Abstract
Introduction:
A number of individual studies in Africa reporting the prevalence of chronic kidney disease (CKD) among people living with HIV are becoming more common. This analysis aims to combine existing prevalence data from various countries across Africa, providing a comprehensive overview of CKD among adults living with HIV
Methods:
This study was conducted in accordance with the PRISMA guidelines. The research protocol was registered on PROSPERO with the reference number CRD42024629601. Various databases, including PubMed, Cochrane, Science Direct, Global Index Medicus, and Google Scholar, were explored for relevant literature from 2000 to December 10, 2024. The data were initially organized in Excel and then analyzed via STATA V.17. Heterogeneity among studies was assessed through statistical methods such as the Cochrane Q test and I2 statistic, visually represented by forest and Galbraith plots, and managed using a random effects model. Publication bias was evaluated using funnel plots and Egger’s test, with bias correction performed through trim and fill analyses.
Results:
A total of 26 studies involving 15,893 study participants were included in the analysis. The pooled prevalence of CKD among adult HIV patients in Africa was 17.2% (95% CI [13.4, 21.0]), indicating significant heterogeneity (I² = 98.76%, p < 0.001) among the studies. Subgroup analyses were conducted to report the pooled prevalence of CKD on the basis of different diagnostic criteria. Modification of diet in renal disease reported a prevalence of 20.2% ([13.5, 26.8]), the CKD Epidemiology Collaboration reported a prevalence of 12.7% ([7.8, 17.5]), and the Cockcroft-Gault method reported a prevalence of 20.2% ([16.4, 23.9]).
Conclusion:
The prevalence of CKD among adult HIV patients in Africa is a significant concern. These findings highlight the urgent need for regular CKD screenings and standardized diagnostic methods.
Keywords
Introduction
Chronic kidney disease (CKD) is a condition that occurs when the kidneys are damaged and unable to filter the blood properly. This impairment leads to the accumulation of excess fluid and waste in the body, resulting in various complications, such as heart disease 1 is common among individuals living with human immunodeficiency virus (HIV). Highly active antiretroviral therapy (HAART) was developed to manage retroviral infections like HIV and to extend the lives of people living with HIV/AIDS (PLWHA). However, with increased longevity among PLWHA, new health challenges are emerging, including the development of non-communicable diseases like CKD. 2 Direct viral infection, immunological dysregulation, and the effects of antiretroviral therapy are mechanisms contributing to the development of kidney disease in HIV patients. 3 Progressive CKD is linked to several complications, like cardiovascular disease, hypertension, anemia, mineral bone disorders, electrolyte imbalances, and acid-base abnormalities. These complications contribute to high morbidity and mortality and poor quality of life. 4 The risk of CKD progressing to end-stage renal disease is estimated to be 2 to 20 times higher among HIV patients than among the general population. 5
By the end of 2023, approximately 39.9 million people were living with HIV, with around 65% of them residing in Africa. During the same year, HIV-related causes led to 630,000 deaths, and 1.3 million people newly contracted the virus. 6 In Africa, a systematic review of studies conducted in Ethiopia and among young people in sub-Saharan Africa revealed that the pooled prevalence of CKD was 18.10% and 12%, respectively.7,8 However, the adult HIV population faces unique challenges, including differing comorbidities, treatment responses, and healthcare needs that are not fully addressed in studies focused on younger individuals. This analysis aims to combine existing prevalence data from various countries across Africa, providing a comprehensive overview of CKD among adults living with HIV. By distinguishing our study from previous youth-focused research, we seek to contribute novel insights into the prevalence of CKD, thereby informing context-specific and effective strategies for its prevention and management within the adult HIV population in Africa.
Methods
Study setting, design, and protocol
This systematic review and meta-analysis were conducted for cross-sectional studies published from 2000 to December 10, 2024 in Africa. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 checklist (S1) 9 was used throughout the review process. The study protocol was prospectively registered on PROSPERO with registration number CRD42024629601.
Search strategy
The literature was searched across various databases, including PubMed, Cochrane, ScienceDirect, Global Index Medicus, and Google Scholar, to identify relevant articles published from 2000 to December 10, 2024. Different search terms related to CKD (“chronic kidney disease” or “CKD” or “renal impairments” or “renal insufficiency” or “chronic renal disease” or “kidney impairment” or kidney insufficiency” or “chronic renal failure”) and HIV (“human immunodeficiency virus” or “HIV” or “AIDS” or “acquired immunodeficiency syndrome” or “HAART” or “highly active antiretroviral therapy” or “ART” or “antiretroviral therapy”) were used.
Eligibility criteria
Cross-sectional studies that defined CKD according to Kidney Disease: Improving Global Outcomes (KDIGO) guidelines and published articles in English were included. Articles that were not fully accessible, did not measure the desired outcomes, or had poor quality scores were excluded. Gray literature, conference abstracts, posters, and clinical trials were excluded.
Outcome of the study
The main outcome of interest was the prevalence of CKD among adult HIV patients in Africa. On the basis of the KDIGO guidelines, 10 CKD was defined as an estimated GFR less than 60 ml/min/1.73 m2 with or without proteinuria or greater than 60 ml/min/1.73 m2 with proteinuria.
Study selection and quality assessment
The search results were managed, and duplicate records were identified via Endnote v.20 (Thomson Reuters, Stamford, CT, USA). Two investigators (GD and MA) independently screened the titles and abstracts of the studies identified through the literature search. Full-text articles deemed potentially eligible were retrieved and assessed for final inclusion. Any disagreements were resolved by consensus or consultation with a third reviewer (TT). To assess the methodological quality of the studies included, a 9-point rating system quality assessment tool, originally developed by Stanifer et al. 11 was used for this research. The scoring criteria examined elements concerning the representativeness of the study participants, sample size, sampling methods, and assessment of possible confounders to the relationship between HIV and CKD. The studies were classified as having high, moderate, or low methodological quality on the basis of scores exceeding 6, falling between 5 and 6, or being 4 or below, respectively. All high- and medium-quality studies were included in this systematic review and meta-analysis (Table 1).
General characteristics of the included studies among adult HIV patients.

Note. CG: Cockcroft-Gault, CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration, HIV: Human immunodeficiency virus, MDRD: Modification of Diet in Renal Disease.
Data extraction
Data extraction was conducted independently by two authors, GD and HA. Discrepancies between their evaluations were rare, and when they arose, the issues were addressed by consulting the other authors, MA and RB, to reach a consensus. The extracted data included the first author’s name, publication year, country, sample size, sex, mean age, method for defining CKD, and number of CKD cases.
Statistical analysis
The data were initially entered into Excel and then exported to STATA V.17 for analysis. The pooled prevalence of CKD was estimated using the Der-Simonian and Laird random-effects model with a corresponding 95% confidence interval. Heterogeneity among the included studies was assessed using the Galbraith plot, and the I² statistic with values of 0%, <25%, 25% to 75%, and >75% were interpreted as no, low, moderate, and high heterogeneity, respectively. 12 To explore potential sources of heterogeneity, subgroup analyses and univariate meta-regression models were conducted. A sensitivity analysis was also performed to evaluate the influence of individual studies by sequentially omitting each study to identify potential outliers. Publication bias was examined using a funnel plot and Egger’s test. 13
Results
Search results
The PRISMA 2020 flow diagram was used to identify, screen, and include relevant studies. A total of 5,542 articles were initially retrieved from various databases covering the period from 2000 to December 10, 2024. Of these, 126 duplicates were removed using EndNote. Following the screening of titles and abstracts, 107 articles remained. Subsequently, the articles were assessed for eligibility, and 26 were included for analysis (Figure 1).

PRISMA 2020 flow chart illustrating the selection of studies for the systematic review and meta-analysis of the prevalence of chronic kidney disease and associated factors among adult HIV patients in Africa.
Characteristics of the included studies and study participants
The included studies reported on 15,893 HIV-infected adults across 14 countries. Specifically, there were nine studies from Nigeria,14–22 three from Tanzania,23–25 and two each from Ethiopia26,27 and South Africa.28,29 Additionally, there was one study from each of the following countries: Cameroon, 30 Burundi, 31 DR Congo, 32 Ivory Coast, 33 Benin, 34 Ghana, 35 Kenya, 36 Malawi, 37 Namibia, 38 and Uganda. 39 These studies reported a total of 2,418 CKD cases. Glomerular filtration rate estimation was done using the Modification of Diet in Renal Disease (MDRD), the Chronic Kidney Disease Epidemiology collaboration (CKD-EPI), and the Cockcroft-Gault (CG) equations, with six studies using at least two equations to estimate GFR.18,24,30–32,36 Ten articles used only the CKD-EPI equation,16,22,23,25–28,33,38,39 six studies used the MDRD equation,14,17,20,21,29,35 and four used the CG equation.15,19,34,37 The overall mean age of the study participants was 38.9 years, ranging from 31.4 to 47. The sample sizes ranged from 103 to 2,468 (Table 1).
Prevalence of CKD
The pooled prevalence of CKD among adult HIV patients in Africa was 17.2% (95% CI [13.4, 21.0]) with high heterogeneity (I2 = 98.76%, p < 0.001) (Figure 2).

Forest plot showing the pooled prevalence of CKD among adult HIV patients.
Factors associated with CKD
Factors identified as significantly associated with CKD include older age, 16,18,19,21,23,27,29,30,32,34,35,38,39 female gender,24,31,34,35 low BMI,18,24,34 low CD4 count, 16,18,19,22,24,29,30,32,39 and high viral load.26,31 The pooled odds ratios for these factors are as follows: older age 1.04 (95% CI [0.98, 1.10]), female 2.80 [1.58, 4.02], low BMI 2.38 [1.24, 3.51], low CD4 count 1.83 [1.32, 2.34], and high viral load 3.27 [0.51, 6.03].
Subgroup analysis, sensitivity analysis, and publication bias
Statistical tests, including the Cochrane Q test and I² statistics (I² = 98.76%, p < 0.001), along with a forest plot and Galbraith plot (S2), revealed heterogeneity among the studies. Subgroup analyses were conducted based on African Union regions, publication year, sample size, mean age, and methods used to define CKD.
The prevalence of CKD according to the African Union region classification was reported as follows: Central Africa 29.1% (95% CI [2.8, 55.5]), Western Africa 19.6% [13.0, 26.1], Eastern Africa 11.9% [6.2, 17.5], and Southern Africa 10.8% [2.1, 19.6] (S3). The pooled prevalence of CKD according to MDRD, CKD-EPI, and CG was reported as 20.2% [13.5, 26.8], 12.7% [7.8, 17.5], and 20.2% [16.4, 23.9], respectively (Figure 3). Subgroup analysis by mean age indicated that studies with a mean age of 37 years or older had a higher prevalence of CKD compared to those with a mean age of less than 37 years (18.0%, [12.8, 23.2] vs. 15.8%, [10.8, 21.2]) (S4). According to the subgroup analysis based on sample size, studies with a sample size of ⩽400 had a higher prevalence of CKD than those with a sample size of >400 (18.2%, [12.7, 23.8] vs. 15.8%, [9.6, 22.0]) (S5). Additionally, the prevalence of CKD was slightly higher in studies published in 2015 or later compared to those published before 2015 (17.3%, [12.5, 22.1] vs. 17.1%, [9.7, 24.5]) (S6).
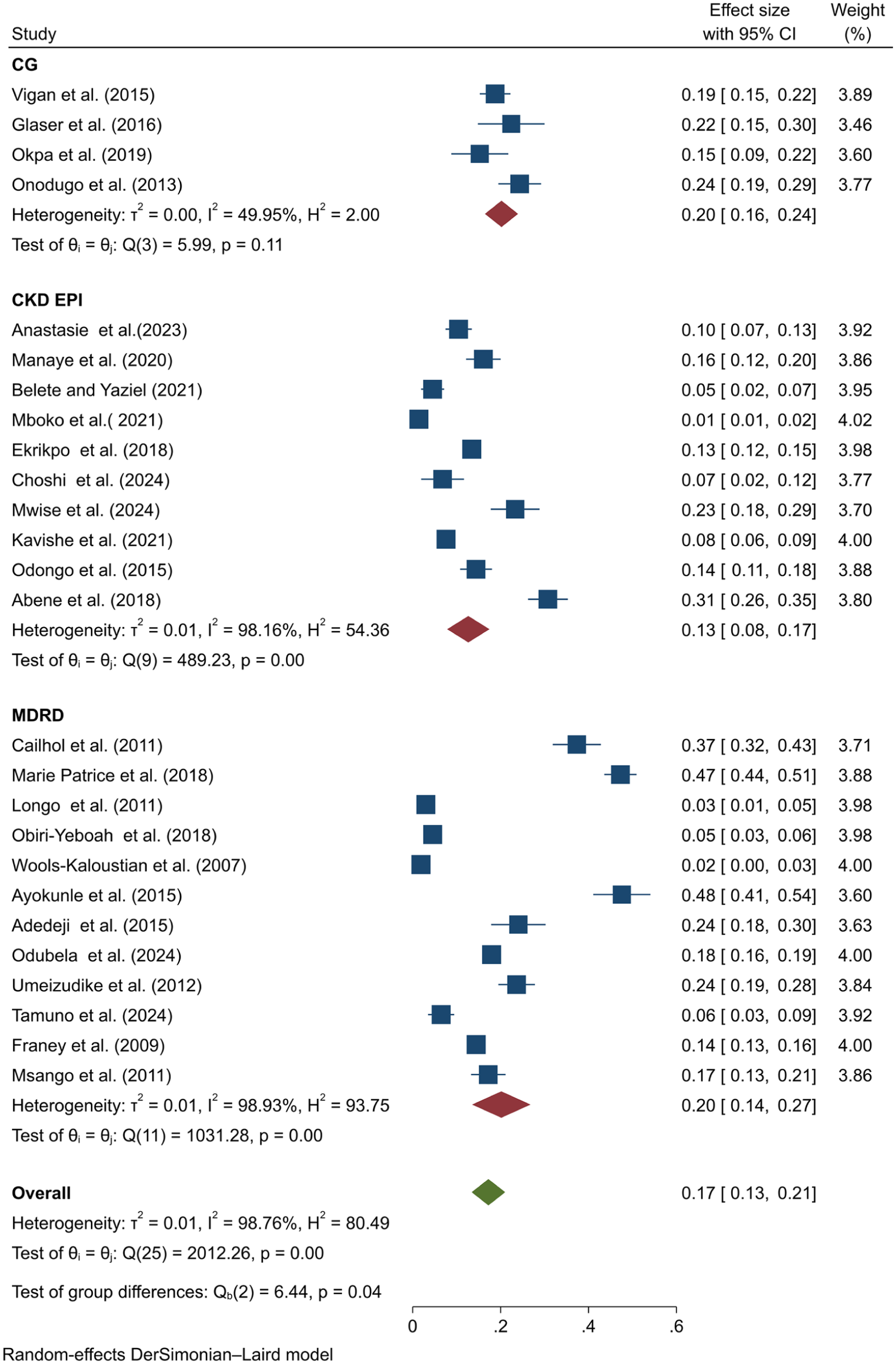
Forest plot for subgroup analysis of methods used to estimate GFR.
Meta-regression was conducted for continuous variables, including year of publication, sample size, and mean age, revealing that these factors did not have a significant impact on the effect size. Additionally, a leave-one-out meta-analysis was performed to evaluate the effect of individual studies on the overall findings of the meta-analysis (Figure 4). The analysis showed that no single study significantly influenced the results, as evidenced by the consistent effect sizes and confidence intervals when each study was omitted individually, with all p-values remaining significant. Both a visual funnel plot (Figure 5) and Egger’s test (p < 0.001) indicated the presence of publication bias. A nonparametric trim-and-fill analysis was conducted, and the pooled estimate remained unchanged despite the detection of bias.

Sensitivity analysis showing the prevalence of CKD among adult HIV patients.
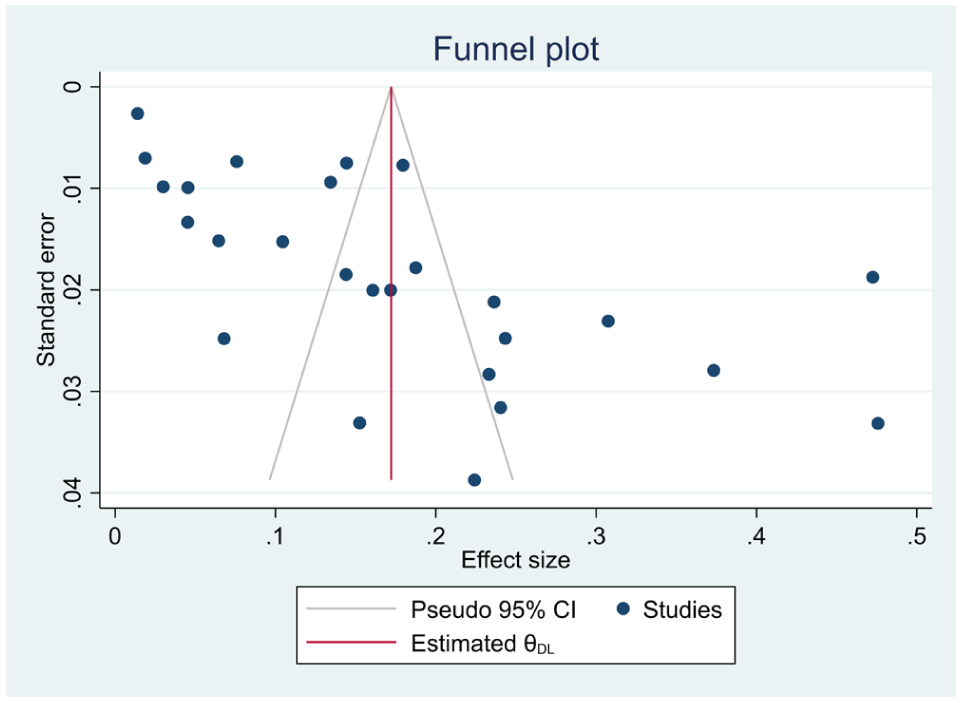
Funnel plot showing publication bias for the prevalence of CKD among adult HIV patients.
Discussion
The study aimed to determine the prevalence of CKD in adult HIV patients across Africa. The pooled prevalence in this demographic was found to be 17.2% (95% CI [13.4, 21.0]). The finding emphasizes the significant burden of CKD within this specific group. The elevated prevalence of CKD in this population can be due to comorbidities and late presentations for testing. 6 HAART plays an essential role in lowering the risk of renal disease by reducing viral replication because viruses themselves have the potential to induce kidney disease by infecting renal cells. 40 Nevertheless, certain antiretroviral medications, such as tenofovir disoproxil fumarate, atazanavir, indinavir, and lopinavir, have been associated with a range of renal complications, including acute kidney injury, CKD or its progression, formation of kidney stones, and Fanconi syndrome.3,41
Several studies conducted in Africa define CKD using the MDRD, CKD-EPI, and CG equations. Based on these definitions, this study revealed that the pooled prevalence of CKD according to the MDRD and CG equations was higher than that determined using the CKD-EPI equation. The prevalence reported based on the CKD-EPI equation (12.7%) is likely more accurate and modern compared to the higher estimates based on MDRD.
The observed pooled prevalence was consistent with the findings of a systematic review and meta-analysis conducted in sub-Saharan Africa 12% (95% CI [6.0, 19.5]) 8 and Ethiopia 18.10% ([14.17, 22.03]), 7 as well as with other cross-sectional studies conducted in Nigeria (15.30%, 15 13.4%, 16 and 16.70% 18 ), South Africa 14.4%, 29 Tanzania 17.2%, 24 and Uganda 14.4%. 39 However, the observed prevalence was higher than the global prevalence of CKD among people living with HIV, which was 6.4%. 42 This discrepancy could be due to financial constraints in Africa and the fact that many studies define CKD based on a single estimated GFR, which may overestimate the true prevalence. Furthermore, comorbidities and population characteristics may contribute to the observed differences.
The study revealed that studies with a mean age ⩾37 years had a higher prevalence of CKD than those with <37 years (18.0%, 95% CI [12.8, 23.2] and 15.8%, [10.8, 21.2]). This could be attributed to age-related factors such as the gradual decline in the number of nephrons, which are influenced by oxidative stress and inflammation, leading to a reduced filtration capacity and a lower GFR. Age-related structural changes in the glomeruli, such as thickening and sclerosis, further impede filtration efficiency, contributing to decreased GFR.43,44
Limitation of the study
The study has several limitations. First, some of the included studies used a single-point measurement of glomerular filtration rate, which may lead to an overestimation of CKD prevalence because persistence (⩾ 3 months) could not be confirmed according to KDIGO guidelines. Additionally, significant heterogeneity among the studies and potential publication bias could influence the conclusions.
Conclusions
The results of this review show that CKD is a public health problem among HIV-infected individuals in Africa. Almost one in six patients with HIV develops CKD. These findings highlight the urgent need for regular CKD screenings and standardized diagnostic methods. Therefore, integrating kidney function testing into routine ART monitoring should become the standard of care. Moreover, it is important to develop prevention strategies tailored to the specific needs of different regions within Africa, ensuring the allocation of resources and the development of guidelines to support CKD prevention and management within HIV programs.
Supplemental Material
sj-docx-1-smo-10.1177_20503121261419300 – Supplemental material for Prevalence of chronic kidney disease and associated factors among adult HIV patients in Africa: A systematic review and meta-analysis
Supplemental material, sj-docx-1-smo-10.1177_20503121261419300 for Prevalence of chronic kidney disease and associated factors among adult HIV patients in Africa: A systematic review and meta-analysis by Gada Diba, Mesay Arkew, Rebuma Belete, Haftu Asmerom and Tewodros Tesfa in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank all authors of the studies included in this systematic review and meta-analysis.
List of Abbreviations
CG: Cockcroft-Gault, CKD: Chronic Kidney Disease, CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration, HAART: Highly Active Antiretroviral Therapy, HIV: Human Immunodeficiency Virus, KDIGO: Kidney Disease: Improving Global Outcomes, MDRD: Modification of Diet in Renal Disease.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
All authors played essential roles in the conception, study design, execution, data acquisition, analysis, and interpretation of the reported work. They participated in drafting, revising, and critically reviewing the article, and they provided final approval for the version to be published. Furthermore, all authors have consented to the final manuscript’s approval for publication in the current journal and take responsibility for all aspects of this work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article [and its supplemental material].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
