Abstract
Objectives
To determine the incidence of opportunistic infections (OIs) and the predictive factors for the development of OIs after antiretroviral therapy (ART) initiation among treatment-naïve patients with HIV infection.
Results
Of 401 HIV-infected patients, 38 (9.5%) HIV-infected patients developed OIs after initiating ART, with an incidence rate of 25.6/1000 person-years. The median time (IQR) from ART initiation to OI occurrence was 26.5 (14-73) days. In multivariate Cox proportional hazard regression, body mass index ≤18.5 kg/m2 (adjusted hazard ratio [aHR] 2.28, 95% confidence interval [CI] 1.18-4.42, P = .015), symptoms at presentation (aHR 13.59, 95% CI 3.24-56.9, P < .001), serum glutamate-pyruvate transaminase >55 U/L (aHR 2.09, 95% CI 1.06-4.15, P = .035), and initiation of a dolutegravir-based regimen (aHR 4.39, 95% CI 1.54-12.48, P = .006) were significantly associated with OIs after ART initiation.
Conclusion
OIs after ART initiation are common. Malnutrition, symptomatic presentation, abnormal liver enzymes, and DTG-based regimens are predictors of OI occurrence after ART initiation. Physicians must monitor and appropriately treat OIs after ART initiation.
Introduction
Human immunodeficiency virus (HIV) infection continues to have devastating health effects globally, with over 39 million HIV-related deaths to date and more than 36 million HIV-infected patients currently. 1 Despite substantial advancements in antiretroviral therapy (ART) and worldwide progress toward implementation of treatment-as-prevention programs, approximately 2 million people become newly infected with HIV every year. 2 Most deaths among HIV-infected patients are attributable to acquired immune deficiency syndrome (AIDS)-defining illnesses. Among non-AIDS-related deaths in HIV-infected patients, most are owing to cancer, followed by stroke, infection, and liver disease. 3
In Thailand, approximately 520,000 adults with HIV-infected patients were reported in 2021. Additionally, 6,500 were newly infected with HIV, 450,000 (86%) were currently on ART, and 9,200 died from AIDS. 4 The main cause of death among HIV-infected patients in Thailand is owing to serious opportunistic infections (OIs). 5 A recent study showed that 42% of newly diagnosed HIV-infected patients were symptomatic and 32% were diagnosed with AIDS; approximately one-third had OIs at diagnosis of HIV infection, including pulmonary tuberculosis (TB, 73.3%), pneumocystis pneumonia (PCP, 55.8%), extrapulmonary TB (45.3%), and cryptococcosis (16.3%). 6 In a systemic review and meta-analysis from Switzerland, the most common OIs in ART-naive participants were oral candidiasis (19.1%), unspecified TB (10.0%), herpes zoster (9.4%), pulmonary TB (9.0%), bacterial pneumonia (6.1%), and genital ulcer disease (6.0%). 7
OIs are the first clinical manifestations that alert clinicians to the occurrence of AIDS. In the context of HIV, OIs are defined as infections that are more frequent or more severe because of HIV-mediated immunosuppression. 8 When OIs occur, plasma HIV viral load (VL) increases; together, these accelerate HIV progression, increase the risk of HIV transmission, and also increase morbidity and mortality.9,10 The most important strategy for preventing OIs is ART, as well as medication prophylaxis for some diseases.11‐13 However, after initiation of ART, immune function improves rapidly and causes systemic or local inflammatory reactions at the site of the preexisting infection or other sites, or unrecognized OIs may develop. 14 Moreover, OIs can develop after virological or immunological failure, despite being on ART. 15 These types of conditions are called “OIs after ART initiation.” Several studies have demonstrated that lower CD4 cell count (≤50 cells/mm3), low baseline bodyweight ≤50 kg, poor adherence to ART, undernutrition, and presentation with advanced World Health Organization HIV clinical stages (III and IV).16,17 Updated studies regarding this issue are limited. The incidence and clinical characteristics of OIs, as well as risk factors of OIs after ART initiation, might change over time for many reasons, such as early ART initiation, more potent antiretroviral drugs, and a higher proportion of screening OIs before ART initiation. In this study, the authors aimed to determine the incidence of OIs after ART initiation, predictors related to the occurrence of OIs after ART, and mortality owing to OIs after ART.
Materials and Methods
Authors conducted a retrospective cohort study among adults (aged ≥18 years) with a first diagnosis of HIV infection based on positive HIV antibody test results, who were naïve to ART, started ART at Ramathibodi Hospital, Mahidol University, Bangkok, Thailand (a 1,200-bed university hospital) from January 2016 to December 2019, and who were followed until 30 June 2022. Individuals were excluded if they had a follow-up visit less than one visit after ART initiation.
Clinical and demographic data, including age when first testing positive for HIV antibody, sex, body mass index (BMI), route of HIV acquisition, comorbidities, clinical presentation, clinical staging, previous OIs before ART initiation, medications for OI prophylaxis, antiretroviral regimen, laboratory investigations, and ART adherence were obtained by reviewing the medical records. Adherence to the latest visit was assessed by the physician and good adherence was defined as more than 90%. 18 Laboratory investigation results, including CD4 cell count, HIV VL, hemoglobin (Hb) level, renal function (creatinine), liver function (serum glutamate-pyruvate transaminase; SGPT), and coinfection with hepatitis B virus (HBsAg), hepatitis C virus (anti-HCV), or syphilis (VDRL and TPHA), were also retrieved and reviewed.
The sample size for a single proportional model 19 was calculated using 95% power to detect an expected prevalence rate of OIs after ART initiation of 44.7% 17 with an expected loss to follow-up of 5% where the significance level (alpha) was 0.05. As a result of this estimation, the sample size was 01. Mean values (with standard deviation [SD]) or median values (with interquartile range [IQR]) and frequency were used to describe patients’ characteristics for continuous and categorical data, respectively. The chi-square test or Fisher's exact test and Student t-test were used to compare categorical variables and continuous variables between the 2 groups, respectively. Baseline characteristics of individuals were compared between HIV-infected patients with and without OIs after ART initiation. The Kaplan–Meier method was used to estimate the cumulative probability of OIs. Univariate and multivariate Cox proportional hazard regression analyses were performed to determine the factors associated with OIs after ART initiation. Variables with a P value <.05 were considered in a multivariate Cox proportional hazard regression model. Variables were entered into a multiple Cox proportional hazard regression model with stepwise forward selection, and variables with P < .05 were retained in the model. The hazard ratio (HR) and its 95% confidence interval (CI) were estimated. A P value <.05 was considered statistically significant. All statistical analyses were performed using Stata Version 17.0 (StataCorp LLC, College Station, TX, USA).
The protocol of this study was reviewed and approved by the Research Ethics Committee of the Faculty of Medicine Ramathibodi Hospital, Mahidol University (No. MURA2021/584). The ethics committee did not require individual consent due to the retrospective study design.
Results
A total of 403 ART-naïve HIV-infected patients were identified from the database. Two HIV-infected patients were excluded due to having follow-up visits less than one visit after ART initiation. Thus, 401 HIV-infected patients were included in the analysis (Figure 1). Among the total, the mean (SD) age was 37.1 (11.9) years, 74.3% were men, and 57.6% had heterosexual risk. The most common comorbidities were hypertension (7%), dyslipidemia (3.7%), and type 2 diabetes mellites (3.5%). Common clinical presentations were asymptomatic (44%), fever (35%), weight loss (30%), pruritic papular eruption (14%), and dyspnea (13.2%). A total of 61.6% were diagnosed with AIDS. Common OIs before ART initiation were pulmonary TB (17%), extrapulmonary TB or disseminated TB (9.5%), candidiasis (13.2%), and PCP (11.5%) (Table 1). As for other coinfections, 10.5% had reactive VDRL, 52% had reactive TPHA, 9.2% had positive HBsAg, and 3.7% had positive anti-HCV. The median (IQR) CD4 cell count at ART initiation was 166 (49-314) cells/µL, and the mean (SD) HIV VL log10 was 4.9 (0.8) copies/mL.
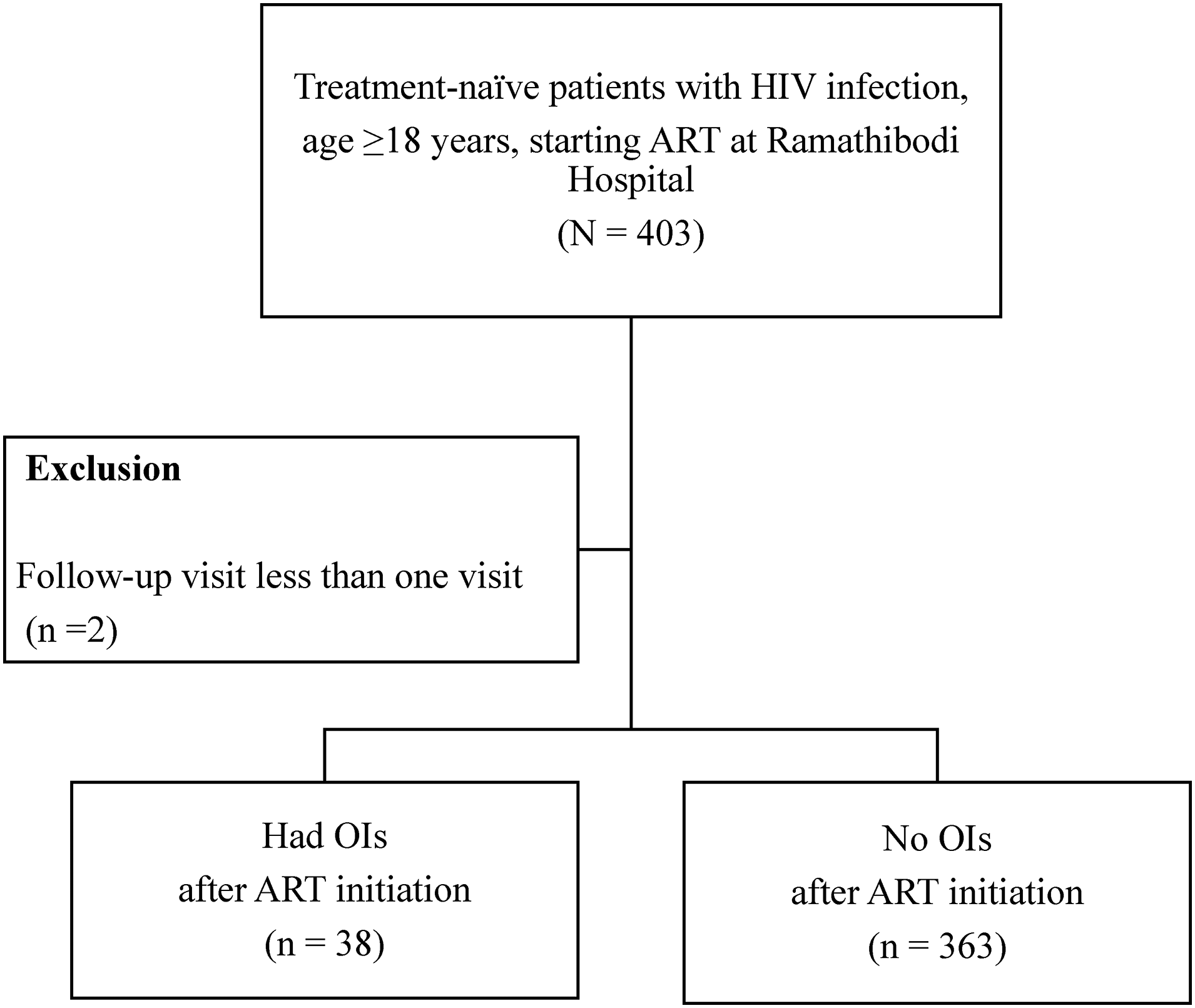
Study flow chart.
Baseline Characteristics of 401 People Living with HIV Stratified by Status of Having Opportunistic Infections after Antiretroviral Therapy Initiation.
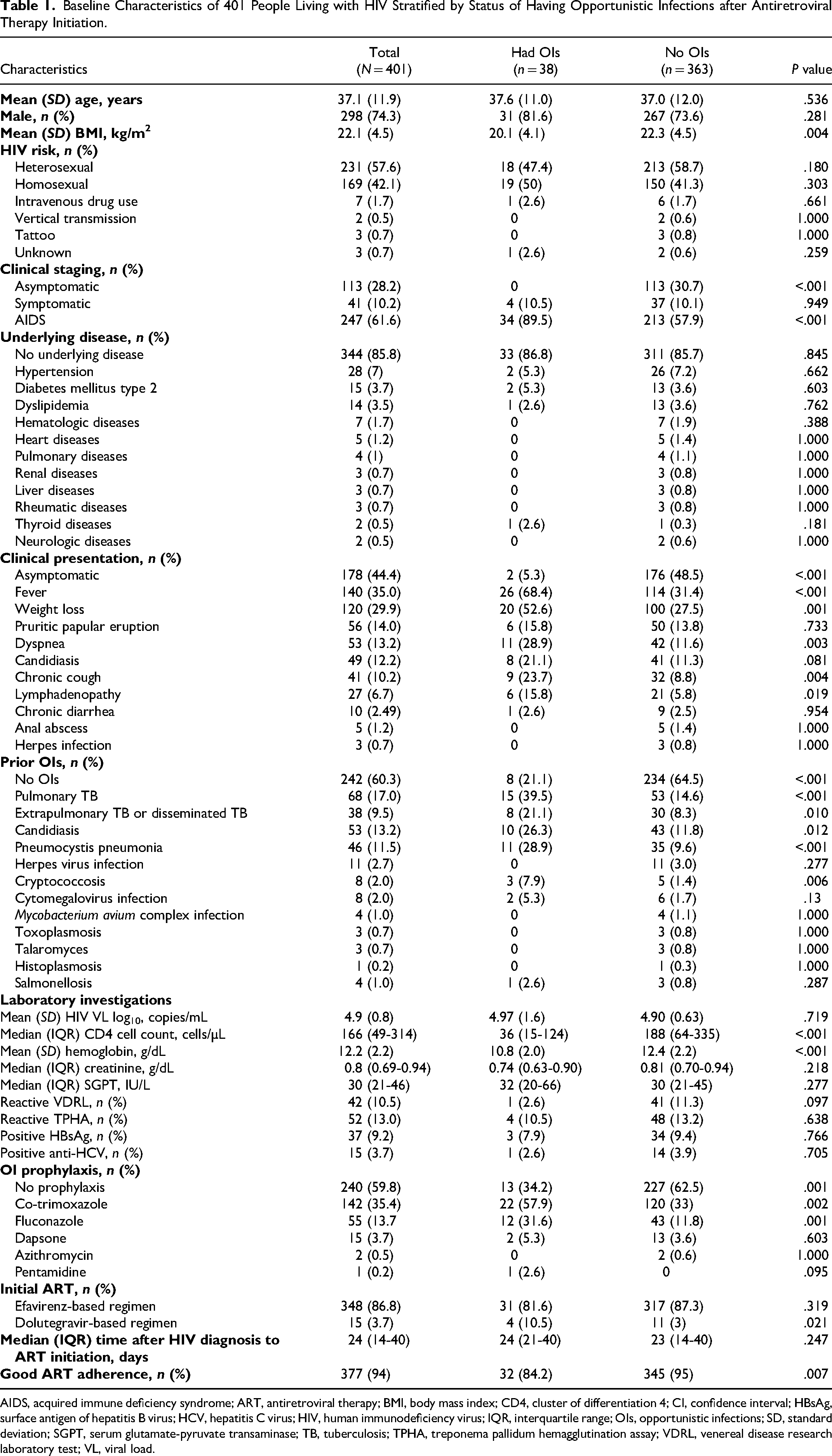
AIDS, acquired immune deficiency syndrome; ART, antiretroviral therapy; BMI, body mass index; CD4, cluster of differentiation 4; CI, confidence interval; HBsAg, surface antigen of hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; IQR, interquartile range; OIs, opportunistic infections; SD, standard deviation; SGPT, serum glutamate-pyruvate transaminase; TB, tuberculosis; TPHA, treponema pallidum hemagglutination assay; VDRL, venereal disease research laboratory test; VL, viral load.
The total follow-up time was 526,893 days, with an average of 1,307 days. The median (IQR) time from initiating ART to the occurrence of OIs was 26.5 (14-73) days. Of the total, 38 HIV-infected patients developed OI after ART initiation, with a cumulative incidence of 9.5%. Time at risk was 1,486.06 person-years and the incidence rate was 25.6 per 1,000 person-years. The most common OIs after ART initiation were extrapulmonary TB (34.2%), pulmonary TB (18.4%), cytomegalovirus (CMV) infection (15.8%), PCP (13.2%), and herpes infection (10.5%) (Figure 2). The highest cumulative incidence of OIs was observed during the first year after ART initiation (31 PLWHA, 82%), with an incidence rate of 78 events per 1,000 person-years (Figure 3). More than half (22 HIV-infected patients, 58%) of OIs occurred within the first 3 months after ART initiation.
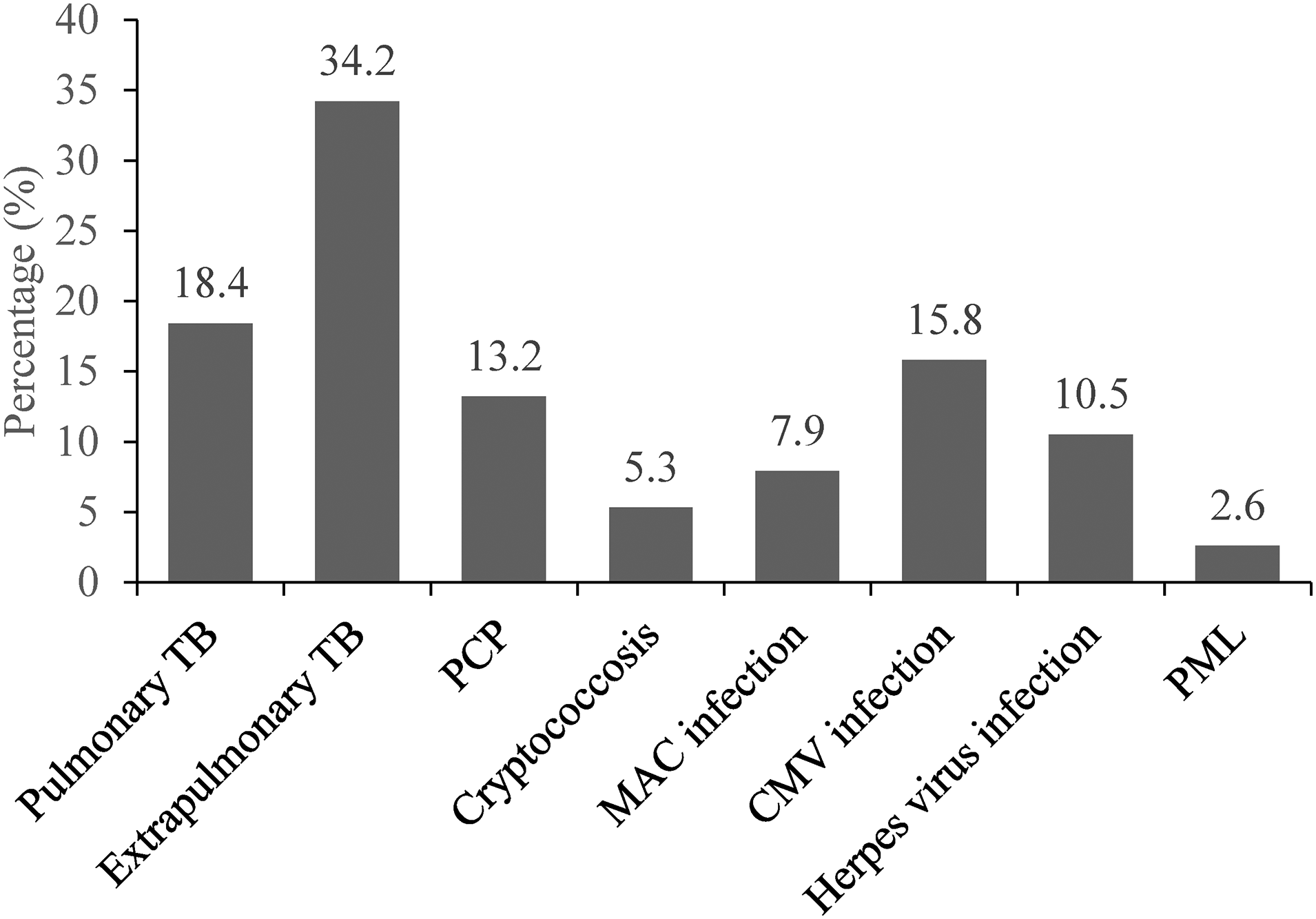
Proportion of opportunistic infections after initiation of antiretroviral therapy.
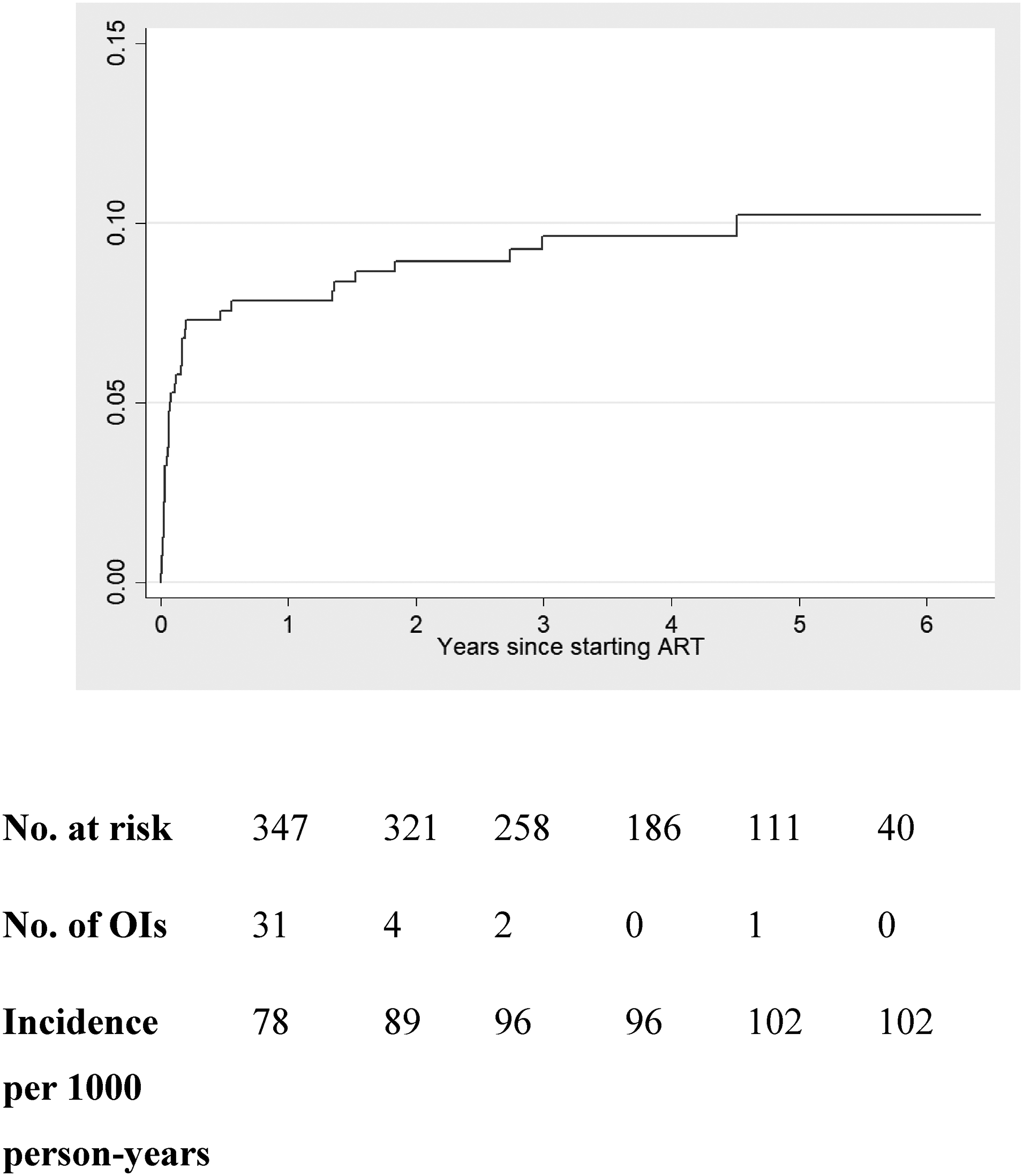
Cumulative probability of opportunistic infections after initiation of antiretroviral therapy.
HIV-infected patients who developed OIs after ART initiation were more likely to have lower mean BMI (20.1 vs. 22.3 kg/m2), lower median CD4 cell count (36 vs. 188 cells/µL), lower mean hemoglobin levels (10.8 vs. 12.4 mg/dL), and lower ART adherence (84.2% vs. 95%) (P < .05 for all). In contrast, those who developed OI after ART initiation had a higher proportion of AIDS diagnosis (89.5% vs. 57.9%), symptoms at presentation (such as fever, weight loss, and dyspnea) (94.7% vs. 51.5%), prior OIs (78.9% vs. 35.5%), OI prophylaxis (65.8% vs. 37.5%), initiation with a dolutegravir (DTG)-based regimen (10.5% vs 3%), and good adherence (84.2% vs. 95%). The baseline characteristics of HIV-infected patients stratified by status of having OIs after ART initiation are shown in Table 1. The OI-associated mortality was 5.3%.
In univariate Cox proportional hazard regression, BMI ≤18 kg/m2 (HR 2.56, 95% CI 1.35-5.00, P = .004), symptomatic at presentation (HR 15.78, 95% CI 3.8-65.56, P < .001), had prior OIs (HR 7.12, 95% CI 3.14-16.19, P < .001), CD4 cell count >200 cells/µL (HR 0.17, 95% CI 0.06-0.44, P < .001), Hb >12 g/dL (HR 0.32, 95% CI 0.16-0.61, P = .001), SGPT >55 U/L (HR 2.60, 95% CI 1.33-5.08, P = .005), received OI prophylaxis (HR 3.05, 95% CI 1.56-5.96, P = .001), DTG-based regimen (HR 4.06, 95% CI 1.43-11.51, P = .008), and good ART adherence (HR 0.32, 95% CI 0.13-0.78, P = .012) were significantly associated with developing OIs after ART initiation (Table 2).
Univariate and Multivariate Cox Proportional Hazard Regression of Opportunistic Infections after Antiretroviral Therapy Initiation.
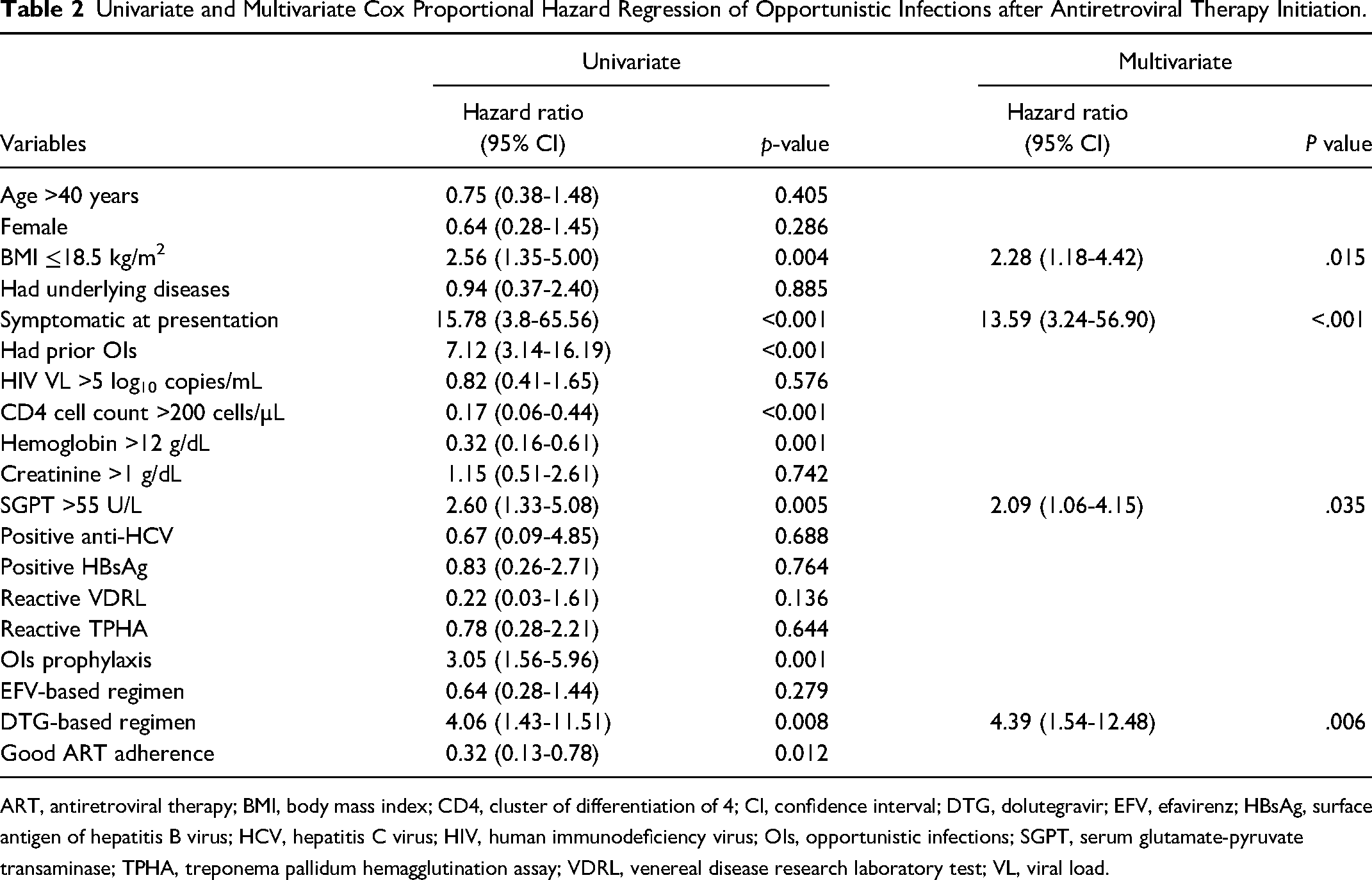
ART, antiretroviral therapy; BMI, body mass index; CD4, cluster of differentiation of 4; CI, confidence interval; DTG, dolutegravir; EFV, efavirenz; HBsAg, surface antigen of hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; OIs, opportunistic infections; SGPT, serum glutamate-pyruvate transaminase; TPHA, treponema pallidum hemagglutination assay; VDRL, venereal disease research laboratory test; VL, viral load.
In multivariate Cox proportional hazard regression, BMI ≤18.5 kg/m2 (adjusted hazard ratio [aHR] 2.28, 95% CI 1.18-4.42, P = .015), symptomatic at presentation (aHR 13.59, 95% CI 3.24-56.9, P < .001), SGPT >55 U/L (aHR 2.09, 95% CI 1.06-4.15, P = .035), and DTG-based regimen (aHR 4.39, 95% CI 1.54-12.48, P = .006) were significantly associated with OIs after ART initiation (Table 2).
Discussion
The results of this study showed that the overall cumulative incidence of OIs after ART initiation was 9.5% and the incidence rate was 25.6 per 1,000 person-years. The most common OIs after initiating ART were extrapulmonary or disseminated TB, pulmonary TB, and CMV infection. BMI ≤18.5 kg/m2, being symptomatic at presentation, SGPT >55 U/L, and a DTG-based regimen were significantly associated with OIs after ART initiation. The OI-associated mortality was 5.3%.
The authors found a cumulative incidence similar to other studies conducted in Thailand (8% and 12%).16,20 In contrast, a study in Ethiopia reported a cumulative incidence of 31.8%, approximately 3 times higher than that in our study. 21 The incidence rate of OIs after ART initiation in the present study was similar to that of reports from Brazil (26.3 per 1,000 person-years), 22 the United States, and Canada (23 per 1,000 person-years). 23 However, the Ethiopian study found an incidence rate of 135 per 1,000 person-years. 21 The higher cumulative incidence and incidence rate in the Ethiopian study might be explained by a more prolonged duration of follow-up (10 years), a larger sample size, and more HIV-infected patients with poor drug adherence (15%). Poor adherence is likely to lead to virological and immunological failure 24 and increases the incidence of OIs after ART initiation. The most common OIs after initiating ART in our study were extrapulmonary or disseminated TB and pulmonary TB. These findings were comparable to those of other studies in Thailand and Ethiopia.16,21 A possible explanation is that TB is associated with low CD4 cell counts and Thailand is a high-burden country for TB and HIV-associated TB.25,26 In contrast, the most common OIs after ART initiation in high-income countries such as the United States and Canada are PCP, candidiasis, and Mycobacterium avium complex. 23
The authors determined that predictive factors for developing OIs after ART initiation were low BMI, being symptomatic at presentation, high SGPT levels, and a DTG-based regimen. Malnutrition can cause immune dysfunction and weaken the immune system through atrophy of the thymus, spleen, and lymph nodes, reducing cell-mediated immunity. 27 These lead to susceptibility to OIs. 28 Previous studies support malnutrition as a risk factor for developing OIs after ART initiation.16,17 Furthermore, symptoms at HIV diagnosis are a clinical clue for preexisting latent OIs. 29 Increased liver enzymes may also indicate the development of an OI because these infections can activate multiple types of immune cells and pathways that cause hepatocyte damage and liver injury. 30
There is no available study determining that a DTG-based regimen is a risk factor for OIs after ART initiation among treatment-naïve HIV-infected patients. Most published studies determined DTG-based regimens were associated with immune reconstitution inflammatory syndrome (IRIS), not OIs, after ART initiation. The DTG-based regimen is a potent regimen with more reduced plasma HIV VL compared to those containing protease inhibitors or nonnucleoside reverse transcriptase inhibitors. 31 It is also associated with faster recovery of CD4 cell counts, which can cause more paradoxical worsening of treated OIs or unmasking IRIS.14,32 A DTG-based regimen is borderline significantly associated with an increased risk IRIS, with an odds ratio of approximately 2-2.5; however, it is not associated with mortality and hospitalization. 33 However, some studies have shown that a DTG-based regimen is not associated with an increased risk of IRIS; thus, rapid virologic decline may not be the trigger for IRIS.31,34,35 The differentiation between simple OIs after ART and IRIS is sometimes difficult. In this study, the authors decided to focus on OIs due to the complexity of diagnosing IRIS. The criteria for diagnosing IRIS were not used in this study due to a lack of consensus on the definitions of IRIS.
The strength of this study is that it is the first study in Asia to evaluate OIs after ART initiation in the era of transition from efavirenz (EFV)-based to DTG-based regimens. However, there are some study limitations. First, the nature of a retrospective cohort study can lead to an underestimation of the outcomes and may introduce selection bias. Second, OI diagnoses were based on medical records. Third, most patients had low CD4 cell counts and had initiated EFV-based regimens; therefore, our results may not be able to be generalized to other populations or settings.
In conclusion, the incidence of OIs after ART initiation is not low. We found that the most common OI after ART was TB. Malnutrition, symptomatic presentation, abnormal liver enzymes, and DTG-based regimens are predictors of OI occurrence after ART initiation. It is crucial for physicians to monitor and provide appropriate treatment of OIs after ART initiation to minimize morbidity and mortality.
Footnotes
Acknowledgments
We thank Ms. La-or Nakgul, nurse and study coordinator, Division of Infectious Diseases, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University for retrieving the electronic data of newly diagnosed HIV-infected patients in Ramathibodi Hospital. We also thank Associate Professor Sasivimol Rattanasiri, Mr. Nattawut Unwanatham, and Assistant Professor Kunlawat Thadanipon, a statistician from the Department of Clinical Epidemiology and Biostatistics, Faculty of Medicine Ramathibodi Hospital, Mahidol University, for the statistical analysis. We thank Analisa Avila, MPH, ELS, of Edanz (![]() ) for editing a draft of this manuscript.
) for editing a draft of this manuscript.
Availability of Data and Materials
The datasets generated and analyzed during the current study are not publicly available owing to institutional policy but are available from the corresponding author upon reasonable request.
Authors’ Contributions
PW and SK: research idea and study design; PW: data acquisition; PW and SK: data analysis/interpretation; PW and SK: statistical analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
