Abstract
Objective
The cortisol concentration in saliva is 10-fold lower than total serum cortisol and accurately reflects the serum concentration, both levels being lowest around midnight. The salivary cortisol assay measures free cortisol and is unaffected by confounding factors. This study analysed published data on the sensitivity and specificity of salivary cortisol levels in the diagnosis of Cushing syndrome.
Methods
Data from studies on the use of different salivary cortisol assay techniques in the diagnosis of Cushing syndrome, published between 1998 and 2012 and retrieved using Ovid MEDLINE®, were analysed for variance and correlation.
Results
For the 11 studies analysed, mean sensitivity and specificity of the salivary cortisol assay were both >90%. Repeated measurements were easily made with this assay, enabling improved diagnostic accuracy in comparison with total serum cortisol measurements.
Conclusions
This analysis confirms the reliability of the saliva cortisol assay as pragmatic tool for the accurate diagnosis of Cushing syndrome. With many countries reporting a rising prevalence of metabolic syndrome, diabetes and obesity – in which there is often a high circulating cortisol level – salivary cortisol measurement will help distinguish these states from Cushing syndrome.
Introduction
Cushing syndrome has an incidence of 13 cases per million people annually 1 and can occur in populations with poorly controlled type 2 diabetes and hirsutism, with or without the presence of polycystic ovary syndrome. 2 Whether screening should be considered in populations without frank signs of Cushing syndrome is a matter of ongoing controversy. 3 One general clinical management approach involves screening individuals with multiple features associated with Cushing syndrome, with progression observed over several months, or with unambiguous clinical signs of the condition (e.g. wide purple striae, proximal myopathy or abnormal patterns of fat deposition). 4 Biochemical assessment of cortisol is always needed in the diagnostic workup of suspected hypercortisolaemic conditions and usually involves venipuncture to obtain a blood sample, or the collection of a 24-h urine sample. 4 On the other hand, salivary sampling is a trouble-free, noninvasive and painless procedure that does not involve stress for the patient. 5 Saliva does not need any special equipment to acquire, therefore it can be obtained quickly and easily by the patient, at home, without assistance (discussed below), and can be stored in the refrigerator at the patient’s home for several days before it is shipped to the clinical laboratory. 6 The salivary cortisol concentration appears to be independent of serum transport proteins such as albumin and cortisol-binding globulin, and is therefore an accurate representation of bioactive free cortisol, 6 in turn helping physicians to understand the usefulness of documenting salivary cortisol levels.
It is interesting to note that salivary cortisol levels follow the diurnal rhythm typical of serum or plasma cortisol levels. 6 This aspect is of paramount importance in diagnosing Cushing syndrome and in distinguishing it from pseudo-Cushingoid hypercortisolaemic states.7,8 For example, diabetes mellitus, obese body habitus and polycystic ovary syndrome – all of which are on the spectrum of dysmetabolic states – can present with increased levels of circulating cortisol or excessive excretion of cortisol in the urine.9–15 However, the key point of distinction is the intact diurnal variation in serum cortisol in these pseudo-Cushingoid states, whereas there is an absence of diurnal variation in Cushing syndrome. The serum cortisol level normally peaks early in the morning and decreases during the day, reaching a nadir between 23.00 h and midnight.16–18 This variation reflects the activity of the adrenal gland in response to environmental conditions, in a normally functioning human adult.
Demonstrating absence of diurnal variation in the serum cortisol concentration is therefore a clinical–biochemical parameter that can be used to confirm the diagnosis of Cushing syndrome, irrespective of any number of confounding factors that may be present.16–18 However, this also poses considerable practical issues, especially the need to obtain blood samples (for serological analysis) around midnight, which is very difficult for both the patient and healthcare personnel. Furthermore, obtaining a blood sample is in itself an invasive procedure that may lead to confounding results being observed: the cortisol concentration may increase due to the stress of blood sampling, but in addition a rise in cortisol may occur because of the technicalities involved in processing the sample (to separate the serum or plasma) immediately after collection. Thus far, no studies have examined the effect of midnight awakening on the serum cortisol concentration. However, it is necessary for patients to remain awake during the period of blood sampling. Herein lies the potential importance of developing a reliable assay for salivary cortisol,16–18 because such procedures would utilize a sample that is easily collected and stored in the home environment. Several studies have shown that saliva samples are well preserved in home-refrigerated conditions, and may be shipped as late as 7 days after being obtained.17,19,20 Importantly, only the midnight sample can indicate the intactness or loss of diurnal variation in circulating cortisol, and thus the serum cortisol sample taken at midnight currently is regarded as the gold standard for distinguishing Cushing syndrome from pseudo-Cushingoid states.
There are several simple means of expressing saliva in the oral cavity. For example, the saliva sample is often collected by drooling or holding an absorbent cotton swab in the mouth until it is saturated. The salivary cortisol assay is therefore convenient not only for patients but also for healthcare or research personnel, as it enables them to collect saliva noninvasively on an outpatient basis (in the patient’s home). Numerous assay techniques have been used to measure salivary cortisol concentrations, including radioimmunoassay (RIA) and liquid chromatography–tandem mass spectrometry (LC–MS/MS). 19 Analytical sensitivity differs between these assay methods, as discussed elsewhere.19,20 Cortisol may cross-react with other steroids, especially topical over-the-counter steroid preparations.18,20 Advice on these aspects can easily be provided at the time of discussing the tests with patients.
The diagnostic accuracy of the midnight circulating cortisol concentration compares very favourably with that of alternative screening tests, such as the 1-mg dexamethasone suppression test and the 24-h urinary free cortisol test. 20 However, obtaining urine samples over a 24-h period presents a number of problems, including a high probability of sample contamination. It is important to be able to demonstrate the loss of diurnal variation in order to detect the confounding presentations of the pseudo-Cushingoid conditions that are now so prevalent. Since a 24-h urine sample is collected in a single container, it is not possible to demonstrate the loss of diurnal variability of cortisol secretion with this method. There are other shortcomings of using the 24-h urinary cortisol test as an indicator of Cushingoid status, as it reflects the intactness of renal function, which may be compromised in conditions such as diabetes mellitus. Furthermore, obtaining a urine sample over a 24-h period also causes considerable aesthetic problems for individuals.
We have argued the case here for the necessity of obtaining a single midnight body fluid sample, to diagnose the presence or absence of diurnal suppression of the circulating cortisol level. This sample needs to be obtained between 23.00 h and midnight. Obtaining a blood sample at this hour is not a practical proposition, leaving the option of obtaining a salivary sample to assay cortisol. The present study provides a critical analysis of published values of the sensitivity and specificity of the salivary cortisol assay for the accurate diagnosis of Cushing syndrome, and makes recommendations for utilizing such assays to diagnose Cushing syndrome accurately and to differentiate it from pseudo-Cushingoid states.
Materials and methods
Data collection
Studies published between January 1998 and December 2012 were used in the present analysis; no language rstrctions were applied. Data contained in studies retrieved through Ovid MEDLINE® were collated and checked for duplication by two independent authors (Q.Z. and J.D.). The rationale for using studies published over this period was that these used the latest methods of either RIA or LC–MS/MS to measure serum cortisol, values of which are usually in the picomolar range and are nearly 10-fold lower than circulating levels. Although some studies reported differences between values obtained using the two assay methods, 19 this was not taken into account in the present analysis.
The following search terms were used: cortisol; cortisol excess; Cushing syndrome; pseudo-Cushing; salivary cortisol; urinary cortisol; serum cortisol; cortisol and confound; cortisol and stress; pain and cortisol; metabolic syndrome and cortisol; sensitivity; specificity; diabetes; depression; syndrome X; hypercortisolism; ACTH; urinary; serum; blood letting and cortisol; McNemar test.
Data analyses
All data analyses were undertaken using SPSS® software, version 19.0 (SPSS®, Chicago, IL, USA). Cumulative databases of sensitivity and specificity in the reported assays of salivary cortisol were created. Analyses of variance were performed in order to examine the variability of the reported sensitivity and specificity values. Each of the calculated variances was considered as an independent data point. These processes, together with estimation of confidence intervals (CIs) of the means (and SDs, which are not reported here), helped to determine whether any of the data points were outliers. Trend lines were created in Microsoft® Excel® software, after plotting the specificity and sensitivity values of the different studies.
Results
The literature search retrieved 11 studies.2,7,8,12,15,19–24 Note that some of the studies did not report either the sensitivity or the specificity data (indicated in Figure 1).
Collated unadjusted sensitivity and specificity values from 11 published studies on the use of a midnight salivary cortisol sample to diagnose Cushing syndrome.2,7,8,12,15,19–24 Note that some of the studies do not report either the sensitivity or the specificity data, not all studies used the same cortisol assay method and two used more than one method (Beko et al.
12
[radioimmunoassay and electrochemiluminescent immunoassay] and Baid et al.
19
[radioimmunoassay and tandem mass spectrometry]). Sensitivity and specificity results for the assay methods used by Beko et al.
12
and Baid et al.
19
were included in the present analyses. The two columns for Baid 2007 represent radioimmunoassay (left) and tandem mass spectroscopy (right); the two columns for Beko 2010 represent the electrochemiluminscence assay and radioimmunoassy (right).
Cumulative sensitivity and specificity of salivary cortisol in diagnosing Cushing syndrome
Cumulative sensitivity and specificity of the use of salivary cortisol concentration for diagnosing Cushing syndrome, derived from the 11 studies shown in Figure 1.
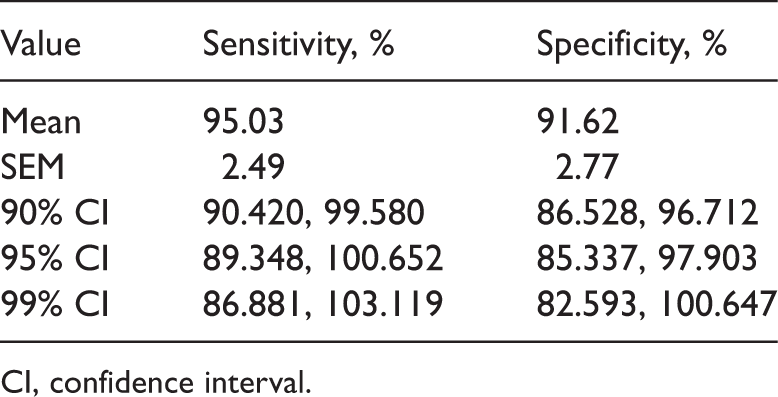
CI, confidence interval.
Graphical representation of collated sensitivity and specificity values
Figure 1 shows the raw sensitivity and specificity values from the 11 studies that were retrieved. Note that not all studies used the same cortisol assay method and some used more than one method. The sensitivity and specificity of each assay method used in each study were included in the present analysis.
Correlation analyses and analyses of variance: Sensitivity and specificity
Analysis of variance showed no significant differences among 10 studies (excluding Baid et al)
19
in the mean values of sensitivity and specificity. Note that all the sensitivity and the specificity values were treated as independent data points (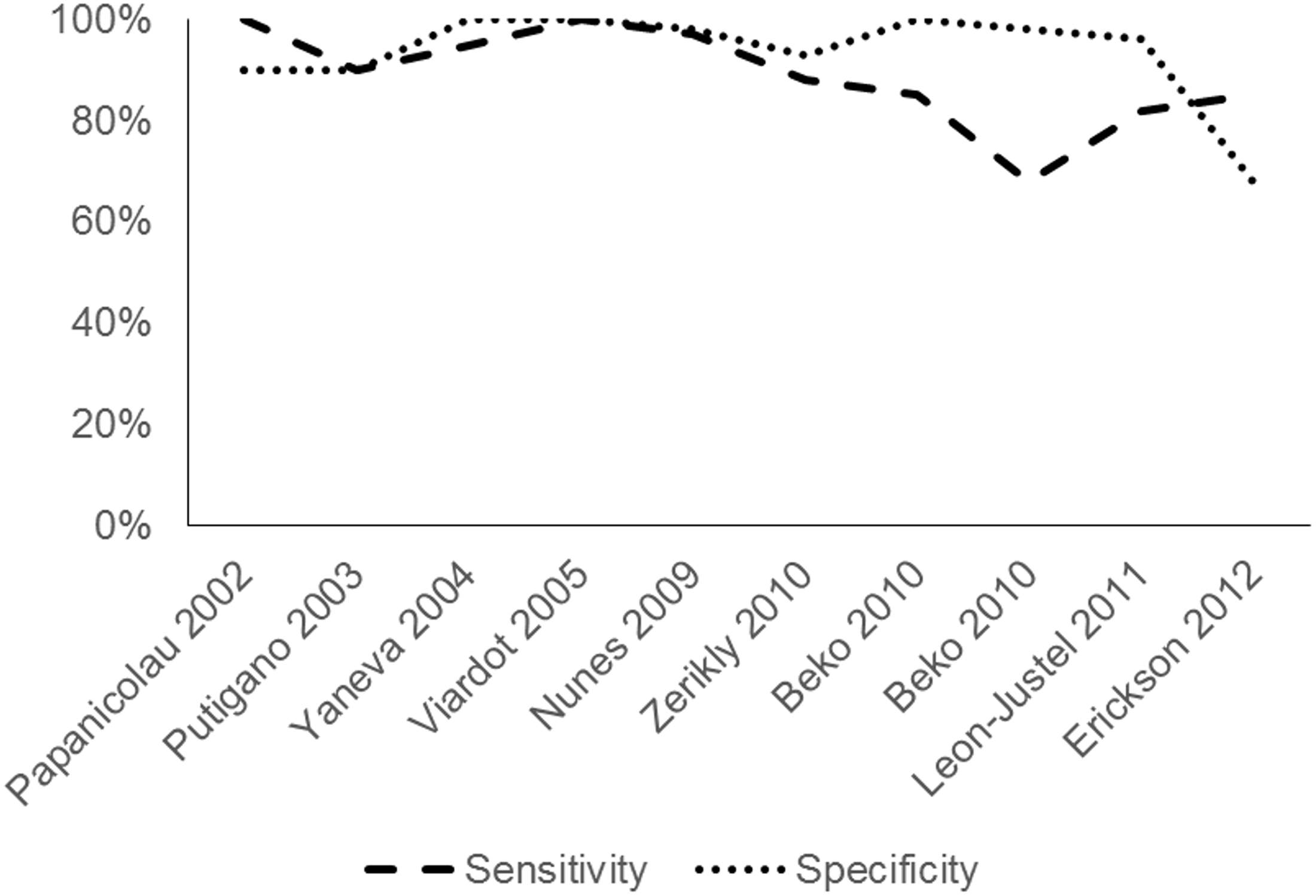
Discussion
Physiological secretion of cortisol is characterized by a daily rhythm, with the circulating cortisol concentration attaining its peak in the morning (06.00–08.00 h) and reaching a nadir at night, during the first half of normal sleep. 6 The normal circadian rhythm of cortisol secretion is absent in patients with Cushing syndrome and an elevated late-night cortisol serum level has been reported to be the earliest and most sensitive marker of the condition.16–18 Thus, analysing the midnight serum cortisol level can be very effective in identifying patients with only mildly elevated levels of cortisol.
Newell-Price et al. 25 analysed plasma cortisol concentrations in blood samples obtained from 150 patients with Cushing syndrome and 20 healthy subjects while they were asleep; the test showed 100% sensitivity. However, sampling blood from sleeping subjects is an impractical approach that can only be performed in a limited research setting, never as a routine clinical assessment. Furthermore, in critically ill in-patients, serum cortisol levels may be elevated due to acute illness and may not be relevant to a Cushingoid diagnosis, 26 thus further highlighting the usefulness of assessing salivary cortisol levels.
Cortisol concentrations in saliva are not dependent on flow rate or secretion dynamics and reflect those in the free fraction of plasma.6,27 Saliva is sampled by noninvasive, stress-free procedures, is stable at room temperature and may be obtained relatively easily at any time during a 24-h period. The present results show that the sensitivity and specificity of the salivary cortisol assay are >90%. This is the first report demonstrating the small amount of variance in published studies of sensitivity and specificity of the salivary cortisol assay in the diagnosis of Cushing syndrome. We recommend obtaining larger series of studies, so that predictive values can be reliably estimated. We consider that analysis of salivary cortisol is a simple and reliable test that could be useful in a number of clinical settings, and in reaching an accurate diagnosis. At present, we are unable to make a recommendation about whether the measurement of salivary cortisol should be used routinely for screening patients with obesity or diabetes, because it does not seem that hypercortisolism needs to be treated in patients with these conditions. In addition, one limitation of our analysis was that we did not strictly abide by the PRISMA guidelines for systematic analysis of publications, which might have affected the overall study findings. However, continued research on this topic would be of clinical interest. The salivary cortisol assay is well suited to repeated measurements, which would increase confidence in the diagnosis of Cushing syndrome. Alcohol intake, major depression and type 2 diabetes mellitus can all present with hypercortisolism but still show diurnal variations in cortisol levels, 28 although more detailed studies are needed to learn the patterns of hypercortisolism associated with these states.
In conclusion, with the increasing prevalence of metabolic syndrome being observed in many countries, we recommend increased use of salivary cortisol tests, which are simpler and easier to perform compared with blood analyses. This would generate a large amount of clinical information that would be useful in defining the baseline cortisol concentration and indicating changes in cortisol levels, in disease processes that have shared phenotypic features.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
