Abstract
Background
We evaluated the impact of the CHORUS™ app on adherence to the cabotegravir and rilpivirine long-acting injectable (CAB + RPV LAI) monthly injections schedule.
Methods
Healthcare centers (HCCs) were randomized to access CHORUS™ CAB + RPV LAI features (intervention) or not (control) from 01OCT2021-31JAN2022. Target window adherence (maintenance injections ≤7 days before/after target day) was assessed with multivariate logistic regression (generalized estimating equations).
Results
CAB + RPV LAI was administered to 188 and 79 individuals at intervention and control HCCs, respectively. Intervention was not associated with improved target window adherence (adjusted odds ratio: 0.61 [95% CI: 0.30-1.25]). However, app use was associated with increased odds of adherence compared to no app use among all intervention HCCs (2.98 [1.26-7.06]) and at smaller HCCs (3.58 [1.31-9.80]).
Conclusions
While access to CHORUS™ CAB + RPV LAI features did not improve target window adherence, app use did, especially at smaller HCCs which may not have established LAI management procedures.
Trial registration
Plain Language Summary
Cabotegravir + rilpivirine long-acting injectable (CAB+RPV LAI) is the first long-acting regimen for HIV treatment, which was approved in the US in 2021. CAB+RPV LAI should be administered ≤7 days before/after the target date. We conducted a trial to evaluate the impact of the CHORUS™ app and web portal on the timing of monthly CAB+RPV injections. The intervention clinics had access to features designed to help with CAB+RPV LAI management, including flagging delayed/missed injections and appointment scheduling status. Control clinics did not have access to these features and managed CAB+RPV LAI administration on their own. Access to the app and web portal features for intervention clinics had no impact on timing of injections compared to control clinics. However, intervention clinics who actively used the app were close to three times more likely to give injections on-time than intervention clinics who did not use the app. The effect of app use was seen specifically among smaller clinics caring for <1000 people with HIV: smaller clinics that actively used the app were 3.58 times more likely to give injections on-time than those who did not use the app. In conclusion, while access to CHORUS™ CAB+RPV LAI features in the app and the web portal did not improve the likelihood of on time injections, actively using the app did make a difference, especially at smaller clinics which may not have established injection management procedures.
Background
HIV treatment options have evolved rapidly over time. Recent clinical development has focused on extended-release and long-acting formulations, which may be dosed anywhere from once a week to every 6 months. Various routes of delivery for ultra long-acting formulations are currently being explored, including oral delivery, intravenous infusion, intra-muscular injection, subcutaneous injection, and implantable/removable device delivery.1–4
Cabotegravir and rilpivirine long-acting injectable (CAB + RPV LAI) was the first long-acting regimen for HIV treatment approved in the United States. It was shown to be non-inferior to daily oral therapy.5–9 Intramuscular injections of long-acting CAB + RPV have a long elimination half-life ranging from 5.6 to 11.5 weeks for CAB and 13 to 28 weeks for RPV. 10 A monthly CAB + RPV LAI schedule was approved by the US FDA in January 2021, with a loading dose of 600 mg CAB + 900 mg RPV, followed by maintenance injections of 400 mg CAB + 600 mg RPV administered once a month, on the same day of the month as the loading dose. 11 As of February 2022, the FDA expanded the label to approve every 2 months dosing, with the first 2 doses of 600 mg CAB + 900 mg RPV administered 1 month apart, and subsequent doses administered every 2 months, on the same day of the month. 12 As of March 2022, in the US, CAB + RPV LAI can also be initiated without an oral lead-in. 13 CAB + RPV LAI should be administered within 7 days before/after the date of the scheduled injections (target window). 14 Lack of adherence to the target window can have a clinically important impact on treatment effectiveness, with longer delays between injections resulting in greater likelihood for CAB or RPV plasma concentrations below therapeutic levels, in particular within the first few months of use. 15 Oral bridging is recommended for delayed or missed injections and should be started 1 month (±7 days) after the last monthly injection or 2 months (±7 days) after the last every 2 months injection. Oral bridging consists of oral formulations of CAB and RPV; alternative oral regimens can also be used. 14
With potential advantages of long-acting formulations including improved treatment adherence and reduced treatment burden for the user, new long-acting formulations such as CAB + RPV LAI have been viewed positively by both people with HIV (PWH) and providers.16–19 However, coordinating injection visits and adherence to the dosing schedule can be an added burden on some medical practices. Reported challenges include the need for personnel to procure, manage, and administer injections, as well as insurance denials causing delays in treatment initiation.18,19 New tools and processes are needed to provide additional adherence benefit and ease staff demands in supporting patients on CAB + RPV LAI. The objective of this study was to evaluate the CAB + RPV LAI features in the CHORUS™ app for the management of monthly CAB + RPV LAI injections in terms of adoption and adherence to the injection target window.
Methods
CAB+RPV LAI Features in the CHORUS™ App
The CHORUS™ App and web portal were developed as a tool for clinicians to easily visualize important patient information from their smart phones. Electronic health record (EHR) data from participating clinics are encrypted, extracted, and processed daily. Relevant information is electronically distributed and placed within the corresponding module in the CHORUS™ mobile app for iOS and Android, and on the CHORUS™ web portal. High levels of encryption are employed to ensure the security and confidentiality of protected health information (PHI) and only authorized clinic personnel have access to CHORUS™. Of note, the primary purpose of the CHORUS™ app and web portal is not research; they are provided as a service and were already used prior to this study for normal healthcare operations and for quality improvement.
New CHORUS™ features were developed to help with the management of long-acting injectables such as CAB + RPV LAI. The CHORUS™ app includes a listing refreshed daily of all patients on the schedule yesterday, today, and tomorrow. With the new features, PWH receiving CAB + RPV LAI are flagged in this list if they missed an injection. In addition, alerts are generated for CAB + RPV LAI recipients based on their injection and scheduling status, indicating that an appointment needs to be scheduled or confirmed, or that injections were missed or administered outside of the target range.
Study Setting and Randomization
This study was conducted within healthcare centers (HCCs) from the AIDS Healthcare Foundation (AHF), a global non-profit organization offering multiple services including HCCs, pharmacies, wellness centers, and specialty Medicare programs. As of March 2022, AHF provided care for 104,945 individuals across 17 US States and territories. To avoid overlap of providers between intervention and control arms, HCCs within the same geographic area were grouped into a single randomization unit. Only groups of HIV primary care clinics with ≥100 active PWH at the time of randomization were included. Selected HCC groups were categorized by size: small (<300 active PWH), medium (300-999 active PWH), and large (≥1000 active PWH). Within each size category, HCC groups were randomly assigned to the intervention or control arm. The randomization schedule was generated using the SAS PLAN procedure.
Intervention arm HCCs were given access to the CHORUS™ CAB + RPV LAI features. Control arm HCCs used their own processes (standard of care) to manage patients on CAB + RPV LAI. They had access to the CHORUS™ app and web portal with all functionalities except the new CAB + RPV LAI features. In both study arms, personal or AHF-provided devices were used to access CHORUS™.
The study was originally intended to take place from 01OCT2021-7JUL2022. However, the FDA approved every 2 months CAB + RPV LAI dosing earlier than anticipated, on 01FEB2022. Because the alerting algorithm had been developed for monthly dosing, alerts were rendered inaccurate if every 2 months dosing was used. Therefore, while the CHORUS™ CAB + RPV LAI features remained available through the end of the study, only data up to 31JAN2022 were assessed.
Measurements
Data on HCC and patient characteristics, as well as timing of CAB + RPV LAI administration were obtained from the Observational Pharmaco-Epidemiology Research & Analysis (OPERA®) cohort, a longitudinal healthcare database of prospectively captured clinical data from EHR, to which AHF takes part.
Adherence to target windows was defined as maintenance injections administered within the target treatment window (ie, within 7 days before/after the target day). For analysis purposes, the target day for maintenance injections was updated monthly and set to the same day of the month as the previous injection. This approach was employed because the interpretation of a custom target day field in the EHR varied across staff members, leading to erroneous entries completely unrelated to the actual injection schedule. For example, the number 28 was often entered in this field even when all injections had been given in the middle of the month, within 31 days of the prior injections, thus maintaining therapeutic levels.
To assess adoption of the CHORUS™ CAB + RPV LAI features, data on the number of interactions per week in each HCC were obtained from the app itself. Interaction with CHORUS™ was defined as navigating to a different screen of the app. Adoption was also assessed via 2 surveys disseminated by the AHF Chief Medical Officer to the AHF workforce at intervention HCCs. The pre-intervention survey was sent before CAB + RPV LAI features were rolled-out and assessed barriers and facilitators to CAB + RPV LAI delivery as well as acceptability of app-based CAB + RPV LAI management. The post-intervention survey was sent after the study ended and assessed barriers and facilitators of CAB + RPV LAI delivery as well as acceptability and adoption of the CHORUS™ CAB + RPV LAI features.
Statistical Analyses
Descriptive statistics were employed to assess HCC and CAB + RPV LAI recipients characteristics stratified by randomization arm (ie, intervention vs control), as well as adoption of the CHORUS™ app within the intervention arm. Target window adherence was assessed in the subset of the population with ≥1 maintenance CAB + RPV injections using logistic regression models fit with generalized estimating equations to account for clustering. 20 First, all HCCs in the intervention and control arms of the study were compared. This model was adjusted for the number of PWH in care at the clinic (linear and quadratic terms), age (linear and quadratic terms), CD4 cell count (linear and quadratic terms), clinic in the Southern US, female sex, Black race, Hispanic ethnicity, any comorbid conditions, and health coverage through Medicaid or ADAP/Ryan White (a proxy for socioeconomic status). Second, analyses were restricted to the intervention group to compare HCCs that ever used the CHORUS™ app during the study period to those who never used it, both overall and stratified by number of PWH in care at the clinic (≥1000 vs < 1000). These models were adjusted for continuous age (linear and quadratic terms), CD4 cell count (linear and quadratic terms), female sex, Black race, Hispanic ethnicity, and health coverage through Medicaid or ADAP/Ryan White.
Ethics Approval and Consent to Participate
Institutional review board (IRB) approval covering patient data contained in the OPERA® database was received from Advarra IRB; a waiver of informed consent and authorization for the use of PHI for patient data was granted (Pro00023648). IRB approval from Advarra IRB was also received for survey data collected from AHF workforce members (Pro00050130). A waiver of documentation of consent for AHF staff was granted. Each site randomized to the intervention was notified of their inclusion in the study. Participation, defined as the active use of the CAB + RPV LAI features in the CHORUS™ app, was on a voluntary basis and no staff member was penalized for not using it. To opt-out, staff members simply abstained from using the CAB + RPV LAI features in the CHORUS™ app. Usage of the CHORUS™ app and CAB + RPV LAI features remained private. The healthcare organization did not have access to information on utilization of the CAB + RPV LAI features in the CHORUS™ app during or after the study. The study was conducted in accordance with HIPAA and HITECH requirements, which expand upon the ethical principles detailed in the 1964 Declaration of Helsinki. The CHORUS™ App and web portal complies with HIPAA and HITECH requirements and operates under a Business Associates Agreement that is consummated between Epividian and each medical organization separately.
Results
Study Population
A total of 37 HCCs providing care to 21,777 PWH were included in the intervention arm; 28 HCCs providing care to 23,907 PWH were included in the control arm. Intervention HCCs tended to have fewer PWH in care (median 466) than control HCCs (median 567) at study launch. However, 46% of intervention HCCs had any experience with CAB + RPV LAI prior to 1OCT2021, compared to 36% of control HCCs (Table 1). During the study period, CAB + RPV LAI injections were administered in 22 (59%) intervention HCCs and 15 (54%) control HCCs.
Characteristics of Healthcare Centers at Study Launch a .
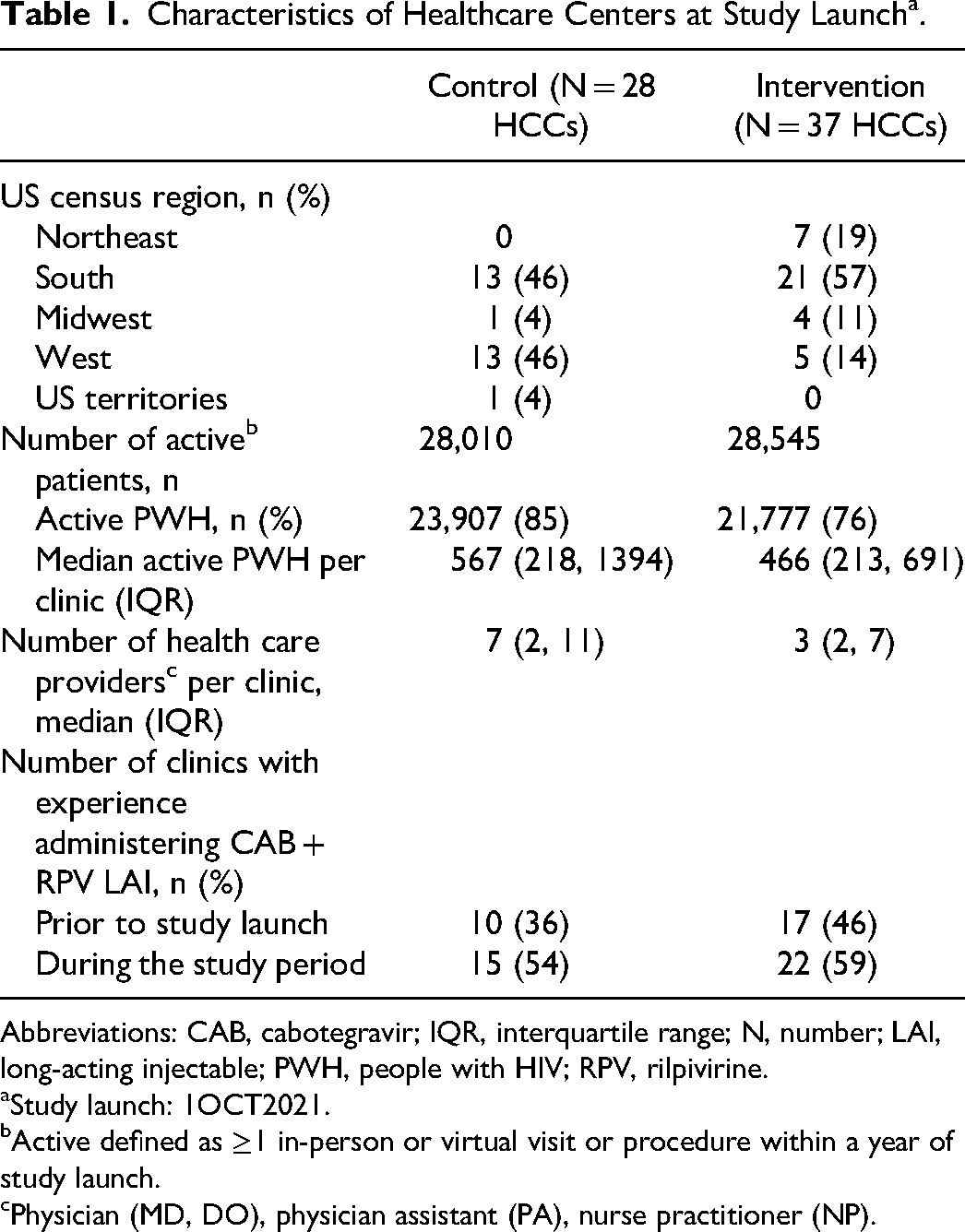
Abbreviations: CAB, cabotegravir; IQR, interquartile range; N, number; LAI, long-acting injectable; PWH, people with HIV; RPV, rilpivirine.
Study launch: 1OCT2021.
Active defined as ≥1 in-person or virtual visit or procedure within a year of study launch.
Physician (MD, DO), physician assistant (PA), nurse practitioner (NP).
Over the course of the study, a total of 188 PWH in the intervention arm and 79 PWH in the control arm received any CAB + RPV LAI. Of note, despite efforts to avoid overlaps, 7 individuals received injections in both intervention and control HCCs. CAB + RPV LAI recipients in the intervention arm were more likely to be women or Black, and less likely to have commercial insurance or switch from an integrase inhibitor (INSTI)-based regimen, compared to those in the control arm (Table 2).
Characteristics of PWH Who Received CAB + RPV LAI During the Study Period.
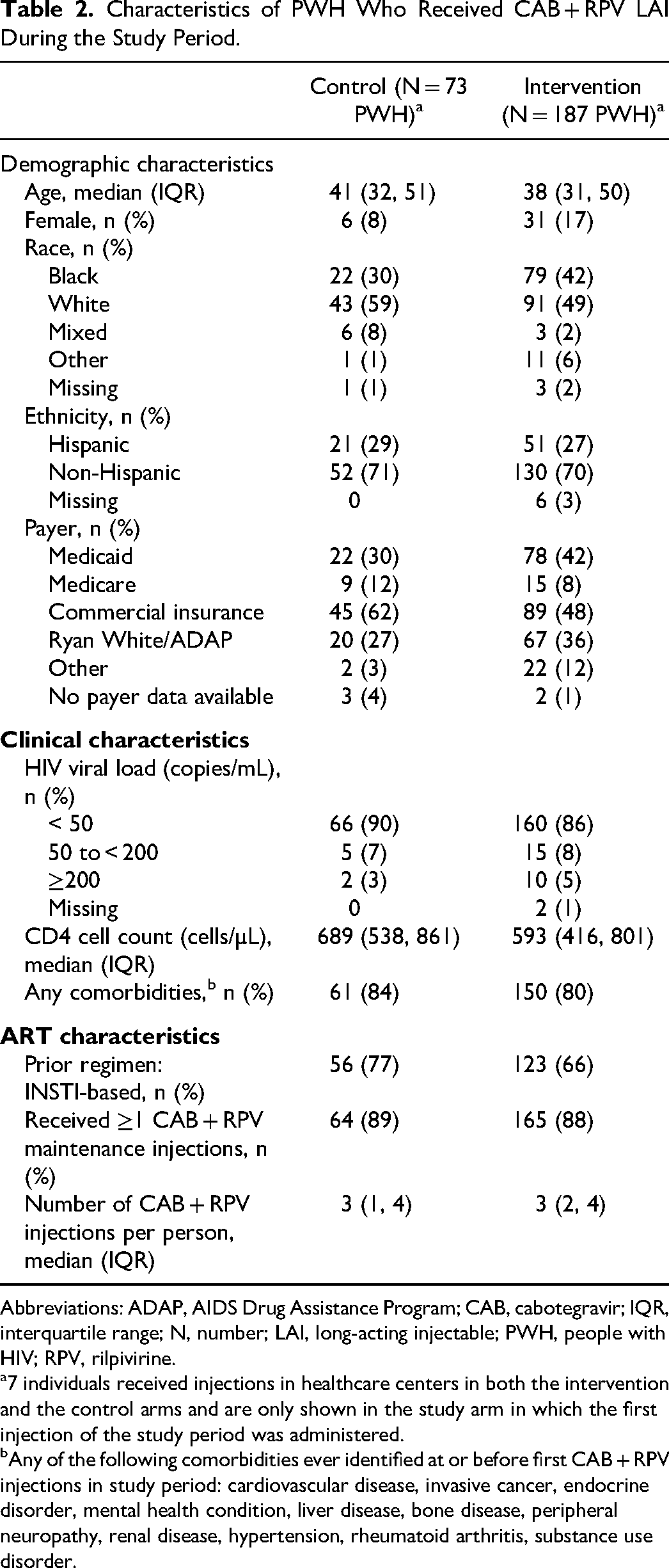
Abbreviations: ADAP, AIDS Drug Assistance Program; CAB, cabotegravir; IQR, interquartile range; N, number; LAI, long-acting injectable; PWH, people with HIV; RPV, rilpivirine.
7 individuals received injections in healthcare centers in both the intervention and the control arms and are only shown in the study arm in which the first injection of the study period was administered.
Any of the following comorbidities ever identified at or before first CAB + RPV injections in study period: cardiovascular disease, invasive cancer, endocrine disorder, mental health condition, liver disease, bone disease, peripheral neuropathy, renal disease, hypertension, rheumatoid arthritis, substance use disorder.
Adoption of the CAB + RPV LAI Features in the CHORUS™ App
Adoption was assessed among HCCs in the intervention arm. Over the course of the study, a median of 46 interactions with the app were recorded per clinic (IQR: 19, 115). As many as 17 HCCs (46%) had no interactions with the app, while 12 (32%) had 1 or 2 interactions per week and 8 HCCs (22%) had at least 3 interactions per week.
Pre- and post-study surveys were completed by 31 and 9 members of the AHF workforce, respectively. The biggest perceived barriers to CAB + RPV LAI delivery were related to scheduling (no-shows, patient unavailability, cancellations), staffing, and insurance coverage. Such barriers were reported both at study launch and after study conclusion. Availability of an integrated system to track target windows was deemed to be the most important facilitator for CAB + RPV LAI delivery, both at study launch and after study conclusion.
Target Window Adherence
In the control arm, 93% (95% CI: 89, 97) of maintenance injections were administered within the target window (3% delayed; Figure 1A), compared to 87% (95% CI: 84, 90) in the intervention arm (11% delayed; Figure 1B). Odds of in-target injections were lower in the intervention than control arm, with an unadjusted odds ratio of 0.50 (95% CI: 0.26, 0.96). After statistical adjustments, the odds ratio was 0.61 (95% CI: 0.30, 1.25; Table 3).
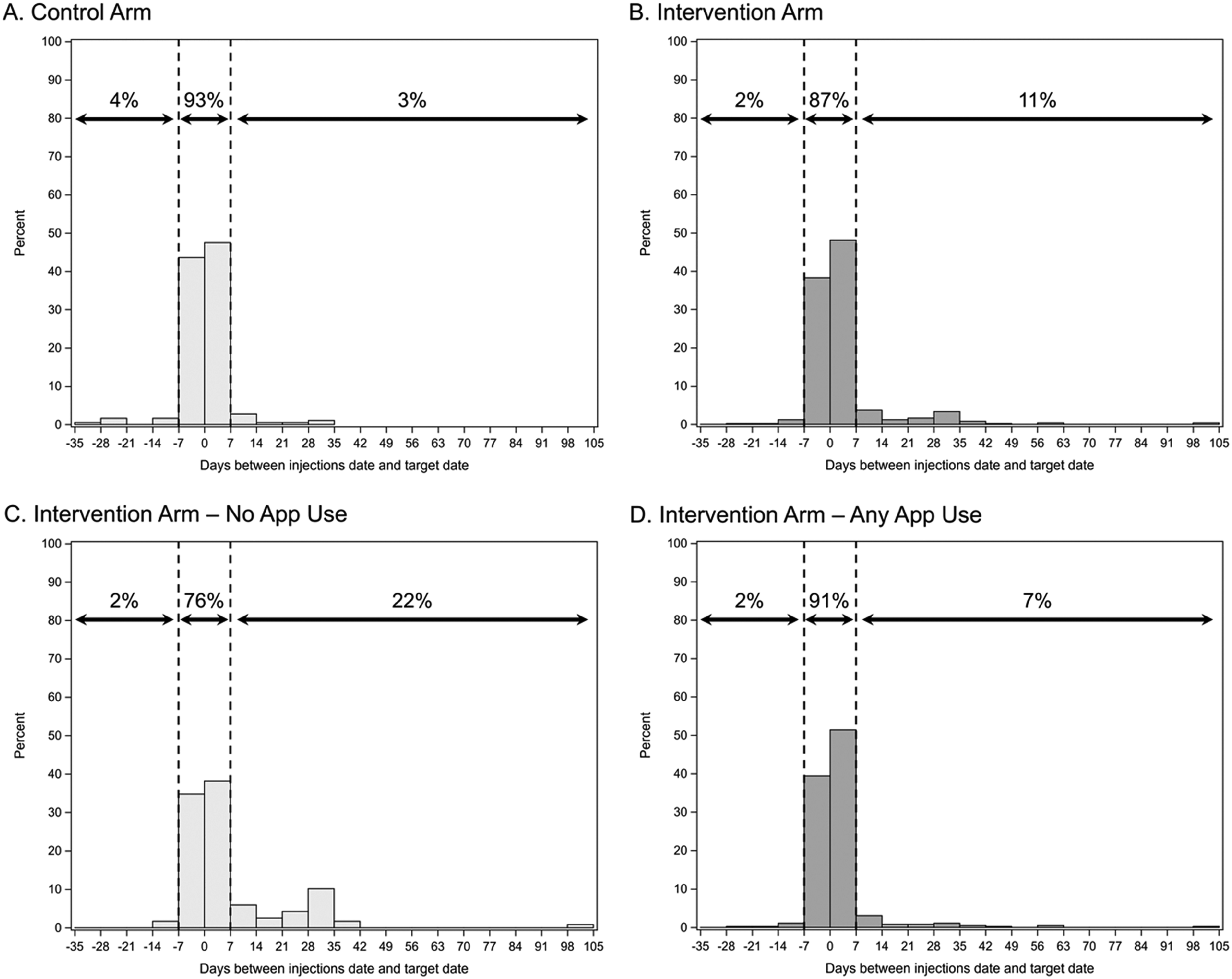
Days between CAB + RPV LAI maintenance injections and target date by study arm ([A] control arm, [B] intervention arm), and by app use within the intervention arm ([C] no app use, [D], any app use).
Association Between Study Arm and Likelihood of In-Target Maintenance Injections, Restricted to Individuals With ≥1 Maintenance CAB + RPV Injections.
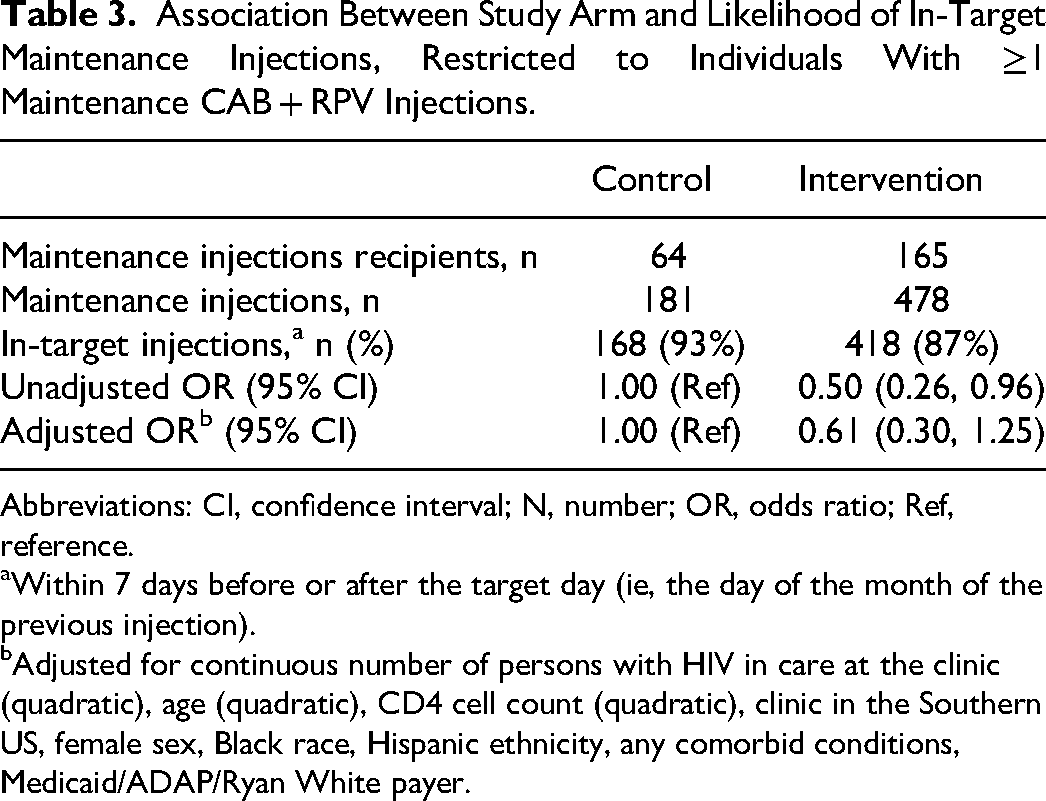
Abbreviations: CI, confidence interval; N, number; OR, odds ratio; Ref, reference.
Within 7 days before or after the target day (ie, the day of the month of the previous injection).
Adjusted for continuous number of persons with HIV in care at the clinic (quadratic), age (quadratic), CD4 cell count (quadratic), clinic in the Southern US, female sex, Black race, Hispanic ethnicity, any comorbid conditions, Medicaid/ADAP/Ryan White payer.
Among intervention HCCs, proportion of in-target injections varied based on the frequency of CHORUS™ app use. Indeed, 76% (95% CI: 69, 84) of maintenance injections were administered within the target window in HCCs that never used the app (22% delayed; Figure 1C), compared to 91% (95% CI: 88, 94) in HCCs with any app use (7% delayed; Figure 1D). Both HCCs with <3 interactions per week and those with ≥3 interactions per week administered 91% of injections within the target window. After statistical adjustment, odds of in-target injections were close to 3 times higher in HCCs that used the CHORUS™ app than those who did not (odds ratio: 2.98 [95% CI: 1.26, 7.06]). The number of PWH in care at the HCC appeared to modify the impact of app use. In HCCs with a larger PWH clientele (≥1000 PWH), there was no association between CHORUS™ app use and in-target injections, and confidence intervals were very wide (odds ratio: 0.72 [95% CI: 0.08, 6.49]). In HCCs with a smaller PWH clientele (<1000 PWH), odds of in-target injections were 3.58 times higher in HCCs using the app than in those that did not use it (odds ratio: 3.58 [95% CI: 1.31, 9.80; Table 4]).
Association Between CHORUS™ App Use and Likelihood of In-target Maintenance Injections, Restricted to Intervention Arm, in the Subset of the Population With ≥1 Maintenance CAB + RPV Injections.
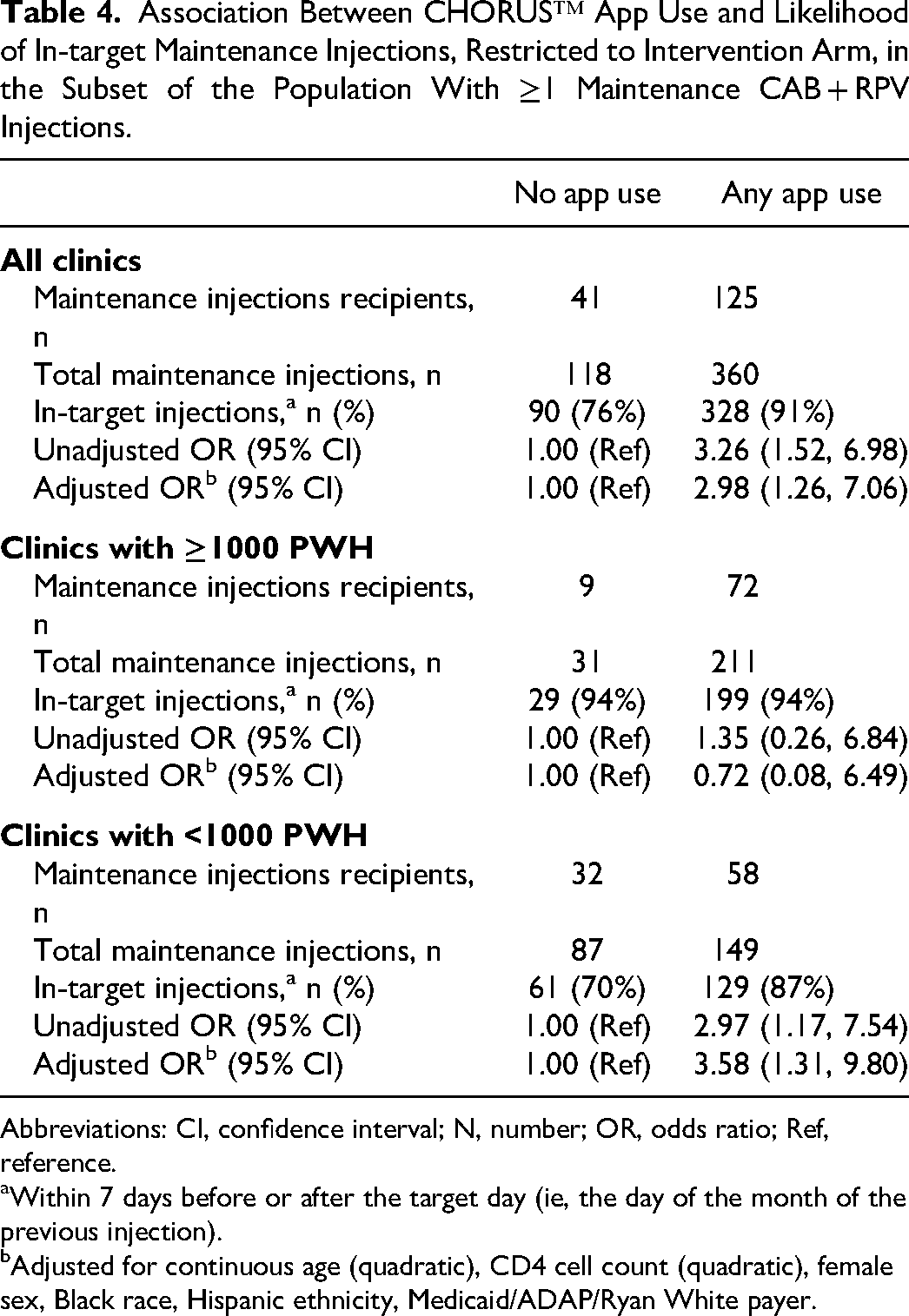
Abbreviations: CI, confidence interval; N, number; OR, odds ratio; Ref, reference.
Within 7 days before or after the target day (ie, the day of the month of the previous injection).
Adjusted for continuous age (quadratic), CD4 cell count (quadratic), female sex, Black race, Hispanic ethnicity, Medicaid/ADAP/Ryan White payer.
Discussion
Over the course of the study, 188 PWH received CAB + RPV LAI injections in 22 intervention HCCs, and 79 PWH received injections in 15 control HCCs. In the intervention arm, 46% of HCCs never interacted with the CHORUS™ app, 32% had 1 or 2 interactions per week, and 22% had at least 3 interactions per week. The biggest perceived barriers to CAB + RPV LAI delivery were related to scheduling, staffing, and insurance coverage, both at study launch and after study conclusion.
Similarly to the concerns raised in this study, health care providers who participated in the FLAIR, ATLAS, and ATLAS-2M clinical trials in the United States and in Spain noted that adherence to injection appointment represented a barrier to CAB + RPV LAI implementation, as did the need for additional staff and logistical issues. 18 Likewise, during a pilot CAB + RPV LAI program at a large Ryan White-funded clinic in the Southeast of the United States, the main implementation challenges faced included the burden on staff to manage the drug supply, administer injections, and support the CAB + RPV LAI recipients, as well as insurance denials delaying treatment initiation. 19
A majority of CAB + RPV LAI injections were administered within the target window, within 7 days before/after the target day in the control (93%) and intervention (87%) arms of this study. Though the adjusted odds ratio pointed towards a lower likelihood of in-target injections in the intervention than the control arm, statistical significance was not achieved. Estimates of target window adherence were consistent with estimates in clinical trials and early reports of routine clinical care. The 5-year results from the LATTE-2 trial revealed that as many as 96% of 9803 expected injection visits occurred within the target window; ≥ 90% target window adherence was achieved by 90%, and 100% adherence was achieved by 54% of participants. 7 Even during COVID-19, target window adherence remained high (94%) in 6 clinical trials. 21 In routine clinical care, of 39 PWH who received at least 2 maintenance CAB + RPV LAI injections, 87% were adherent to the target window; 2 individuals experienced a single delay greater than 7 days after the target, and 2 others experienced a delay twice. 22 In another OPERA study following 321 individuals who received ≥1 CAB + RPV maintenance injection, 90% of injections were received on time. 23
While access to the CHORUS™ CAB + RPV LAI features did not improve adherence to target windows, use of the app itself did. In the intervention arm, 76% of maintenance injections were administered within the target window in HCCs that never used the app, compared to 91% in HCCs with <3 app interactions per week and 91% in those with ≥3 interactions per week. HCCs that used the app were statistically more likely to adhere to the target window overall. Moreover, HCCs with a smaller PWH clientele (ie, < 1000 PWH) also saw a benefit to the app use, although those with a larger PWH clientele did not. The difference in target window adherence observed based on the number of PWH served by the HCC may be explained by clinics’ experience with CAB + RPV LAI. HCCs caring for more PWH may have participated in CAB + RPV-LAI clinical trials or have a higher demand for CAB + RPV LAI and may thus have a more robust system in place for its delivery and management. In addition, such HCCs may have more resources and staff members dedicated to HIV services who can manage the administration of monthly injections. All these factors have been identified as playing an important role in successful implementation of LAI ART.16–19 Thus, these HCC characteristics may reduce the need for additional tools such as those provided by the CAB + RPV LAI features in the CHORUS™ app. In contrast, HCCs with fewer PWH may benefit from these tools to set up a system and ensure target window adherence.
This study is not without limitations. Indeed, it had to be stopped early due to the FDA approval of every 2 months dosing for CAB + RPV LAI in February 2022. Transitioning from receiving injections once a month to once every 2 months dosing occurred frequently at the study sites, although the alert system could not be modified in time to accommodate this change. Therefore, starting in February 2022, the alerts were no longer pertinent for anyone transitioning to an every 2 months dosing schedule. Ending the study prematurely was the best solution to assess the intervention as intended. Training on the CHORUS™ app and CAB + RPV LAI features was done via an online training platform already in use at this organization. Emails were sent by the Chief Medical Officer informing providers and staff of their participation in the intervention and directing them to the training material; 75% completed the training. A more hands-on approach to training may have been more effective to ensure everyone at intervention HCCs were aware and able to use the CAB + RPV LAI features in the CHORUS™ app. Participation in the surveys was low, limiting the generalizability of their findings. However, barriers and facilitators identified by survey participants were aligned with the literature. Use of the CAB + RPV LAI features in the CHORUS™ app could not be measured directly. Instead, any interaction with the app (ie, every time one navigates to a different screen) was used as a proxy. However, it is not known if the provider interacted with the CAB + RPV LAI features specifically. Control HCCs also had access to the app, but not to the CAB + RPV LAI features. Therefore, results from the comparison between the intervention and control arms may be attenuated by the potential benefits of using the general CHORUS™ app for CAB + RPV LAI management even without access to the CAB + RPV LAI features. However, the analysis restricted to the intervention arm, which focused on comparing HCCs using the app to those that did not, provided a clearer understanding of the benefits of the CHORUS™ CAB + RPV LAI features for CAB + RPV LAI management. The determination of target dates also proved to be complex. While AHF created a custom EHR field to record the target day, upon closer inspection of the data, it became apparent that some providers set the target date to 28, regardless of the actual injection dates. For example, someone who received injections on 12OCT2022, 10NOV2022, 9DEC2022, and 11JAN2023 is receiving their injections consistently every month, ensuring the maintenance of therapeutic levels. However, their target day may be set to 28 in the EHR, resulting in seemingly out-of-target injections every month. Therefore, the alerts received in the app for this individual would not correspond to the care provided or needed. For this reason, for analysis purposes, the target day was reset every month to the same day as the previous injection, ensuring that the injections described above are interpreted as being in-target. In addition, both the alerts and EHR fields were tweaked throughout the study. While the nature of the intervention did not change, it was not uniform throughout the study. This suggests that the greatest benefit of the app use was likely the feature notifying the staff of CAB + RPV LAI users on the schedule yesterday, today, and tomorrow, rather than the alerts. Finally, oral bridging regimens and prescriptions are not uniform: some providers chose to maintain the prior ART prescription, some wrote a new prescription at CAB + RPV LAI start, and a few wrote a new prescription before a gap that corresponded with the duration and timing of bridging medication use. Further, the EHR does not include a field to note if oral bridging was taken. Therefore, information about oral bridging in the EHR was inconsistent and often missing; and could not be used to characterize the dosing delays observed.
Strengths of this study include its cluster randomized trial design, which allowed for the comparison of HCCs with and without access to the CAB + RPV LAI features in CHORUS™. The use of clusters minimized the risk overlap between the intervention and control arms, although providers and PWH did occasionally provide or receive care in more than 1 HCC, sometimes in a different study arm. In addition, the study was conducted within the AHF network, which provided access to 65 HCCs throughout the US and Puerto Rico meeting the inclusion criteria for the study. These HCCs represent a wide variety of practices serving different populations, but they are all part of the same organization, and therefore share a similar structure, and the same EHR and training capabilities. Moreover, CAB + RPV LAI-related directives were uniform across sites. CHORUS™ app use was already established as part of the tools offered for routine clinical care at AHF. App use variation within the intervention arm allowed for the assessment of the impact of frequency of app use on target day adherence. This analysis provided clarity on the benefits of the CHORUS™ app for CAB + RPV LAI management.
Conclusions
In conclusion, a large number of PWH on a CAB + RPV LAI regimen had the potential to be reached by the intervention, although adoption was not optimal. Importantly, CHORUS™ app use had a strong impact on adherence to injection target windows, specifically among HCCs caring for fewer PWH. Indeed, within intervention HCCs, app use was associated with a 3-fold increase in the likelihood of in-target maintenance injections. However, while app use improved target window adherence, simple access to the CAB + RPV LAI features did not. This study shows that technology can facilitate the management of CAB + RPV LAI in routine clinical care in the United States.
Footnotes
Acknowledgments
This research would not be possible without the generosity of people living with HIV and their OPERA® caregivers. We are especially grateful for the support of the providers and staff at the participating AHF Healthcare Centers, including Elizabeth Balet, Hans Foy, Robert Heglar, and Christine Uranaka. Additionally, we express a special thanks to the following individuals at Epividian: Robin Beckerman, Bernie Stooks, and Lisa Lutzi.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MBW has participated in post-conference advisory boards for the Conference on Retroviruses and Opportunistic Infections (CROI) and International AIDS Conference (IAC) and also serves as a principal investigator on ViiV Healthcare clinical trials but does not receive personal compensation for this work, which goes directly to the AIDS Healthcare Foundation. MBW is also a member of the Epidemiology and Clinical Advisory Board for Epividian. LB, JSF and GPF are employed by Epividian, Inc.; Epividian has had research funded by AIDS Healthcare Foundation, EMD Serono, Gilead Sciences, Janssen Scientific Affairs, LLC, Merck & Co., TheraTechnologies Inc., and ViiV Healthcare. QC has no competing interest to declare. RKH has received research grants from Gilead Sciences, Janssen and ViiV Healthcare, speaker honoraria from ViiV Healthcare, Merck, Gilead Sciences and Janssen, and advisory board participation with ViiV Healthcare, Gilead Sciences, Janssen, and Epividian.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by ViiV Healthcare.
