Abstract
Iatrogenic Cushing syndrome (CS) is a well-known complication of treating patients with systemic steroids. More rarely, it has been described in HIV-positive patients on ritonavir (RTV) while using the inhaled corticosteroid fluticasone, which is metabolized through the cytochrome P450 3A4 (CYP3A4) enzyme system. In the presence of RTV, a known CYP3A4 enzyme inhibitor, the interaction can result in impaired metabolism and systemic accumulation of inhaled fluticasone resulting in iatrogenic CS. Iatrogenic CS has been less often described with inhaled budesonide compared to inhaled fluticasone. Therefore, inhaled budesonide is often used as an alternative therapy for patients on RTV to avoid iatrogenic CS. We report the fifth case report of budesonide-induced iatrogenic CS in an HIV-positive patient on RTV. We highlight the importance of early recognition of the syndrome and distinguishing it from HIV lipodystrophy. Finally, we review the literature for cases of iatrogenic CS involving RTV and commonly used steroids.
Introduction
Iatrogenic Cushing syndrome (CS) is typically a systemic side effect of exogenous corticosteroid use. Medications that inhibit the cytochrome P450 (CYP450) enzyme pathway can induce CS by causing serum accumulation of exogenous corticosteroid. Ritonavir (RTV) is a potent inhibitor of CYP450 3A4 and can precipitate iatrogenic CS in patients using inhaled corticosteroids. Prior case studies of iatrogenic CS focused mainly on fluticasone interacting with RTV. We present, to our knowledge, the fifth case report of iatrogenic CS involving inhaled budesonide and RTV and review prior cases. We also discuss the pathophysiology, differential diagnosis and management of the condition in such patients, as well as how it can be distinguished from HIV-associated lipodystrophy. We also review the literature concerning iatrogenic CS involving RTV and commonly used steroids.
Case Report
A 47-year-old well-controlled HIV-positive female (CD4 count of 691/cmm and viral load of less than 20 copies/mL) with chronic obstructive pulmonary disease (COPD) presented to her primary physician’s (PCP) office, describing a 20-kg weight gain over a 6-month period. She had not changed her exercise or eating habits. The patient was diagnosed with HIV 15 years earlier, and her highly active antiretroviral therapy (HAART) regimen consisted of raltegravir, emtricitabine, atazanavir (ATV), and RTV. Shortly before she started to gain weight, she was switched from tenofovir (TDF) to raltegravir after developing Fanconi syndrome, with resolution of this complication.
Her COPD was treated with an albuterol inhaler used as needed, a tiotropium bromide inhaler used daily, and a combination budesonide/formoterol inhaler (160/4.5 µg/act). Her PCP had prescribed the budesonide inhaler to be used 2 puffs twice a day, but the patient had been using it incorrectly, only rarely as needed when she was feeling short of breath. On examination in the office, the patient exhibited central adiposity. Her PCP was concerned for lipodystrophy secondary to her recently modified HAART regimen as raltegravir can cause lipodystrophy. He referred her back to the infectious disease (ID) clinic for evaluation and possible modification of her HAART therapy.
Several weeks later, her ID specialist found her to have a rounded face, a dorsocervical fat pad, and proximal muscle weakness. Upon further history, he noted that prior to her weight gain, she began to correctly use her inhaled corticosteroid twice daily. Iatrogenic CS was suspected and she underwent a laboratory evaluation. Random serum cortisol during the morning office visit was 0.3 µg/dL (normal = 5-23 µg/dL) and potassium was 2.7 mmol/L (normal = 3.5-5.0 mmol/L) despite being on daily 20 mEq oral repletion with potassium chloride. The patient was referred to the endocrinology clinic where her serum cortisol was measured to assess for diurnal variation. Cortisol level in the late afternoon was 1.1 µg/dL, and adrenocorticotropic hormone (ACTH) was 6.7 pg/mL (normal ≤ 10 pg/mL). The next morning, cortisol was 1.3 µg/dL, indicating adrenal suppression. Her inhaled budesonide was stopped, she was started on 5 mg prednisone daily to support the hypothalamic-pituitary-adrenal (HPA) axis and prevent adrenal insufficiency. Over the next 6 months, the patient’s prednisone therapy was tapered and discontinued, and her cushingoid appearance resolved. Seven months after she stopped using budesonide, the patient’s morning serum cortisol was 10.4 µg/dL and ACTH was 21.3 pg/mL, both within normal range, indicating recovery of her HPA axis.
Discussion
Clinical Presentation and Workup
Our patient presented with physical findings consistent with CS, including central obesity, “moon facies” and a “buffalo hump” (dorsocervical fat pad). The differential diagnosis for central adiposity and a buffalo hump in patients on HAART includes HIV-associated lipodystrophy, as discussed below, which can occur in up to 50% of HIV-positive patients. In noniatrogenic cases of CS, serum cortisol levels will be elevated secondary to either an ACTH-dependent or ACTH-independent process, and thus diagnosis is made by checking late-night salivary cortisol or urinary cortisol levels, followed by a dexamethasone suppression test to determine true pathogenesis. However, in cases of exogenous administration of synthetic corticosteroids, as in our patient, measurement of serum and urinary cortisol levels will be low or normal. The 2008 Endocrinology Society Clinical Guidelines recommend 2 late salivary cortisol tests, which can be collected by patients at home, stored in the refrigerator, and mailed to a reference laboratory. A dexamethasone suppression test is unnecessary as the exogenous cortisone provides constant negative feedback to the pituitary gland, suppressing ACTH (Figure 1).

This graphic demonstrates the potential derangement of the Hypothalamic-pituitary-adrenal axis when excess cortisol accumulates via inhaled corticosteroid in the setting of ritonavir (RTV) use. ACTH indicates adrenocorticotropic hormone; CRH, cortisol-releasing hormone; ICS, inhaled corticosteroid.
Pathophysiology
Ritonavir is used in low doses as a component of HAART to “boost” the concentration of another protease inhibitor by inhibiting its hepatic metabolism and increasing its plasma concentration. Ritonavir should be used with caution along with a number of medications that are metabolized by the CYP3A4 pathway, including antiarrythmics, statins, ergot alkaloids, sedatives, and steroids. Inhaled corticosteroids are eliminated largely by first pass metabolism as well as through this pathway (Figure 1). Systemic accumulation of the medication can occur when its bioavailability is increased by impedance of this hepatic metabolism. The effective dose of inhaled and intranasal corticosteroids can also be inadvertently increased through improper administration techniques.
Budesonide can be used as an appealing alternative to fluticasone in patients on boosted protease inhibitors, as it has a milder effect on the HPA axis with regular use. Budesonide has a shorter half-life than fluticasone, on the order of 3 hours compared to 10. It is less lipophilic by an order of 300 and has half of fluticasone’s affinity for the corticosteroid binding receptor. 1,2 Many cases of iatrogenic CS described in the literature resolved once the patient was switched from inhaled fluticasone to budesonide. However, this case demonstrates the potential to overwhelm the CYP450 enzymes with inhibition and cause sufficient plasma accumulation of budesonide to develop signs and symptoms of hypercortisolism. Inhaled beclomethasone has been implicated in iatrogenic CS and is also metabolized via the CYP450 3A4 pathway. 3 However, it is thought to be extensively hydrolyzed by esterases and thus not subject to significant accumulation after inhibition of the enzymes and first-pass metabolism in the liver. Clinical trials are currently underway assessing beclomethasone as a safe alternative to fluticasone and budesonide.
Management
After cessation of the inhaled corticosteroid, replacement with a low-dose oral steroid is required until the HPA axis recalibrates, in this case demonstrated by normal ACTH and serum cortisol levels at follow-up.
Distinguishing Lipodystrophy from CS
Lipodystrophy, a common side effect of several HIV medications (didanosine, stavudine, early protease inhibitors), may cause fat redistribution in a cushingoid pattern such as buffalo hump, increased abdominal girth, and breast enlargement. Additionally, lipoatrophy is often noticeable in the limbs, face, and buttocks. These changes typically persist even after the medication is discontinued and are associated with a significant increased risk of insulin resistance, dyslipidemia, and cardiovascular disease. Lipodystrophy develops in about 40% of patients treated with a protease inhibitor for longer than 1 year, making HIV-associated lipodystrophy by far the most prevalent form of lipodystrophy. 4
While the clinical picture is similar, a practitioner should make note of subcutaneous fat on the face (an abundance being associated with CS), facial plethora, striae, hirsutism, acne, easy bruising, and other subtle physical examination findings and have a low threshold for testing for hypercortisolism and referring to endocrinology.
Literature Review
Iatrogenic CS secondary to exogenous steroid use is by far the most common etiology of CS, and it has been well described in patients receiving protease inhibitors to treat HIV. Most documented interactions have occurred with inhaled fluticasone (Table 1), and the use of fluticasone with RTV is relatively contraindicated. 5 Cases involving budesonide are less common but have been noted in the literature. Gray et al described 3 cases of children in South Africa undergoing HIV treatment with RTV who were concurrently treated with inhaled budesonide for respiratory ailments and developed CS. 6 Frankel and Packer described a case of iatrogenic CS in a patient receiving RTV and oral budesonide and speculated that inhaled budesonide may have potential to cause a similar effect. 7 Kedem et al, Yoganathan et al, and Blondin et al have described cases of interaction between RTV and inhaled budesonide in Israel, United Kingdom, and Canada, respectively, that caused iatrogenic CS. 8 –10 Le Roux et al reported on a case from the United Kingdom in which a patient was suspected to have HIV-associated lipodystrophy for 19 months until CS was diagnosed. 11 We thus present the fifth case description of iatrogenic CS caused by an interaction of inhaled budesonide and RTV in an adult HIV-positive patient.
Summary of English-Language Published Cases of Cushing Syndrome as a Result of Drug Interactions with Ritonavir (RTV).
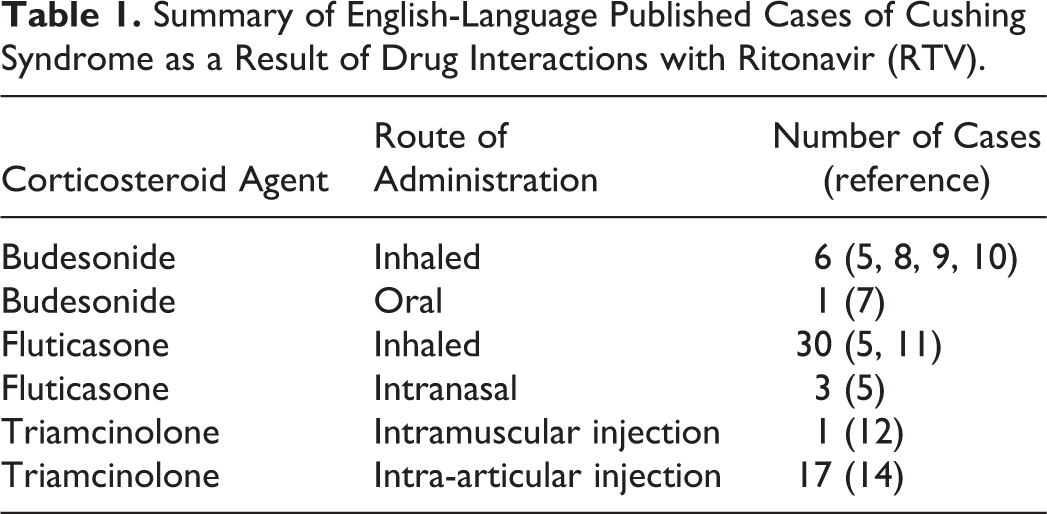
While we classically think of prednisone as the primary offender in iatrogenic CS, patients on RTV developed the condition from inhaled corticosteroids, corticosteroid eye drops, intranasal corticosteroids, and intra-articular joint injections. 12,14
Conclusion
As HIV has become a common chronic disease in the modern medical age, physicians prescribing HAART are increasingly comanaging patients on inhaled corticosteroids for conditions such as asthma and COPD. Respiratory agents were the eighth most common class of drug prescribed in 2015, 13 and inhaled corticosteroids are a therapeutic cornerstone in treating patients with obstructive lung disease. This case represents a significant drug interaction of RTV inducing the development of CS, by the unconventional mechanism of systemic accumulation of inhaled budesonide. We hope to have highlighted the importance of being cognizant of the unexpected drug interactions of a CYP 450 inhibitor such as RTV, especially iatrogenic CS in the presence of corticosteroids.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
