Abstract
Introduction
Acquired immune deficiency syndrome (AIDS) is a disease caused by the human immunodeficiency virus (HIV). AIDS is a syndrome that appears in the final stages of HIV infection. In AIDS, the immune system changes which makes people more vulnerable to other infections and diseases. 1 According to the Global Burden of Disease study in 2013, HIV/AIDS with 10.4% is the first cause of death in 10- to 14-year-old children in the world. 2
In 2013, the incidence and death of the disease for all ages and in both men and women around the world were 1,847,654 and 1,340,974, respectively, with the annual percentage changes declining between 2000 and 2013, and estimated to be −3.92 and −1.54, and this proportion of developing country was higher than developed countries. The standardized rates of incidence, prevalence, and mortality of HIV infection in the world were estimated to be 98.65, 159, and 19.24 per 100,000 in 2013. 3 In Iran, the incidence and deaths of HIV in 2013 were reported to be 1398 and 325, respectively, which had an increased annual percent change of 5.64 and 12.98, respectively, from 2000 to 2013. Since HIV-related death in Iran occurs in younger age groups, the burden of disease is high. 4
AIDS is a negative phenomenon in Iranian culture and may not be easily presented in general population. 5 Sexually transmitted infections are one of the problems that may lead to social exclusion. Despite the social stigma on the sexual transmission of AIDS in Iran, in recent years, this transmission has increased by 30%. Social stigma is a kind of prejudice and a negative attitude toward people living with HIV (PLHIV) and AIDS. According to previous studies, in most cases, these people experience social stigma in various forms, including social disapproval, discrimination, embarrassment, and fear of revealing their illness. 5 Such behaviors may lead to hiding the morbidity, which may lead to very serious problems such as depression, low self-efficacy, noncompliance with treatment, reduced quality of life, drug abuse, and reduced awareness of the ways of transmitting the disease 6 and even the possibility of transmission of the infection to others.
According to the statistics released by the Vice-Chancellor of Health Affairs of Arak University of Medical Sciences, the prevalence of HIV in Markazi province has been reported to be lower than in other provinces of Iran, which suggests a special context for social stigma studies in this province. 7 “The HIV Stigma Scale” developed by Berger et al 8 has been used in Persian and for the Iranian population.5,6,9,10 This questionnaire is consisting of 40 items. In big studies, short questionnaires can be preferred both for convenience and for the accuracy of collected data. 11 Therefore, Reinius et al 12 designed and developed a 12-item abbreviated questionnaire in English. Considering the necessity of using such a short questionnaire that requires less time to collect data, we aimed to translate this questionnaire into Persian and make its cultural adaptation among PLHIV in the Markazi province.
Methods
Participants
Arak is a metropolis located in the center of Iran. Most residents of Arak speak Persian. The people of Arak are culturally similar to the rest of the Iranian cities, and they hide having infections involving high-risk behaviors including HIV. Therefore, they are also potentially exposed to HIV stigma.
The data of this study were collected from January 2019 to May 2019. PLHIV were selected from among those registered at the surveillance system of Arak Medical Science University and were enrolled in the study. The participants of this study were those who had confirmed HIV status and were 18 years of age or older.
This was a validation study on the PLHIV to standardize the stigma questionnaire from English into Persian. In this study, a questionnaire on the determination of the status of social stigma associated with HIV was used, developed by Reinius et al. 12 They have reconstructed the questionnaire with the aim of reducing the number of the 40-item questionnaire designed by Berger et al. 8 The new version of the HIV social stigma scale in Sweden was evaluated psychometrically using national study data on PLHIV, which had comparable psychometric characteristics with the 40-item questionnaire. The new scale has 12 items that is designed in 4 subscales including personalized stigma (PS; Questions 10, 11, and 12), negative self-image (NS; Questions 1, 2, and 5), disclosure concerns (DCs; Questions 3, 4, and 8), and concerns about public attitudes (CP; Questions 6, 7, and 9). Each item in the scale is answered using the 4-point Likert scale (strongly agree, agree, disagree, and strongly disagree). The higher scores indicate a higher agreement with that item. The reliability of its original version has been determined using Cronbach's alpha in the range of 0.80 to 0.88 for subscales.
The Process of Intercultural Adaptation
In this study, the intercultural adaptation process recommended by the American Association for the Consequences of Orthopedic Surgery 13 has been used.
The following processes are used to implement this study.
Step 1: Initial Translation
At this stage, forward translation was carried out. Two fluent English translators, whose mother tongue was Persian, were selected. One of the translators was an expert familiar with AIDS and social stigma and the second translator was a nonmedical expert who was unfamiliar with concepts of stigma and AIDS. After the translation was independently done by the 2 translators, the results of their translations were compared. The weak words selected at this stage were identified by the translators and resolved through conversations between the translators. The content of the items and the response options were translated at this stage
Step 2: Combining Translations
At a meeting, 2 translators and an observer were invited to combine the translation results. At this stage, a common translation version was achieved.
Finally, disagreements were resolved based on unanimity. The finalized questionnaire from this step was used for the next step.
Step 3: Back Translation
The final version of the previous step was translated into English by another translator who was blind to the original version of the questionnaire. This step is for a kind of checking the validity that is displaying conceptual errors in the translation. At this stage, 2 translators were selected independently of each other. They did not know the concepts of the questionnaire and its goals, without specialization in the field of medicine and social stigma.
Step 4: Expert Committee
The committee members included a methodologist, a health professional, an English language specialist, and translators for the forward translation and back translation stages. During this process, an author of the original paper, 12 Lars E. Eriksson reviewed the activities of the committee of experts.
In the following, all questionnaire versions were merged at this stage and a prefinal questionnaire was developed for being tested. Therefore, the main questionnaire in English, the 2 questionnaires translated in the forward translation stage, the combined questionnaire of the forward translation stage, and 2 backward questionnaires were considered at this stage. Thus, the following 4 steps were taken at this stage: semantic, idiomatic, experiential, and conceptual balance (to achieve the balance of the original version in English and the target version in Persian).
At this stage, in the committee, the original questionnaire and the backward translation were assessed for all balances.
Step 5: Test the Prefinal Version
At this stage, the finalized questionnaire was administrated to PLHIV. Three respondents were asked to examine the meanings of the items and the responses.
Step 6: Providing a Committee for Criticizing the Adaptation Steps
The goal of this step was to confirm the steps recommended in the American Association guidelines, and the reports of all processes were approved. The purpose of this stage was not to change the content, but it was assumed that by doing this, a logical translation could be achieved.
A More Accurate Evaluation of the Adapted Version
In order to measure the content validity of the Persian version of the questionnaire, 10 experts were invited to assess the questionnaire in terms of its validity indices. These experts included 2 health education experts, 2 epidemiologists, 2 sociologists, and 4 healthcare professionals familiar with HIV care. Then, for each of the items, based on clarity and relevancy, they chose 1 of the 4 options: very high, high, low, or very low. For content validity analysis, we categorized options very high and high as relevancy or clarity and other options as no relevancy or no clarity. Also, for measuring the reliability, data from the Persian version of the questionnaire was collected twice at a 2-week interval from the PLHIV.
Considering all the necessary points, the final questionnaire was read out to the participants by 2 trained people, and the patients’ responses were recorded. Two data collectors were selected from HIV/AIDS experts working in the HIV clinic. These experts are in close contact with these people for providing HIV routine care. Necessary training on how to complete the questionnaire 14 was provided to these experts.
Statistical Analysis
Summary statistics including frequency, mean and standard deviation (SD) were obtained. Content validity was measured using the scale-level content validity index (S-CVI) and item-level content validity index (I-CVI) recommended by Polit et al 15 for the whole instrument and each of the questions. I-CVI was obtained for each question according to a proportion of 10 experts who had given each question a complete score of relevancy or clarity. In order to obtain the S-CVI, all I-CVIs of the questions were summed and then divided by 12 (number of questions). Cronbach's alpha was used for the internal consistency. 14 Intraclass correlation coefficients (ICCs) and 95% confidence interval (CI) were used to obtain its reliability through test–retest. Data analysis was performed using SPSS 16.0.
Ethical Approval and Informed Consent
This study was approved by the Ethics Committee of Arak University of Medical Sciences, Arak, Iran (Approval ID: IR.ARAKMU.REC.1398.009). Additional approval was received by the Deputy of Research and Technology of Arak University of Medical Sciences, Arak, Iran (Code: 3304). Informed consent was obtained from all participants before the study began. PLHIV were told that they could leave the study at any time, for any reason, and there was no compulsory participation in the study. Also, the questionnaires did not include the names and identity information of the patients.
Results
A total of 32 patients participated in this study. Descriptive results are shown in Table 1. The average age of the participants was 44 years (with an SD of 7.9 and a minimum and maximum of 28 and 57 years). The average time from the diagnosis to the interview was 72 months (with a SD of 66 and minimum and maximum of 1 and 216 months). Precisely, 59% of the participants were men. Precisely, 59% of them were married. Heterosexual transmission was the most commonly reported transmission way of the infection (60%). The education degree of most of the participants was primary or secondary education (81%). Precisely, 94% of the participants were covered by insurance, 78% of them, at the time of initial diagnosis, were at an asymptomatic stage, and 78% of them, at the time of the interview, were at the asymptomatic stage of HIV.
Demographic Characteristics of the Study Sample (n = 32).

Abbreviations: SD, standard deviation; HIV: human immunodeficiency virus.
Ten experts evaluated the content validity of the questionnaire in terms of relevancy and clarity. Accordingly, the scores given to each of the questions were in the range of 0.9–1 (Table 2), a score of 0.9 was given to the question “Some people avoid touching me once they know I have HIV,” which was related to the word “touching” and the changes were made according to the study questionnaire, to reach the proper equivalent in the Persian language. The S-CVI of the instrument had an acceptable validity value of 0.99.
Scale-Level and Item Content Validity Index of the Persian Version of the Stigma in Patients With HIV.

Abbreviations: HIV, human immunodeficiency virus; ICC, intraclass correlation coefficient.
The reliability results of the instruments are presented in Tables 2 and 3. Accordingly, in order to measure the internal consistency, Cronbach's alpha index was 0.84 for the entire instrument and 0.69, 0.81, 0.82, and 0.77 for the NS, CP, DC, and PS subscales, respectively.
Cronbach's Alpha and ICC of the Persian Version of HIV-Related Stigma Questionnaire (N = 32).
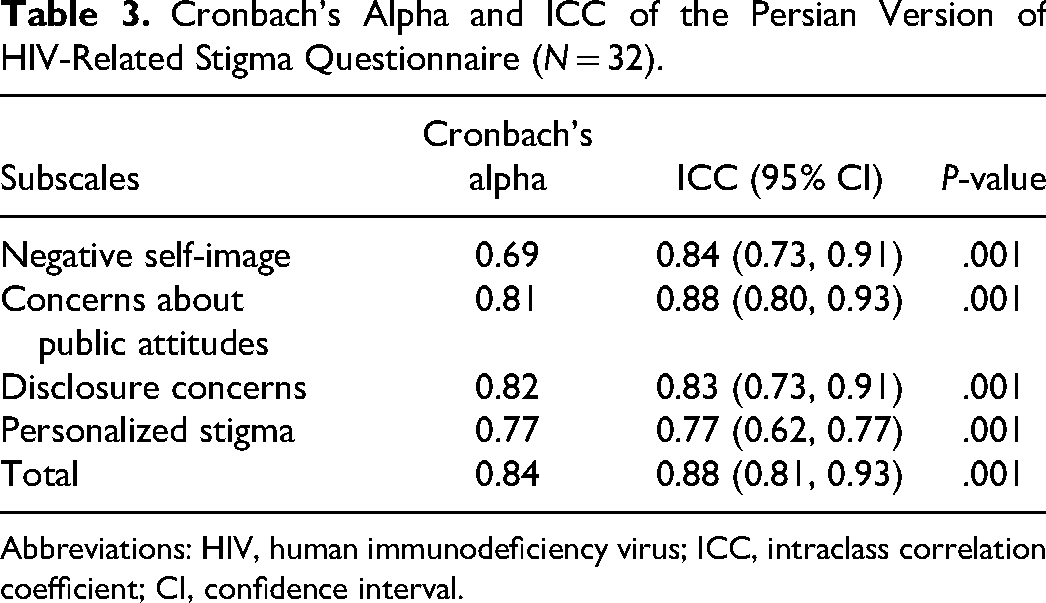
Abbreviations: HIV, human immunodeficiency virus; ICC, intraclass correlation coefficient; CI, confidence interval.
In measuring the reliability through a test–retest in a 2-week interval, the ICC for the entire instrument was 0.88 (0.81, 0.93), and for each of the subscales including NS, CP, DC, and PS, values of 0.84 (0.73, 0.91), 0.88 (0.80, 0.93), 0.83 (0.73, 0.91), and 0.77 (0.62, 0.77) was obtained, respectively. The ICCs for each of the questionnaire items are shown in Table 2, which range from 0.64 (0.25, 0.82) to 0.89 (0.77, 0.94).
Discussion
This was the first study to assess the validity and reliability of the 12 items stigma scale in a Persian-speaking population and showed that this instrument has acceptable reliability and validity in measuring HIV-related stigma.
In the evaluation of content validity, the CVI coefficients obtained for each single question and the entire instrument were high, which happened after the cultural adaptation of some words in Persian. Some of the changes that seem to be helpful even in the original version include changing the structure of the last 3 questions into conditional statements in the Persian version. For example, we changed the question “I have lost friends by telling them I have HIV” into “If I tell my friends that I have HIV, I will lose them.”
The internal reliability of the Persian version was acceptable both in general and in the subscales and was comparable to the English version in the Reinius et al 12 study, so the reliability coefficients of the subscales were similar or slightly smaller. In this study, as in the Reinius et al 12 study, the lowest reliability coefficient was obtained for the NS subscale. The reason why the reliability coefficient of this subscale was lower in our study may be the lack of homogeneity of our participants, as a proportion of the participants (35%) included wives of injecting drug users who were victims of their husbands’ addiction and were therefore infected with HIV. They answered questions related to this subscale much differently than those who were infected with the infection through other ways. However, this subscale had a relatively high reliability in test–retest reliability testing.
Test–retest reliability of this instrument demonstrates its acceptable stability over time. Therefore, the cultural and racial differences and the experiences that Iranian HIV patients get, have not led to a different assessment of HIV-related stigma. The short version that was standardized in this study, while covering all 4 major subscales of the original questionnaire, in addition to being shorter than the original version, can even have better psychometric properties since it has deleted the items that have been overlapping between multiple subscales. 12 Among other instruments investigated in this field, the 16-item HIV/AIDS Stigma Instrument—PLWHA was used by Ebrahimi-Kalan et al. 9 This instrument evaluates the 3 subscales of internalized stigma including blaming and distancing, discrimination, and fear. This instrument does not have sufficient integrity in evaluating the stigma, and the authors of the study concluded that the shortening of this instrument could have made it appear as a single-dimensional scale and suggested future studies to investigate the instruments with meaningful subscales.6,9 In another study, SeyedAlinaghi et al, 22 translated and used another questionnaire to investigate stigma in Iran. The tool included 10 key areas and 45 questions, which being too extensive and time-consuming are the major constraints to using it. 16 In another study, Pourmarzi et al 6 evaluated a 20-item instrument and the findings of this study did not achieve a high ICC coefficient and internal consistency coefficients for a number of subscales of this instrument, and the important limitation of this study is the lack of the co-layering of stigma (related to social position and risky behavior). The psychometric assessment of a short version of the Berger HIV stigma scale entitled Swahili version of the new 10-item HIV stigma scale, was conducted in the Kenyan coast. The Swahili version of the HIV stigma scale was presented as a good fit. Therefore, it is appropriate for evaluating HIV stigma among perinatally HIV-infected adolescents on the Kenyan coast. Their study results support the unidimensional model and measurement invariance across gender and age groups of the Swahili version of the new 10-item HIV stigma scale. 17 Moreover, the validity and reliability of the Internalized AIDS-related stigma scale (IA-RSS) were assessed in Turkish. This scale consists of 6 items that measure internalized stigma. IA-RSS is understandable and easily applicable in Turkish society and it is aimed to reach the most appropriate meaning of scale items in Turkish. In the internal consistency test of IA-RSS, the total Cronbach's alpha was obtained to be 0.70, and in test–retest reliability, ICCs ranged between 0.872 and 0.949, 18 and these findings were similar to this study. Similar to this study, Chan et al 19 evaluated the reliability, validity, and factor structure of the Internalized AIDS-related stigma scale in India. The findings on Cronbach's alpha and validity indices were similar to this study. Furthermore, Mokhtarabadi et al 20 and Aziz et al 21 assessed the validity and reliability of other HIV-related stigma scales among nonpeople with HIV; that is, general population and healthcare providers. These studies may be related to HIV stigma among PLWHA, so it is important to consider them in any study related to stigma validation, especially in confirmatory and exploratory factor analysis.
Among the strengths of this study is its short, yet comprehensiveness of the instrument. Moreover, the use of skilled people in the expert committee to examine the questionnaire, which carefully examined the meanings and clarity of the words to prevent possible misunderstandings. Another point of strength is that during all these processes, the authors of the original research paper reviewed the activities of the committee of experts. This study has also some limitations. Since the HIV population is hidden and is difficult to estimate and access, 16 we invited PLHIV under the care of the health center to participate in the study which may be potentially nonrepresentative for all Iranian PLHIV. In addition, a number of PLHIV might refuse to answer the questions correctly due to HIV and its high stigma, so there is a risk of misclassification bias.11,22 In order to reduce the likelihood of the occurrence of this bias, the study interviewers were advised to ensure the PLHIV that their information will remain confidential to the researchers so that they can answer the questions accurately in the data collection phase.
For a lot of reasons including that stigma is associated with depression in PLHIV, which may lead to noncompliance with treatment, measuring of stigma is important among PLHIV. In addition, stigma may cause fear of rejection, which ultimately leads to a PLHIV not taking advantage of potential social support. 8 Therefore, by using an efficient instrument for estimating the stigma among PLHIV, special HIV-related interventions may be used to help them.
Conclusion
The Persian version of the 12-item HIV-related stigma questionnaire was found to be, in addition to being short and comprehensive, acceptable reliability and high validity to use in order to determine the stigma related to Persian-speaker PLHIV. Further studies are suggested to determine other reliability and validity indices such as convergent, divergent, and predictive validities for the scale validation, as well as further studies in more sample sizes than this study.
Footnotes
Acknowledgments
The authors appreciate the contributions and the valuable comments of Dr. Lars E. Eriksson at Karolinska Institutet. The authors thank the expert committee for the process of content validity. The authors also thank the study volunteers for their participation and all of the staff at the Arak HIV clinic for their great help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Arak University of Medical Sciences, Arak, Iran (Approved code: 3304).
