Abstract
Established antiretroviral therapy (ART) programs in sub-Saharan Africa have well-defined first-and second-line therapies but no standard third-line ART regimen. The impact of third-line ART on patients with multiclass-resistant HIV in resource-limited settings has not been well characterized. We conducted a retrospective review of patients on third-line ART at the University Teaching Hospital in Lusaka, Zambia. We assessed virologic and immunologic outcomes following 6 months of third-line therapy and found among those with a documented viral load, viral suppression (≤1000 copies/ml) at 24 weeks was 95% (63/66) with a mean increase in CD4 count of 116 cells/mm3 and viral suppression of 63% (63/100) by imputation of missing data. This study suggests that third-line therapy is clinically and virologically effective among patients with multiclass-resistance in a resource-limited setting in sub-Saharan Africa.
Keywords
What Do We Already Know about This Topic?
Prior to introduction of dolutegravir (DTG), third-line antiretroviral therapy (ART) regimes typically included raltegravir (RAL), darunavir (DRV-r), and etravirine (ETR). Currently, little data exists on the efficacy of such regimens in sub-Saharan African (SSA) and few countries have clear guidelines or access to third-line care.
How Does Your Research Contribute to the Field?
This study aims to add to the limited body of knowledge on third-line ART’s effectiveness, impact on clinical, immunologic, and virologic outcomes in treatment-experienced third-line patients.
What Are the Implications of Your Research toward Theory, Practice, or Policy?
This research will be beneficial for third-line ART management in SSA and provide insight into management of HIV in resource-limited settings where access to HIV genotypes may be limited and empiric third-line switches may be warranted.
Introduction
The global HIV pandemic continues to impact sub-Saharan Africa (SSA); of the 38 million people living with HIV (PLWH) worldwide, over 25.7 million are in SSA as of 2018. 1 An estimated 1.2 million PLWH are in Zambia, of which 700,000 (58.33%) are women. 2 In 2018 UNAIDS reported the percentage of PLWH among persons 15-49 years old was 11.3%; at its peak, the death rate from HIV/AIDS was 1.02%. 3 Zambia has scaled up ART since 2003, initiated at the University Teaching Hospital (UTH) in the capital city of Lusaka and Ndola Central Hospital in Copperbelt Province.
Despite improved ART access, many patients fail therapy after prolonged exposure. In 2016, the World Health Organization (WHO) updated their guidance on third-line therapy to include darunavir/ritonavir (DRV/r), etravirine (ETR), dolutegravir (DTG), and/or raltegravir (RAL), with 1 or 2 nucleoside reverse transcriptase inhibitors (NRTIs) in adults and adolescents over 10 years old. 4 WHO report highlights increasing resistance to first-line ART in SSA; in Zambia, an estimated 4.3% of individuals on ART for 12-24 months have some ARV resistance. 5 Among those on first-line ART with unsuppressed viral load (VL), 47.3% had non-nucleoside reverse transcriptase inhibitors (NNRTI) resistance and 46.9% had both NNRTI and NRTI resistance. 5 Second-line ART failure rates have increased across SSA; some studies have reported higher failure rates on second-line than on first-line. 6,7 Multiclass-resistant HIV is an emerging threat in SSA; however, rates of sustained VL suppression in PLWH on third-line regimens who failed second-line ART in SSA have not been well characterized. We evaluated measures of viral suppression and CD4 increase in patients started on third-line ART at UTH.
Methods
Study Design and Population
We conducted a retrospective chart review of HIV patients treated with third-line regimens at UTH Advanced Treatment Center (ATC) from January 1, 2012 through December 31, 2015 to evaluate rates of viral suppression. Patients 18 years and older with a baseline VL of ≥1000 copies/ml and receiving darunavir/ritonavir (DRV/r) and/or raltegravir (RAL) and/or etravirine (ETR) on third-line ART for ≥6 months were included.
Data Acquisition
Patient records were screened from paper charts, pharmacy records, and SmartCare (the national electronic medical record). Patients without complete data on third-line regimens, baseline VL, or duration of third-line treatment were excluded. For individuals who met study criteria, demographic and clinical data were extracted and entered into Microsoft Excel 2010.
Laboratory Evaluation
VL suppression was defined as HIV-1 RNA levels ≤1000 copies/mL by week 24. 8 HIV VLs were performed on either: COBAS® AmpliPrep/COBAS® TaqMan® HIV-1 Test, version 2.0 or Abbott real-time HIV-1 (Automated), version m2000sp. According to national guidelines, VLs are recommended 24 weeks after initiation while on or with any change in ARV regimen. Data showed variation in timing possibly due to clinician management decisions, test availability, and clinic attendance. Therefore, VLs done 8 weeks before or after scheduled initial VL at 24 weeks were considered “24 weeks,” while any VL outside this time frame was considered an “any other VL measurement.”
Data Analysis
All statistical analyses were performed using Stata version 13.0 (StataCorp, College Station, TX). Descriptive statistics were calculated and compared between patients with follow-up VLs less than and greater than 1000 copies/mL. Chi-square and Fisher’s exact test were applied to test for statistical significance for categorical variables and Mann-Whitney/Kruskal-Wallis for parametric and non-parametric variables continuous variables, respectively. A sensitivity analysis was conducted where baseline VL was assumed unchanged from third-line initiation to follow-up to impute missing follow-up VL.
Ethical Approval and Informed Consent
The study was reviewed and approved by the institutional review boards at the University of Zambia Biomedical Research Ethics Committee (Ref No. 1243-2020), the National Health Research Authority (NHRA000010/11/11/2020), and the University of Maryland School of Medicine (HP-00090308). Informed consent was waived by the IRBs as this study was a retrospective chart review.
Results and Discussion
Demographic and Clinical Characteristics
A total of 205 patient files were screened for eligibility. Of those, 100 patients met inclusion criteria, patients were predominantly female (52%) with a mean age of 40.5 years at the start of third-line ART (Table 1). ART median duration prior to starting a third-line regimen was 8 years. Median HIV RNA level was 4.91 log10 copies/ml (IQR, 4.22-5.42), with a median CD4 count of 76.5 cells/µl (IQR, 24.3-250.8).
Baseline Characteristics at the Initiation of Third-Line Antiretroviral Therapy.

VL, viral load; PI, protease inhibitor; DRV, darunavir; ETR, etravirine; RAL, raltegravir; RTV (r), ritonavir; ABC, abacavir; 3TC, lamivudine; AZT, zidovudine; TDF, tenofovir disoproxil fumarate; FTC, emtricitabine.
NRTI backbone consisted of *3TC, AZT/3TC, TDF/FTC, AZT/TDF/3TC.
Baseline Genotypes
A total of 92 patients had genotypes available. The most frequently reported NRTI mutations were 184 V, 215F, 67 N, and 70 R (Table 1). Of NNRTI mutations, 181C, 103 N, 190A, and 101E were most frequent. 46I, 54 V, and 82A were the predominant protease inhibitor (PI) mutations. The median number of PI mutations per patient was 3 (IQR, 0-4), NRTI was 4 (IQR, 3-5), and NNRTI was 2 (IQR, 1-3) (Table 1).
Third-Line Drug Regimens
Most patients (90.0%) were maintained on an NRTI backbone, most commonly tenofovir/emtricitabine (TDF/FTC). RAL was the most common third-line agent (94.0%), DRV/r (79.0%), and ETR (42.0%) (Table 1).
Virologic and Immunologic Response
A statistically significant reduction in VL and an increase in CD4 count by week 24 was observed in this cohort (Table 2). Of 100 patients meeting inclusion criteria, 34 had no follow-up VL by 24 weeks, illustrating virologic monitoring challenges. Of 66 patients with available VL by week 24, 63 (95%) had VL <1,000 copies/ml, of 86 patients with any available VL, 85 (99%) were virally suppressed. The median time from the start of third-line therapy to next available VL was 32 weeks (IQR, 19-53). There was a statistically significant mean VL change from baseline in log10 of −2.95 and −2.98 by 24 weeks and next VL (p < 0.01). Mean CD4 count increased by 130.5 cells/mm3 by week 24 and by 217.8 cells/mm3 by week 48 (Table 2 & Figure 1). 92% and 96% of patients achieved VL ≤400 copies/ml; 73% and 77% achieved VL ≤50 copies/ml by 24 weeks and next available, respectively. Sensitivity analysis resulted in lower estimates of suppression at 83% ≤400 copies/ml and 66% VL ≤50 copies/ml. A bivariate analysis of potential factors associated with viral suppression defined as ≤50 copies/ml did not identify any statistically significant differences between the 2 groups at week 24 (data not shown).
Virologic and immunologic characteristics at baseline, week 24, and 48 weeks or next available.

* p < 0.01.
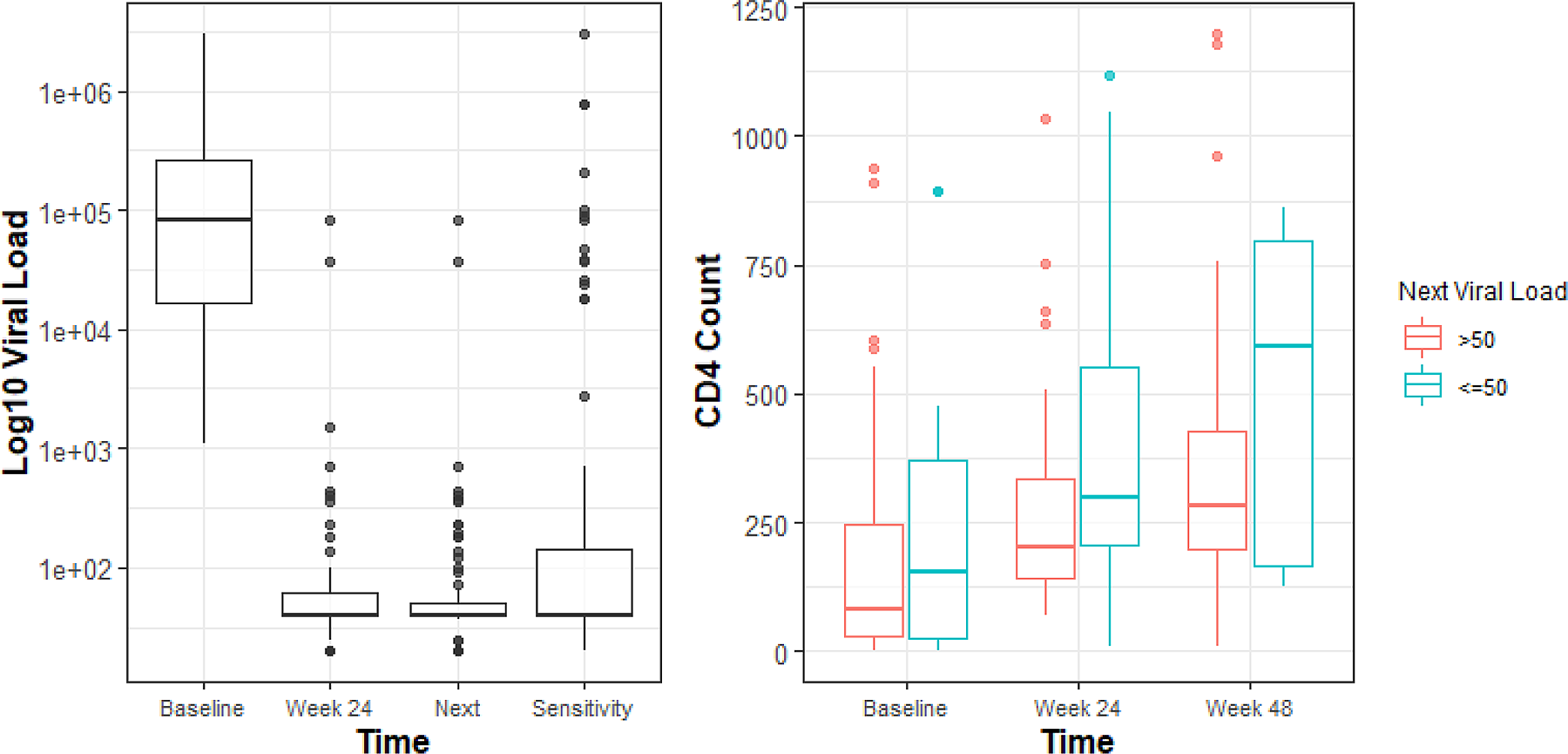
log10 HIVVL at baseline, week 24, 48 weeks or next available, and sensitivity analysis with corresponding immunologic recovery in CD4 counts.
Discussion
In this cohort, among those with a documented follow-up VL, suppression was 95% (63/66) at 24 weeks and 99% (85/86) with any VL. In sensitivity analysis, imputing missing values from baseline found 63% (63/100) viral suppression at 24 weeks and 85% (85/100) suppression among those with any VL. The proportion of patients achieving improved clinical parameters and virologic suppression in our study was comparable to Meintjes et al, who found that 82.9% of patients in South Africa were documented to have suppressed ≤400 copies/ml and 71.1% VL ≤50 copies/ml. 9 Another South African study showed that patients on third-line ART within the first year with at least 1 VL after initiation had 83% and 79% suppressed to below 1000 and 400 copies/mL, respectively. 10 The prospective open-label phase 4 intervention study, conducted at 19 urban sites in 10 countries in SSA, including Kenya, Malawi, South Africa, Uganda, and Zimbabwe, found that 64% of participants achieved viral suppression after 48 weeks. 11 Comparing our results to high income countries, RAL reduced HIV-1 RNA levels to <400 copies/mL in 72.3% and <50 copies/mL in 62.1% at 48 weeks. 12
There were several limitations to this study; missing data and inconsistent follow-up labs for virologic and CD4 monitoring were notable. Assessing loss to follow-up (LTFU) or death was difficult given missing data or files contributing to a selection bias as data reported is of those who had post-initiation VL. Prior studies on mortality among patients LTFU in Zambia reveal substantial under-reporting among patients on ART with rates of 7%. 13 Lack of standardized third-line therapy and differences in time points of VLs also posed challenges. Despite these limitations, this study underscores an effective third-line ARV program in producing favorable virologic and immunologic outcomes in Zambia.
Conclusions
In this study, over 95% of patients with a follow-up VL attained virologic suppression on third-line regimens including RAL, DRV/r, and ETV. Additionally, the suppression rate was significant even when using viral suppression defined as ≤400 copies/ml, demonstrating 92% and 96% virally suppressed by 24 weeks and next available VL, respectively. We observed a robust increase in CD4 by 24 weeks and 48 weeks, with a median CD4 count of over 300 cells/mm3 in individuals tested at week 48. In the pre-DTG era, third-line ART was an effective approach to achieving viral suppression among patients with multiclass-resistant HIV in Zambia. This study represents a summary of Zambian third-line ART outcomes prior to introduction of DTG; future research should include a repeat analysis of data and outcomes in the DTG era.
Footnotes
Authors’ Note
Paul M. Zulu conceived and designed the manuscript concept, collected the primary data, and led the writing and coordination. Mona-Gekanju Toeque provided critical manuscript revision and contributed to design, analysis and interpretation of data. Lottie Hachaambwa, Lameck Chirwa, Sombo Fwoloshi, Mpanji Siwingwa, Melody Mbewe, Lloyd Mulenga, and Cassidy Claassen aided in initial study design and primary data collection. Kristen Stafford, Brianna Lindsay, and Joelle Rosser aided in data analysis, interpretation, and manuscript development. Lottie Hachaambwa, Lloyd Mulenga, and Cassidy Claassen provided overall supervision and direction for this project. All authors aided in reviewing and editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
