Abstract
Kidney disease is the fourth most common cause of non-AIDS-related mortality in people living with HIV. Combination antiretroviral therapy (cART) remains the cornerstone of treatment. However, little is known about the impact of cART on disease outcomes in patients with HIV-associated nephropathy (HIVAN) and HIV-immune complex kidney disease (HIVICK). This systematic review evaluates the impact of cART on progression to end-stage kidney disease (ESKD) and other outcomes in HIV-infected individuals. We conducted a literature search utilizing PubMed, and Cochrane database and 11 articles met inclusion criteria for analysis of which nine HIVAN studies showed decreased progression to ESKD or death for subjects when treated with cART versus those untreated. However, two studies showed no survival advantage with cART. Three HIVICK studies showed improvement in delaying ESKD in subjects on cART compared to untreated subjects. cART appeared to reduce the risk to ESKD or death in patients with both HIVAN and HIVICK.
Introduction
Approximately 1.2 million people in the United States are currently living with Human Immunodeficiency Virus (HIV) infection. 1 It has been transformed from an inexorably fatal disease to a relatively controllable chronic condition, primarily due to advancements in antiretroviral pharmacotherapy (Figure 1). HIV affects multiple organs, including the kidney, heart, skin, and lungs. Kidney disease is the fourth most common cause of non-acquired-immune-deficiency syndrome-(AIDS)-related mortality in the people living with HIV (after oncologic, cardiac, and liver disorders). 2 HIV infection can result in a diverse clinical and histologic spectrum of renal diseases; HIV-associated nephropathy (HIVAN), HIV-associated immune complex kidney disease (HIVICK), thrombotic microangiopathy-related renal disease, and others. 3 Research in the last two decades has shown that early initiation of combination antiretroviral therapy (cART) in people living with HIV may result in a near-normal life expectancy. 4 The advent of cART in the 1990s produced a paradigm shift in the HIV, with pandemic, striking reductions in patient mortality and morbidity. 2 Nevertheless, along with co-morbidities like diabetes mellitus (DM) and hypertension (HTN), chronic HIV infection may play a substantive role in the rising prevalence of chronic kidney disease (CKD) and is associated with poor health outcomes. 5 Several treatment modalities are available for people living with HIV with end-stage kidney disease (ESKD), including renal replacement therapy (RRT) and renal transplantation, with excellent survival rates. 6

Evolution of ART, HIV-associated mortality and inclusive dates of studies reviewed. Key:
HIVAN
HIVAN is the predominant renal manifestation in people living with HIV seen in approximately 3.5–10% of the US population infected with HIV, 7 chiefly African Americans. The apolipoprotein L1 (APOL-1) gene predicts strong genetic predisposition in individuals with sub-Saharan African ancestry, especially those who carry renal risk variants G1 and G2 mutations. 8 Clinically, HIVAN presents with heavy proteinuria, declining renal function, hematuria, and peripheral edema. 9
HIV targets glomerular and tubular epithelia, causing podocyte dysregulation and tubular-epithelial cell dysregulation, ultimately leading to collapsing focal segmental glomerulosclerosis (Figure 2). Histologically, HIVAN presents as collapsing focal segmental glomerulonephritis (FSGN) with features of tubular microcyst formation (containing proteinaceous casts), tubular injury, endothelial tubule-reticular inclusion and interstitial inflammation. 10
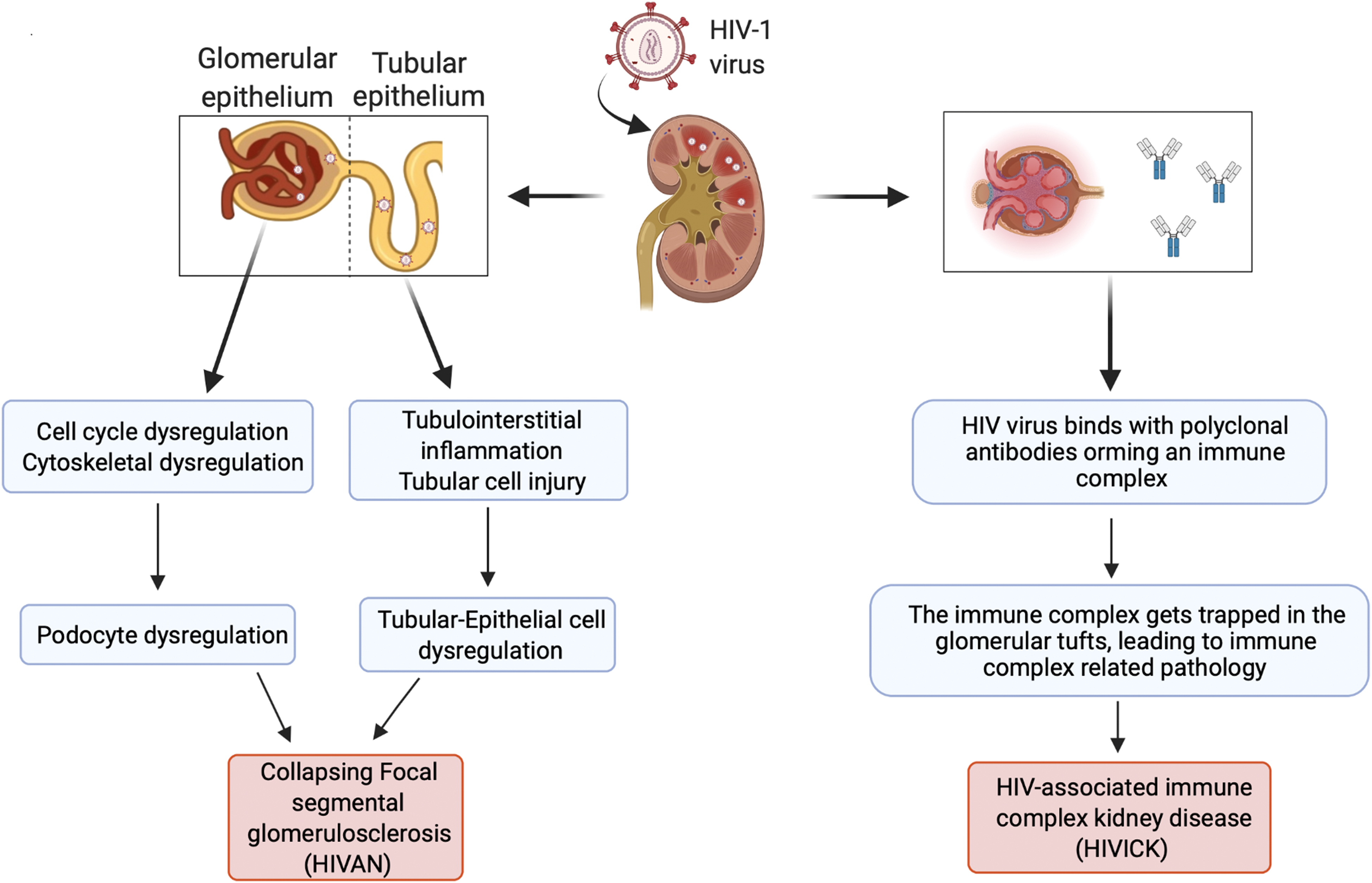
Pathophysiology of human immunodeficiency virus-associated renal disease. HIV-1, Human immunodeficiency virus; HIVAN, HIV-associated nephropathy.
HIVICK
HIVICK is the broad term connoting renal diseases other than HIVAN in people living with HIV – including membranous nephropathy, membranoproliferative nephropathy, and thrombotic microangiopathy, but excluding IgA nephropathy. 11
HIVICK is the result of immune complex-mediated renal damage and on histology it presents as global glomerular immune complex deposition. The mechanisms that drive HIVICK include trapping circulating immune complexes in glomerular tufts (which may accompany polyclonal hypergammaglobulinemia and immune reconstitution), or in-situ formation of immune complexes, causing complement activation and tissue injury (Figure 2). 12
HIVICK is not associated with increased frequency of APOL1 risk alleles, and frequently occurs in non-African populations. Clinically, HIVICK is diagnosed when lupus-like renal lesions are present in people living with HIV, without serological evidence of systemic lupus erythematosus. Patients present with nephrotic-range proteinuria, hematuria, and low complement levels.8,11 They tend to have lower viral loads, higher CD4 counts, and higher estimated glomerular filtration rates (eGFR), with better prognosis than individuals with HIVAN. 8
For both HIVAN and HIVICK, cART remains the cornerstone of therapy. 3 This qualitative systematic review evaluates the impact of cART on progression to ESKD, other clinical outcomes and death in people living with HIV.
Materials and Methods
Design
Qualitative systematic review was conducted in accordance with ENTREQ (Enhancing Transparency in Report the Synthesis of Qualitative Research) guidelines (Table S1). 13 The study (developed with agreement of all authors), involving three stages: 1) Initial consultation with a medical librarian for assistance with development of a search strategy, including exclusion and inclusion criteria; 2) quality and critical appraisal and data extraction; 3) data aggregation and findings synthesis.
Search Terms and Search Methods
A systematic search for this review was conducted in PubMed, EMBASE, SCOPUS, and Cochrane databases. The search strategies employed a combination of subject terms, controlled vocabulary (MeSH and EMTREE) and keywords, including but not limited to: “HIV-associated nephropathy,” “antiretroviral drugs,” “HIV immune complex kidney disease,” and “viral load.” Each database was searched from inception through January 2021, employing no date or language filters. All search results were imported into Covidence Systematic Review Software (Veritas Health Innovation Ltd, Melbourne VIC 3000, Australia) for review. The literature was reviewed based on pre-established inclusion and exclusion criteria (described below) by two independent reviewers. A third independent reviewer resolved any conflicts.
Inclusion and Exclusion Criteria
Studies of people living with HIV aged older than 18 years, with either HIVAN or HIVICK (diagnosed clinically or histologically) and receiving cART as a treatment modality for HIVAN or HIVICK were included for review. The case definitions of HIVAN and HIVICK in each study was also considered to ensure data consistency
Quality Appraisal and Data Extraction
All included studies were evaluated for study quality independently using a quality assessment tool, Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Qualitative Research [summarized in Table S5]. This tool is designed to validate internal studies designed for critical appraisal. 14 The review was conducted by two independent reviewers and any discrepancy was resolved by a third reviewer. A standardized data collection format, using Microsoft Excel (2021) was used to extract and summarize relevant information from individual articles for both HIVAN and HIVICK, including the author's name, sample size, age, ethnicity, use of cART, and various laboratory results.
Data Analysis and Synthesis
The study data were analyzed based on outcomes of each study. Three reviewers created keywords based on selected studies to analyze the outcomes. Interpretive synthesis, that allows for methodical analysis and interpretation, allowed methodical data analysis interpretation. 15 The studies with words including “improvement in outcome,” “decreased mortality,” or “delay in dialysis,” were considered positive outcomes compared to words including “worsening outcome,” “no improvement,” or “no change.” All keywords were identified by two independent reviewers (HA and NA) after detailed reading of each included article. Any disagreement was resolved with the help of third reviewer (AK). Furthermore, all similar study observations were aggregated into categories. After detailed discussion of the synthesized findings, conclusions were derived by all co-authors and data analysis was done on the included studies.
Results
The initial database search yielded 4819 articles. After screening for duplications, 119 articles were selected for eligibility screening. Eventually, 11 articles were selected based on the inclusion criteria, as depicted in the PRISMA flow diagram (Figure 3). Data from all included articles were extracted (details outlined in Tables 1 and 2).

PRISMA flow chart.
Summaries of all the Included Studies for HIVAN.

Key:
Summaries of all the Included Studies for HIVAN or HIVICK.

Key:
A total of 11 studies, eight with data from subjects with HIVAN alone, and three with data from subjects with either HIVAN or HIVICK were included. The total sample size was 787 subjects [692 HIVAN patients (11 studies), 95 HIVICK subjects (three studies)]. The mean ages of HIVAN patients ranged from 36.1 to 45.4 years [10 studies] and with HIVICK 42.6 to 44.8 years [three studies]. Of subjects with HIVAN, 94.40% were African American (AA), and 5.60% were of other ethnicities [based on nine studies, Table S6]. Of subjects with HIVAN, 77.37% were proven via biopsy, and 22.65% were diagnosed clinically [based on seven studies]. However, for HIVICK, 100% were biopsy-proven [based on two studies]. Only seven of 11 HIVAN-related studies specified the exact number of patients receiving ART (48.7%) (Table S6).
Of 11 studies including 692 subjects addressing HIVAN, nine showed decreased progression to ESKD or death for subjects treated with cART versus those untreated. Atta et al (2006) clearly stated in his article
16
: “Renal survival was significantly higher in group treated with ART compared to untreated group. Median renal survival was 552 days (18.4 months) for treated group compared with 117 (3.9 months) for the untreated group.”
However, two studies showed no survival advantage with cART. As stated by the authors of the two articles, Abott et al (2003) and Bige et al (2012) reported, respectively17,18: “Neither the use of multiple antiretroviral drugs (AHR, 0.62, 95% CI, 0.10, 3.86, p = 0.06), or ACE inhibitors were associated with a survival advantage”. “Renal outcome was poor despite the fact that the patient received HAART after HIVAN diagnosis and a half of them achieved viral suppression in the peripheral blood during follow-up”.
Nonetheless, it is important to note that, Abott et al. 17 mainly focused on small post-ESKD patient population (N = 31) who received the earlier regimens and for shorter duration (1996-2000), these factors likely have underpowered the study to show significant survival benefits.
All three studies addressing HIVICK showed improvement in delaying ESKD in subjects on cART, compared to untreated subjects. For example, Booth et al (2016) reported
11
: “Patients with HIVICK who initiated ART and achieved suppression of HIV RNA experienced improvements in estimated glomerular filtration rate and proteinuria.”
Discussion
This systematic qualitative review analyzes the impact of cART in individuals with HIVAN and HIVICK. Of the Eleven studies meeting our eligibility criteria, nine studies addressed HIVAN, while three studies addressed HIVICK and HIVAN. Atta et al showed the significance of cART treatment in HIVAN patients. Thirty-six HIVAN patients were studied, with subjects divided into group I (receiving cART) versus group II (not receiving cART). Compared to group II, significantly increased renal survival was seen in group I (univariate, p = 0.025 and multivariate, p < 0.05). This study provided crucial data favoring cART in individuals with HIVAN and its positive impact on renal function, irrespective of CD4+count and plasma viral load. Early detection and treatment in HIVAN patients played an essential role in preventing renal loss. 16
A retrospective study by Halle et al investigated 156 people living with HIV with CKD, of whom 27.6% were HIVAN patients. 48.7% of subjects received cART and were followed for 12 months. Unfortunately, the authors did not specify how many subjects with HIVAN received cART. However, the study showed significant renal function improvement and delayed mortality and morbidity (p = 0.021). 19 Similarly, Post et al conducted a multi-center retrospective study on 61 subjects with HIVAN. After a median 4.2 years, there was significant improvement in overall patient survival and renal outcomes, which were associated with HIV RNA suppression (p = 0.001). There were no additional benefits reported in patients with early cART initiation. 20 In addition, Cosgrove et al and Lucas et al showed statistically significant improvement in renal function (based upon serum creatinine measurements p = 0.027 and p < 0.001, respectively) in patients receiving cART versus those not on cART.21,22
On the other hand, Abbott et al observed no survival advantage in their cohort study reviewing the United States Renal Data System (USRDS). The investigators studied 36 subjects with HIVAN, of whom 22 patients received single-agent ART and nine received cART. The study found no significant survival advantage with cART (p = 0.06). The authors attribute their findings to a relatively short study duration (four years) and fewer subjects receiving ART. Additionally, the authors noted underutilization of cART, early NRTIs (stavudine and zalcitabine) and PIs (indinavir and saquinavir) and angiotensin converting enzyme (ACE) inhibitors (medications known to improve outcomes in HIVAN patients) in their subjects which might have underpowered the results and failed to show the survival benefits of newer and robust therapies available. 17 Similarly, Bigé et al analyzed 57 HIVAN patients who received cART between January 2000 and July 2009. On follow-up, 58.8% of the patients had ESKD, and the median renal survival was 40 months. There was no significant association between use of cART and better renal outcome, irrespective of the viral load. According to the authors, this could be attributed to renal dysfunction in patients, delayed HIVAN diagnosis (>1 year) and/or delayed initiation of ART. However, patients who also received renin-angiotensin system (RAS) blockers had better renal survival (p < 0.05). 18
Three studies focused on both HIVICK and HIVAN. Booth et al conducted a multi-center retrospective study on 65 HIVAN and 55 HIVICK patients diagnosed by renal biopsy. Forty-eight weeks after initiation of cART, there was a significant drop in proteinuria in both groups [89% (p = 0.003) and 100% (p = 0.009), respectively]. 28.1% (9/32) of HIVAN patients progressed to ESKD compared to progression seen in HIVICK patients [7.3% (3/41)]. 11
Another retrospective cohort study by Da Silva et al was conducted on 61 people living with HIV, of whom 14 had HIVAN and 14 had HIVICK. All subjects with HIVAN received cART. At the end of two-year follow-up, there were significant delays in progression to ESKD in both populations. The investigators did not specify outcomes for individual groups. 23 Szczech et al conducted a multi-center retrospective cohort study with 89 people living with HIV (42 with HIVAN). Only 11 of the subjects with HIVAN were reportedly taking cART; despite this, cART use resulted in a trend toward decreased progression to the use of RRT (p = 0.06). 24
Secondary outcome measures, including proteinuria, viral load, CD4+ count, and eGFR were utilized in defining the prognosis in HIVAN and HIVICK. Some studies utilized viral load as one outcome predictor for HIVAN, as HIV is known to invade renal epithelial cells directly. Interestingly, Bige et al demonstrated presence of persistent replicating virus in the renal epithelium that was difficult to control with cART alone (despite undetectable plasma viral loads), leading to poor renal outcomes despite using cART. 18 This could delay cART initiation. Additionally, they highlighted the importance of early diagnosis and treatment before the development of irreversible renal injury. 18
Our qualitative analysis is consistent with previous studies that demonstrate improvement in renal function with the initiation of cART. Bookholane et al conducted a long-term single-center prospective study in South Africa. A total of 246/419 patients were identified with biopsy-proven HIVAN. 163 patients with HIV received cART (although the study did not specify how many HIVAN patients received cART). HIVAN diagnosis was associated with mortality [adjusted hazards-ratio (HR) 1.77, Confidence Interval (CI): 1.07–2.91, p = 0.025], and cART use was shown to be protective (adjusted HR: 0.52, p = 0.0008). Moreover, the study concluded that the proportion of biopsy-proven HIVAN patients declined over time, likely due to greater cART availability. 25
Evolution of ART
There have been substantial advances over the past four decades in antiretroviral formulations, and their utilization in people living with HIV (Figure 1). In 1987, the first class of ARVs-the nucleoside analog reverse transcriptase inhibitors (NRTIs) were introduced. 26 As additional NRTIs were developed, emerging viral drug resistance, toxicities and lower potency limited their clinical impact. 27
Succeeding decades brought introduction of newer classes of ART, including protease inhibitors (PIs) in 1995 and non-nucleoside reverse transcriptase inhibitors (NNRTIs) in 1996. 28 Triple-drug combinations comprised of two NRTIs plus either a PI or NNRTI became the new treatment standard, which also became known as highly active antiretroviral therapy (HAART) or cART. Such combination regimens introduced in the mid-1990s led to a subsequent marked decline in HIV-associated mortality rates. 29 However, diminished enthusiasm for lifelong cART surfaced due to the growing concern about drug interactions, complex regimes, pill burden, and decreasing quality of life. In 2006, the first FDA-approved single-tablet regimen (STR), was launched. It contained efavirenz, an NNRTI and a combination of dual NRTIs- tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC). 30 With its success, many other STRs were launched as they demonstrated effectiveness in suppressing HIV replication. 31
Other antiretroviral classes, including fusion/entry inhibitors and integrase inhibitors, continued to fortify the HIV treatment armamentarium. Integrase strand transfer inhibitor (INSTI)-containing three-drug regimens are well tolerated and are currently recommended for initial cART by National Institute of Health (NIH) guidelines, USA. 32
Newer potent agents and co-formulated products have resulted in decreased ART pill burden and overall reduced adverse effects. The STRs proved to be safe, convenient, and easy to tolerate, hence reducing selective nonadherence to components of the regimen. 33 In addition, there is lower potential for resistance and cross-resistance with the use of STRs. 34 These critical observations have led to improved outcomes related to viral load. The studies included in our analysis show data from different eras of ART. Therefore, the differences between studies in progression to ESKD and mortality outcomes may reflect different adherence rates to therapy and treatment efficacy. The studies were conducted between 1989–2018 (Table S6), during which time a constant drop in HIV-related mortality was seen.
Despite their excellent efficacy, certain factors may limit the use of STRs, including inability to adjust dosages in patients with renal insufficiency. TDF, lamivudine (3TC), and FTC require dose reduction in patients with creatinine clearance less than 50ml/minute.35–37 Another limitation may be the individual patient's viral resistance profile and intolerance to any component of the STR. 38 Additionally, drug interactions, ART-induced rhabdomyolysis, and metabolic complications may contribute indirectly to kidney injury. 39
This review has some limitations. Due to the absence of some necessary data points (such as lack of data before and after cART initiation), we were unable to perform a meta-analysis. No additional information was obtained on contacting the corresponding authors of the studies. Thus, we pursued a qualitative systematic review. Secondly, due to the limited understanding of the pathogenesis and the heterogeneity of diseases, there is no consensus or guidelines for categorization of HIVICK. As a result, there remains uncertainty as to whether conditions like IgA nephropathy are part of HIVICK.10,11 Moreover, as the diagnosis of HIVAN has evolved over the years, there is a lack of universal case definitions of HIVAN and HIVICK (Tables 1 and 2). Establishing case definition would facilitate the recognition of all clinical variants of HIVAN and allow better screening, treatment, and prevention methods. Lastly, process surveillance measures were absent, resulting in our inability to assess the adherence to cART in the study population.
Conclusion
cART appears to reduce risk of progression to ESKD or death in both HIVAN and HIVICK and should be considered standard of care. Focused prospective studies will help determine the overall risks and benefits of cART in people living with HIV with renal disease.
Supplemental Material
sj-docx-1-jia-10.1177_23259582221089194 - Supplemental material for Impact of Antiretroviral Therapy on Kidney Disease in HIV Infected Individuals – A Qualitative Systematic Review
Supplemental material, sj-docx-1-jia-10.1177_23259582221089194 for Impact of Antiretroviral Therapy on Kidney Disease in HIV Infected Individuals – A Qualitative Systematic Review by Harsha Adnani, Nirav Agrawal, Akshay Khatri, Jaclyn Vialet, Meng Zhang and Joseph Cervia in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Data Availability
Available on reasonable request.
Research Ethics
Our study did not require an ethical board approval because it did not contain human or animal trials.
Contributorship
HA and NA designed the study selection criteria, search keywords, drafted the initial manuscript, performed data analysis, and created the illustrations. AK performed qualitative assessment, revised, and reviewed the manuscript for intellectual content. JV conducted literature search, designed methodology, supervised, and contributed to qualitative assessment. MZ performed data analysis and reviewed the manuscript. JC conceptualized and designed the study, supervised the process, revised and reviewed the manuscript for intellectual content. All authors approved and agreed on the final manuscript as submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
