstitution, the patient's CD4+ T-cell count peaked at only 110 cells/μL, with HIV RNA levels consistently below the detection threshold after 9 years of ART, which is inconsistent with conventional expectations.
Clinicians often find themselves at a loss when dealing with INRs, as there are currently no interventions that are clearly effective at aiding in the recovery of CD4+ T cells. However, the diagnosis and treatment of this case provides new insights: HIV DNA and DNA resistance testing may support the achievement of immune reconstitution. HIV DNA refers to the viral reservoir, which is established early during viral infection. Current ART drugs target HIV RNA but with the success of ART treatment, HIV DNA may undergo changes. Total HIV DNA reflects the overall level in the HIV reservoir in an infected individual, and its load is associated with disease progression and treatment efficacy.10,11 Research by Yue YS et al indicated that ultralow HIV DNA levels were the only factor correlated with higher CD4/CD8 ratios after 96 weeks of ART. 12 Pan et al reported a negative correlation between HIV DNA levels and CD4+ T-cell counts, which is consistent with the findings of other studies.13,14 Low HIV DNA levels have also been used as an important inclusion criterion in studies on functional cures. In this case, after 9 years of ART, the patient was classified as an INR. Comprehensive testing indicated a relatively high level of HIV DNA (550 copies per million cells), and further testing revealed the presence of resistance. Based on the resistance results, the ART regimen was adjusted to include two fully effective drugs with high resistance gene barriers. Six months after the adjustment to the treatment regimen, the patient's CD4+ T-cell count had increased to 281 cells/μL, and after 1 year, it had increased to 319 cells/μL. Compared to before, the mechanism of improved immune function in patients may be due to a decrease in HIV DNA. This therapeutic approach significantly improved the patient's long-standing status as an INR.
In January 2022, the patient consulted the outpatient clinic seeking a single-tablet ART regimen, which would make adherence to the medication regimen more convenient. According to the research results, 50 eligible patients were included in the study. Among them, 46 patients had HIV RNA levels <200, and 4 patients had unsuppressed HIV viral loads (including 2 who had not initiated ART but carried transmissible resistance). All 50 patients had at least one NRTI resistance mutation, specifically M184V, but none had mutations in the integrase resistance site. After switching to Betuximab, changes in HIV RNA levels were observed. With an average follow-up of 18.6 months, 48 patients achieved HIV RNA levels <50 copies/mL. Previous studies have shown that patients with NRTI resistance can maintain high levels of virological suppression even after switching to BIC/FTC/TAF, following long-term successful virological suppression. 15 Therefore, the patient's regimen was changed to BIC/FTC/TAF. Subsequent follow-ups over the next 3 years consistently showed HIV RNA levels below the detection threshold, confirming the feasibility of this smooth transition in treatment. Additionally, the patient's HIV DNA levels further decreased, and the CD4+ T lymphocyte count rose to 360 cells/μL, indicating a significant improvement in immune function. We will continue monitoring these indicators in the future.
Conclusions
In our case, after optimizing the treatment regimen based on the HIV DNA resistance results, the patient's immune function improved significantly. This case provides a new approach to the management of patients who are classified as INRs without conventional risk factors. However, more data are necessary to validate and support this new approach.
Footnotes
Acknowledgements
We would like to thank all the colleagues in the Division of Infectious Diseases, Chongqing Public Health Medical Center for their assistance with the related data and literature collection.
Ethical Considerations and Informed Consent
This study was conducted in compliance with the Declaration of Helsinki and was approved by the Institutional Review Board of Chongqing Public Health Medical Center (approval number: 2022-055-01-KY). Written informed consent was obtained from the patient.
Author Contributions
KH, ML, and XD contributed to the study conception & design, organized, and revised the manuscript before submission to the journal. QC, ML, and JQ contributed to patient management and data collection. All authors proofread the final manuscript and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the first batch of key Public Health Key Discipline Construction Project (Junior College), Chongqing Science and Technology Bureau (CSTB2022TIAD-KPX0180), Chongqing medical scientific research project (Joint project of Chongqing Health Commission and Science and Technology Bureau, 2024MSXM117), Chongqing medical scientific research project (Joint project of Chongqing Health Commission and Science and Technology Bureau, 2025MSXM135).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
