Abstract
Plain language summary
Why was the study done? Some PLHIVs discover their HIV status later after being infected, and others delay starting treatment once a diagnosis is made. These situations could lead to AHD at the start of antiretroviral treatment. AHD is a severe form of HIV disease, and people who start antiretrovirals with AHD could be at risk of several complications, including death, opportunistic infections, and higher cost of treatment. There is limited evidence about AHD among PLHIV who start antiretrovirals in the DRC and related disparities between children, adolescents, and adults in the country. What did the researchers do? We analyzed data from an HIV program implemented in Kinshasa, DRC, from 2005 to 2020. The analysis examined how common AHD is among PLHIVs, how it affects them, and how AHD and its consequences differ between children, adolescents, and adult PLHIVs. What did the researchers find? The study found that a third of all PLHIVs who started antiretrovirals had AHD. Adolescents were more affected by AHD than adults, and there were no differences between adults and children. Despite their higher risk of AHD than adults, adolescents had lower chances of dying than adults. What do the findings mean? These findings have significant implications for HIV interventions in the DRC. The study highlights the need for more effective HIV interventions targeting PLHIVs, with a focus on early diagnosis and treatment initiation. The results also suggest that interventions tailored explicitly for adolescents may be necessary to address the disproportionate impact of AHD on this population. Overall, the study provides important information on the burden of HIV in the DRC and highlights the need for continued efforts to address this public health challenge.
Introduction
The optimal population- and individual-level benefits of antiretroviral therapy (ART) for human immunodeficiency virus (HIV) are gained when diagnosis is made early and treatment is promptly initiated.1-4 This principle led the WHO to recommend in 2016 that treatment be initiated immediately upon HIV diagnosis, regardless of the level of CD4 cell count—the test and treatment strategy.5,6 This strategy is essential to reach the UNAIDS 95-95-95 targets, namely that 95% of people living with HIV (PLHIV) are to know their status; 95% of PLHIV who know their status are to receive treatment; and 95% of PLHIV who are receiving treatment are to have a suppressed viral load by 2025. 5
Although progress towards the UNAIDS 95-95-95 targets has generally lagged in developing countries since the test and treat strategy was adopted, 7 the Democratic Republic of Congo (DRC) reported significant improvement in treatment coverage from a low rate of 8% in 2010% to 82% in 2021.8,9 In addition, the country adopted in 2017 the integrase inhibitor dolutegravir (DTG) as part of first-line HIV treatment, a regimen that is easy to use, with durable virologic efficacy and favorable toxicity profile. All these factors contributed to an estimated 71% decline in AIDS-related deaths since 2010.8,9
However, this positive news may mask an enduring burden of advanced HIV disease (AHD), defined as having CD4 cell count < 200 cells/μL or WHO clinical stages III and IV. 10 Approximately 30% of PLHIV globally have AHD10,11; these patients are at increased risks for mortality (even after treatment initiation), opportunistic infections, and higher treatment costs than PLHIV without AHD.10-12 Late linkage to care, delayed ART initiation, ART failure, and poor in-care retention are the main driving factors of AHD.11,13 The high prevalence of these factors in low-income countries exposes all PLHIV to AHD; however, with lower ART coverage than adults, children, and adolescents living with HIV (CALHIV) in these settings might be at a disproportionately higher risk of initiating ART with AHD compared to adults.14,15
PLHIV in the DRC experience higher levels of AHD compared to global averages. A study reports that 97% of PLHIV ≥ 13 years old hospitalized in one health facility in Kinshasa, DRC were admitted with AHD, 44% of whom were ART-naïve with or without a prior HIV diagnosis. 16 Similarly, a prospective cohort study of 468 PLHIV under a DTG-based first-line regimen in Bunia (Eastern DRC) found that 53% of patients had AHD. 17 Also, only 58% of CALHIV in the country are on treatment compared to 82% of adults,8,18 suggesting that more CALHIV face delayed linkage to care than adult PLHIV, with potentially higher rates of AHD at treatment initiation. This partial evidence suggests that AHD is a significant threat to PLHIV in the DRC. However, to better inform local policies and programs, additional data is needed, particularly about AHD at ART initiation among all PLHIV, regardless of age groups, treatment regimen, or hospitalization status. There is also a need for more evidence about inequalities in AHD prevalence and associated outcomes between CALHIV and adults.
Using routine service data from a PEPFAR-funded program in Kinshasa, DRC, we estimated the prevalence of AHD at treatment initiation among CALHIV and adults and the risk of mortality among patients with AHD. We hypothesized that CALHIV would have higher risks of AHD (Hypothesis 1) and higher mortality (Hypothesis 2) compared to adults living with HIV.
Methods
Study Design, Setting, and Population
We conducted a retrospective cohort analysis of program data routinely collected between 2005 and 2020 among adults, adolescents, and children living with HIV at health facilities in 6 health zones of Kinshasa, namely the Limete, Matete, Lingwala, Ndjili, Mont-Ngafula and Kinshasa health zones. The analytical population consisted of patients from Kinshasa, DRC enrolled in an ART program.
Our primary analysis aimed to compare the prevalence of AHD in children (< 11 years) and adolescents (11-19 years) to that in adults (age ≥ 20 years), based on age at ART initiation. We included patients who met the following criteria: (1) ART naïve and (2) follow-up data available. AHD was defined as CD4 cell count < 200 cells/μL or WHO clinical stages III and IV or stage III (CD4% < 20%) for children. Follow-up continued from initiation of ART until either a change in ART regimen or the patient was lost to follow-up/transferred or death occurred. ART is defined as taking a minimum of 2 nucleoside reverse transcriptase inhibitors (NRTIs) plus either one non-nucleoside reverse transcriptase inhibitor (NNRTI) or a boosted protease inhibitor (PI) or Integrase Strand Transfer Inhibitor (INSTI) (eg, DTG). ART initiation eligibility in the DRC evolved over time in line with the various changes in WHO guidelines. Before 2015, the initiation of patients on ART was based on the CD4 cell count threshold (200 cells/μL, then 350 cells/μL, and later 500 cells/μL) and the WHO clinical stages III and IV. From 2015, when the country adopted the test and treat strategy, all patients became eligible for ART upon documentation of HIV diagnosis, irrespective of CD4 count or WHO clinical stage.
Our secondary objective was to quantify mortality among AHD patients and associated factors. We explored several factors, including viral suppression, defined as viral load < 50 copies/mL. The analysis related to this objective focused on patients who had AHD and met the 2 criteria described above.
Statistical Analysis
Continuous variables were expressed as median with interquartile range since they were nonnormal data. Categorical variables were expressed using frequencies and percentages. Chi-squared and Fisher's exact test were used to assess the association between the categorical variables and the age groups. The Kruskal-Wallis test was used to assess the equality of the median of the continuous variables among the age categories. A log-binomial regression model was used to assess the association between demographics, clinical variables, and AHD. Factors associated with AHD at P < .15 in unadjusted bivariate log-binomial regression were included in a multivariable model to identify predictor variables associated with AHD. Adjusted risk ratios and their 95% confidence intervals (CIs) were used as a measure of association.
Cumulative event graphs were used to assess differences between age categories and time-to-death. Time-to-death was evaluated by competing risk analysis using cumulative incidence function, with loss to follow-up as a competing event. Conventional methods for survival analysis, such as the Kaplan–Meier method and Cox's proportional hazards regression, ignore competing event(s), which can lead to biased risk estimates in the presence of competing risks (eg, loss to follow-up). Variables with P-values < .10 and a priori factors in the bivariate analyses were included in a multivariable proportional subdistribution hazards model to estimate the adjusted subdistribution hazard ratios (aSHRs) and associated 95% CIs.
Statistical analyses were performed using Stata (V.16, Stata Corp, College Station, Texas, USA), R (V, 4.1.0, R Core Team) with R Studio (V.1.4.1, R Studio Team) statistical software, and graphical representations were created in Microsoft Excel.
Ethical Approval and Informed Consent
This secondary data analysis was approved by the DRC Ministry of Health Ethics Research Committee (No404/CNES/BN/PMMF/2022 of 11/3/2022) and was deemed as “research, not involving human subjects” by FHI 360 Protection of Human Subjects Committee (Project # 2024014-1)
Results
Demographic Characteristics
We included 24,092 eligible patients on ART, of whom 1136 were children (< 11 years), 1523 were adolescents (11-19 years), and 21,433 were adults (20 years and older). The median age was 6 (IQR: 2-8) years for children, 15 (IQR: 13-18) years for adolescents, and 42 (35-50) years for adults. The characteristics of the study cohort are shown in Table 1.
Demographic and Clinical Characteristics of the Study Population by Age Category.

Note: For each variable, “missing” indicates observations with no available data.
Adolescents were less likely than adults to be female (53% vs 70.8%, P < .001); this was similar for children (70.8% vs 51.9%, P < .001). The Lingwala Health Zone had the highest proportion of participants in the children and adolescent categories (31.2% and 51.3%, respectively). The Ndjili Health Zone had the highest proportion of adults (24.2%). Compared to adults, children were more likely to have shorter follow-up duration (median 21 months; IQR [6-50] vs 29 months [10-70], P < .001) and adolescents a longer follow-up duration (median 60 months; IQR [15-125] vs 29 months [10-70], P < .001).
Advanced HIV Disease and Other Clinical Characteristics at Treatment Initiation
Of the 24,092 patients included in the study over the observation period, 32.0% (n = 7701) had AHD (Table 2). The proportion of patients with AHD was higher among adolescents (42.8%) than adults (31.2%, P < .001). There were no differences in levels of AHD between children and adults.
AHD and Clinical Characteristics of the Study Population by Age Categories.

Note. For each variable, “missing” indicates observations with no available data.
AHD: advanced HIV disease; ART: antiretroviral therapy; HIV: human immunodeficiency virus; WHO: World Health Organization.
The WHO disease stage was documented for 82% of patients (n = 19,686), 33.1% of whom were in stages III or IV (Table 2). Adolescents had a higher proportion (40.7%) and children had a lower proportion (23.9%) of patients with HIV stage III or IV at initiation as compared to adults (33.0%, P < .001). Among the 25.8% of patients with recorded CD4 count results at ART initiation, adolescents and adults had an overall median CD4 count of 309 cells/μL (IQR: 179-503). Children had an overall CD4 percent count of 0.3 (IQR: 0.16-0.92). Over time, the median CD4 count at ART initiation among children decreased from 0.275 in 2005 to 2010 to 0.17 in 2010–2015 and increased to 0.91 in 2015–2020 (Figure 1A). Adolescents and adults experienced an overall increase in all the years (Figure 1B and C).

(A) Overall temporal trends in median CD4% for children antiretroviral therapy (ART) initiation from 2005 to 2020. (B) Overall temporal trends in median CD4 count (cells/µL) for adolescents ART initiation from 2005 to 2020. (C) Overall temporal trends in median CD4 count (cells/µL) for adults ART initiation from 2005 to 2020.
The viral load test result was available for only 38.1% (n = 9198) of patients. Viral load suppression was low (63.2%) overall, and worse for children (43.9%) and adolescents (48.1%) than adults (65.2%, P < .001). Treatment outcome was documented for 33.6% (n = 8084) of patients, most of whom (73.0%) were lost-to-follow-up; this was slightly lower among adolescents (68.5%) compared to adults (73.3%). Conversely, adolescents showed a higher proportion of deaths than adults (21.8% vs 18.1%, respectively, P = .029). The efficacious DTG-based regimen accounted for 47.9% (n = 11,539) of all ART regimens and was higher for adults (49.5%) compared to children (20.7%) and adolescents (45.9%, P < .001). Overall, 91.9% of DTG prescriptions went to adult patients (Table 2).
Individual and Clinical Factors Associated With AHD at Treatment Initiation
In log-binomial regression (Table 3), the risk of AHD at ART initiation was 15% higher for adolescents compared to adults (RR: 1.15; 95% CI: 1.08-1.21; P < .001). Advanced HIV disease at ART initiation was also significantly higher among males (RR: 1.11; 95% CI: 1.07-1.15; P < .001) versus female; residents of Lingwala (RR: 1.14, 95% CI: 1.06-1.22; P < .001) and Matete (RR: 1.41, 95% CI: 1.32-1.51; P < .001) versus Kinshasa health zone; patients on Efavirenz-based (RR: 1.16, 95% CI: 1.12-1.21; P < .001), Nevirapine-based (RR:1.29, 95% CI: 1.21-1.38; P < 0.001), and other ART regimens (RR:1.17, 95% CI: 1.08-1.26; P < .001) versus DTG-based regimen; and among patients enrolled in care between 2005 and 2010 (RR: 1.27, 95% CI: 1.11-1.46; P = .001) versus those enrolled in care during the period 2015–2020. Advanced HIV disease at ART initiation was negatively associated with being a resident of Limete (RR:0.82, 95% CI: 0.76-0.88; P < .001) and Mont-Ngafula (RR:0.54, 95% CI: 49-0.60; P<.001) versus Kinshasa health zone. As shown in Figure 2, the risk ratios of AHD decreased from 2005–2010, to 2010–2015, and to 2015–2020 significantly more among children and adolescents than adults.
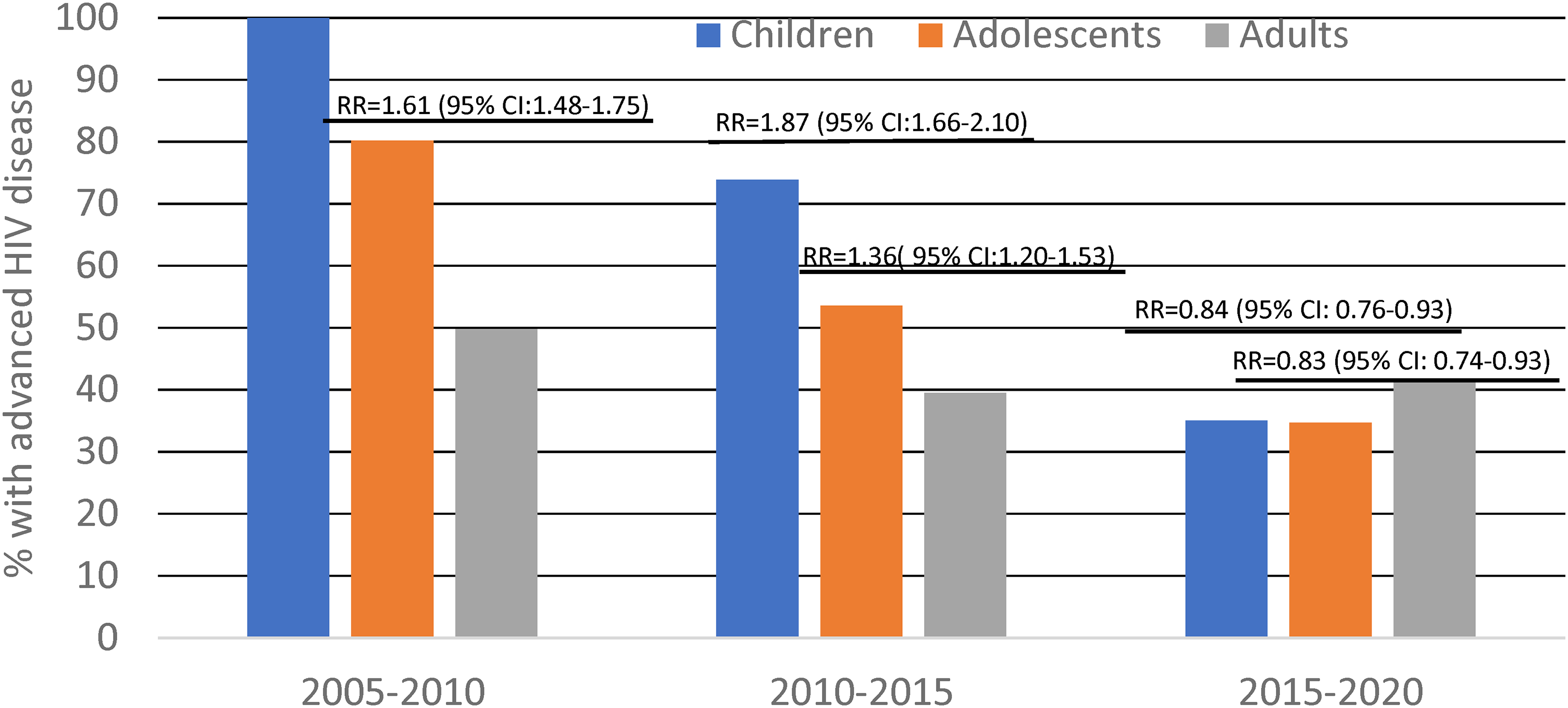
Risk ratios of advanced HIV disease at ART initiation for children, and adolescents compared to adults in 2005 to 2010, 2010 to 2015, and 2015 to 2020.
Factors Associated With AHD in Unadjusted and Adjusted Multivariable Binomial Regression.

Abbreviations: AHD, advanced HIV disease; ART, antiretroviral therapy; HIV: human immunodeficiency virus.
Mortality Risks Among Patients With AHD at Treatment Initiation
The adjusted competing-risk analysis of time-to-death (Table 4) shows that the risk of dying among patients with AHD was lower for adolescents versus adults (asHR, 0.72; 95% CI: 0.52-0.99; P = .047). Mortality among patients with AHD was also associated with living in Lingwala (asHR, 0.60; 95% CI: 0.40-0.88; P < .009) and Ndjili (asHR, 0.69; 95% CI: 0.48-0.98; P < .038) versus in Kinshasa health zone; taking Efavirenz-based (asHR, 0.68; 95% CI: 0.48-0.95; P = .025), Nevirapine-based (asHR, 0.53; 95% CI: 0.35-0.81; P = .003), or other ART regimens (asHR, 0.50; 95% CI: 0.32-0.79; P = .003) versus DTG-based ART regimen; having started ART between 2005 and 2010 (asHR, 0.44; 95% CI: 0.32-0.63; P < .001) or between 2010 and 2015 (asHR, 0.58; 95% CI: 0.46-0.73; P < .001) compared to having started ART between 2015 and 2020. These factors were associated with reduced risks of dying.
Factors Associated With Mortality among Patients With AHD, Adjusting for Loss to Follow-Up as Competing Risk.

Abbreviations: AHD, advanced HIV disease; ART, antiretroviral therapy; HIV: human immunodeficiency virus.
Finally, adolescents experienced low cumulative death events over the follow-up period as compared to adults [SHR = 0.44; 95% CI: 0.34-0.59; P < .001] (Figure 3). Children experienced more cumulative deaths as compared to adults; however, it was not statistically significant [SHR = 0.88; 95% CI: 0.58-1.34; P = .542].

Cumulative event of time to death among patients with advanced HIV disease (AHD), stratified by age categories (children, adolescents, and adults).
Discussion
This analysis explored the prevalence of AHD at ART initiation and related mortality among PLHIV in Kinshasa, DRC, and associated age inequalities. Our findings reveal that AHD at treatment initiation among PLHIV was high, at 32%, which is consistent with global estimates10,11 and a similar study in South Africa. 12 In partial support of our first hypothesis, AHD was more common among adolescents, males, and those who began treatment before 2015. Over time, there was an increase in the median CD4 count at ART initiation, with most gains seen among children and adolescents. The risk of AHD at treatment initiation decreased among CALHIV compared to adult patients over the same period. Surprisingly, the observed higher risk of AHD at ART initiation among adolescent patients contrasted with a significantly lower mortality risk when compared to adults. This finding goes against our second hypothesis that CALHIV would have a higher risk of adverse outcomes than adults.
Enrolment into care with AHD could be due to late diagnosis or delayed linkage to care once diagnosed.19-21 Given the high rate of AHD among Congolese, innovative interventions are needed to scale-up community-based HIV testing coupled with timely linkage to care with the goal of promoting rapid ART initiation as per the latest WHO HIV guidelines. For a more comprehensive approach, these strategies should incorporate components addressing the socioeconomic factors of delayed ART initiation. In Kinshasa, for example, stigmatization, religious beliefs, and limited economic resources are some known reasons for delayed ART initiation. 22
Our data indicate that efforts by local HIV programs to reach more adolescents and children are headed in the right direction, as evidenced by the decreased risk of AHD at treatment initiation over time for CALHIV and the lower mortality among adolescents with AHD versus adults. However, more efforts are needed to sustain this progress and to reach the UNAIDS 95-95-95 targets on time in 2025. There is a need to increase the coverage of integrated youth- and adolescent-friendly testing services, raise HIV status awareness, and multiply differentiated entry points in the care system for this population. 23 In South Africa, clients of adolescent-friendly services were more likely to be virally suppressed than those in conventional health facilities. 24 These services should provide linkages to the health system and offer a menu of other services, as these factors were linked to more HIV testing in the past year and facility visits for sexual and reproductive health services among adolescents in similar settings. 25
Male–female disparities in risks of AHD are in line with reports from similar sub-Saharan contexts. 26 The disparity could be explained by the higher likelihood of HIV-positive women engaging in health-seeking behaviors resulting in HIV diagnosis and care linkage as well as the increased uptake of prevention of mother-to-child HIV transmission.27,28 Moreover, in some sub-Saharan African settings, low HIV testing among men was linked to gender attitude (especially tolerance towards gender-based violence), low HIV knowledge, and holding stigmatizing views of HIV.29,30 Health policies and programs need to address these areas to reduce gender disparities in AHD.
The lower mortality risk among adolescents despite experiencing higher risks of AHD compared to adults deserves further investigation. Children and youth with AHD are highly susceptible to opportunistic infections 31 and studies report that they have lower ART coverage compared to adults. 15 These 2 factors can increase the susceptibility of children and adolescents to adverse outcomes, including early death. Our results could be explained by a lower incidence of opportunistic infections and other clinical complications in adolescents with AHD compared to adults by the time their treatment was initiated. Also, our dataset may have been subject to reporting issues, which would lead to biased results.
The strengths of this study include the comparison between children and adolescents to adults in risks of AHD and their trends over time. Although informative, our study has several limitations with potential implications for the validity of the results. First, the retrospective study design prevented the collection of selected information of interest, such as the underlying causes of late HIV presentation. Secondly, the programmatic nature of our dataset poses some challenges as the data was not primarily intended for research purposes. Furthermore, about three-quarters of patients were lost to follow-up, and a substantial proportion had missing data on CD4 cell count results on record at ART initiation. Likewise, data on clinical outcomes and ART regimens were not consistently reported for all patients. Finally, we cannot claim the generalizability of our results to other settings in DRC as Kinshasa is the capital city and likely to have better access to CD4 count lab testing and ART treatment than the remote cities in the country. Our results could also differ from other health zones in Kinshasa with HIV programs covered by other donors.
In summary, we documented a high prevalence of AHD at ART initiation in Kinshasa, DRC, with significant disparities between adolescents and adults. Although age disparities in AHD risks decreased between 2005 and 2010, 2010 and 2015, and 2015 and 2020, the scaling up of high-impact interventions targeting CALHIV is needed to sustain the current efforts and reach the UNAIDS 95-95-95 goals by 2025.
Footnotes
Acknowledgments
JBN is supported by the US National Institutes of Health (NIH), National Institutes of Allergy and Infectious Diseases (NIAID), and Fogarty International Center (FIC) grant numbers NIH/FIC #1R25TW011217-01; NIH/FIC #1D43TW010937-01A1; NIH/FIC #D43TW011827-01A1; NIH/FIC #1R21TW011706-0; and NIH/NIAID #5U01AI096299-13.
Authors’ Contributions
Conceptualization: JBN, JD, OC, and DB; data acquisition: Amu and AMb; data analysis: LNS; drafting: OC, DB, and JN. All the authors contributed to the overall critical review, writing, and approval of the final version.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The secondary analysis on which this manuscript is based was funded by FHI 360 Family Health International (grant number internal funds).
