Abstract
Numerous cases of Cushing syndrome have been reported as a result of the interaction between ritonavir (RTV) and exogenous steroid medications. Another complication that frequently occurs is secondary adrenal insufficiency, which can be profound and has not been well described. Here, we report 6 cases of adrenal suppression caused by RTV and exogenous steroids, all of which required corticosteroid replacement therapy and 2 of which were severe enough to require hospitalization. These cases add to the body of literature on the dangerous interaction between RTV and corticosteroids and highlight the risk of secondary adrenal suppression. We also review the literature on this complication and make a recommendation for managing and monitoring such cases.
Introduction
Ritonavir (RTV), a commonly prescribed antiretroviral (ARV) medication in the protease inhibitor (PI) class, is a component of many first-line and salvage HIV treatment regimens. Its primary function is to boost the concentration of other PIs through CYP3A4 enzyme inhibition. This potent blockage of CYP3A4 raises concentrations of other medications that are metabolized by the same pathway, including corticosteroids. 1 Numerous cases of iatrogenic Cushing syndrome or systemic hypercortisolemia have been reported as a consequence of the interaction between RTV and various forms of exogenous corticosteroids, including inhaled, intranasal, injectable, and intraocular preparations. 2 –4 In some of these cases, withdrawal of either RTV or the corticosteroid led to symptomatic suppression of the hypothalamic–pituitary–adrenal (HPA) axis and necessitated corticosteroid replacement therapy.
Here, we report 6 cases in which patients being treated for HIV with a RTV-containing regimen plus a corticosteroid medication developed clinical and/or biochemical evidence of adrenal suppression, all of whom required corticosteroid replacement therapy. Two of these patients developed adrenal insufficiency (AI) that was severe enough to require hospitalization. These cases add to the growing literature on the danger of RTV–corticosteroid interactions and highlight the significant risk of adrenal dysfunction that occurs after the RTV or exogenous steroid medication is withdrawn. We also provide a recommendation for management of such cases.
Case 1
A 44-year-old man (CD4 count 191 cells/mm3) with HIV-related dementia and past history of Pneumocystis jirovecii pneumonia was started on inhaled and intranasal fluticasone for chronic obstructive pulmonary disease (COPD) and allergic rhinitis, respectively. His other medications included tenofovir (TDF)/emtricitabine (FTC), stavudine (d4T), fos amprenavir (FPV), RTV, dapsone, ranitidine, risperidone, metoprolol, and inhaled albuterol.
Following the initiation of the corticosteroids, he began to gain weight and developed a dorsocervical fat pad, purple abdominal striae, easy bruisability, hypertension, and worsening of his baseline anxiety, agitation, and psychosis. Fluticasone was discontinued and replaced with inhaled triamcinolone acetonide, which led to resolution of his symptoms over the next 12 months. Subsequently, he was hospitalized several times for respiratory complaints and received multiple courses of inhaled and systemic corticosteroids. He then developed weakness, malaise, diarrhea, abdominal pain, and weight loss, leading providers to suspect AI. A 250-mcg cosyntropin-stimulation test demonstrated cortisol levels of 0.7 and 4.0 mcg/dL at baseline and 30 minutes, respectively (a 60-minute test was not performed), consistent with the clinical diagnosis of AI. Thyroid function was normal.
Soon thereafter, but before corticosteroid replacement therapy was initiated, the patient was hospitalized for worsening symptoms of AI including malaise, nausea, weakness, polyuria, diarrhea, and salt craving. Intravenous corticosteroids were administered with quick resolution of symptoms, and he was discharged from the hospital on hydrocortisone 60 mg every morning and 30 mg every evening. After 12 months, the dose was tapered to 30 mg every evening and 15 mg every morning, but persistent hypotension prevented further decrease and fludrocortisone 50 mcg daily was added with good effect.
Eighteen months after initiation, oral corticosteroids were discontinued. A repeat 250-mcg cosyntropin-stimulation test demonstrated adrenal recovery with cortisol levels of 6.5, 16.9, and 21.2 mcg/dL at baseline, 30 minutes, and 60 minutes, respectively.
Case 2
A 66-year-old man (CD4 count 562 cells/mm3) with progressive multifocal leukoencephalopathy, seizures, hypertension, hyperlipidemia, coronary artery disease, hypogonadism, osteoporosis, and hemochromatosis was started on inhaled and intranasal fluticasone for COPD. His other medications included efavirenz (EFV), lopinavir/ritonavir (LPV/r), abacavir/lamivudine (ABC/3TC), lamotrigine, topiramate, levetiracetam, furosemide, aspirin, escitalopram, losartan, alendronate, topical testosterone, pravastatin, fenofibrate, tiotropium, and albuterol.
Over 5 months, he developed clinical signs of steroid excess with weight gain, lower extremity edema, central adiposity, peripheral muscle wasting, and weakness. Fluticasone was empirically switched to beclomethasone dipropionate. Ten days later, he was hospitalized for diffuse weakness, mental status changes, and diarrhea. His thyroid function was normal and the morning cortisol level was 2.2 mcg/dL. He was started on hydrocortisone 20 mg every morning and 10 mg every evening, which led to resolution of his symptoms.
He was discharged with instructions to slowly taper the steroids, however, he was subsequently hospitalized multiple times for recurrent Clostridium difficile colitis and the taper was extended. Efforts to decrease the hydrocortisone to less than 10 mg daily were prevented by worsening fatigue and recurrent medical illnesses, and it has yet to be stopped.
Case 3
A 47-year-old man (CD4 count 492 cells/mm3) with coronary artery disease, panic disorder, and history of cytomegalovirus retinitis was being followed for painful cervical radiculopathy. His medications were atazanavir (ATV), RTV, TDF/FTC, oxycodone, aspirin, atorvastatin, ezetimibe, methocarbamol, metoprolol, ranitidine, and isosorbide mononitrate.
He underwent 9 C7-T1 interlaminar epidural injections of triamcinolone acetonide, receiving 480 mg of steroid in total. Over a 10-month period, he developed full facies, dorsocervical fat pad, and abdominal protuberance with purple abdominal striae. Fatigue, thigh weakness, and myalgias worsened after each injection but completely resolved prior to the subsequent injection. After a random cortisol returned low at 1.6 mcg/dL, a 250-mcg cosyntropin-stimulation test demonstrated cortisol values of 0.4, 5.1, and 6.9 mcg/dL at baseline, 30, and 60 minutes, respectively. Thyroid function was normal.
The epidural steroid injections were stopped and when his symptoms began to worsen again, he was started on hydrocortisone 15 mg every morning and 5 mg every evening. His ARV medications were not changed. Due to adverse psychiatric symptoms, the patient stopped the hydrocortisone after a short period of time. Over the next several months, his symptoms slowly abated. Morning cortisol levels measured 1 and 4 months after diagnosis were 1.2 and 2.6 mcg/dL, respectively. At 7 months postdiagnosis, a repeat cosyntropin stimulation test demonstrated partial adrenal recovery with cortisol values of 5.0, 12.3, and 17.1 mcg/dL at baseline, 30, and 60 minutes, respectively.
Case 4
A 54-year-old man (CD4 count 216 cells/mm3) with hepatitis B and asthma was taking TDF, 3TC, ATV, RTV, and fluticasone when he transferred care to our clinic. The exact date at which he had started fluticasone was unclear, but he estimated he had been taking all these medications together for approximately 3 years.
He complained of generalized pain, skin fragility, and easy bruising as well as weight gain of 40 pounds over approximately 6 months, with fat deposition primarily around the face and trunk. New episodes of hypertension were documented in clinic.
Inhaled fluticasone was changed to beclomethasone due to concern for iatrogenic Cushing syndrome. Random cortisol level was less than 0.5 mcg/dL and adrenocorticotropic hormone (ACTH) was less than 5 pg/mL. He returned 1 month later reporting improvement in skin fragility and bruising, stable weight, and improved blood pressure. However, he then began to experience recurrent episodes of nausea and vomiting, raising concern for symptomatic adrenal suppression, so was prescribed 5 mg daily oral prednisone replacement, leading to resolution of his symptoms.
Four months later, he reported continued improvement in skin fragility and weight loss of 5 to 10 pounds. Ten months later, it was noted that his blood pressure had normalized and he had lost 40 pounds. He reported continued return to his normal body shape. Prednisone has not yet been stopped in this patient.
Case 5
A 34-year-old woman (CD4 count 513 cells/mm3) with cervical dysplasia, antiphospholipid antibody syndrome without thrombosis, tobacco dependence, alcohol use, and asthma was started on fluticasone/salmeterol. Her other medications included TDF/FTC and LPV/r.
Two and a half years later, she presented for 70-pound weight gain, lower extremity weakness, and amenorrhea. She was noted to have central adiposity, moon facies, and abdominal striae. Shortly thereafter she also developed nausea and vomiting. Both cortisol and ACTH levels were below the detection limits of the assay.
Fluticasone was changed to beclomethasone, and she was started on prednisone 5 mg daily due to concern for AI. One month later, she reported improvement in her facial and central adiposity, striae, pedal edema, and lower extremity strength, but complained of nausea and vomiting so prednisone was increased to 10 mg daily. One month later, her nausea and vomiting were resolved. One month after that, she continued to lose weight and her menstrual periods resumed. The prednisone was changed to 7.5 mg every morning and 2.5 mg every evening, which the patient took for 2 months and then self-discontinued. A random cortisol 5 months after discontinuation of prednisone was 10.0 mcg/dL. One year after stopping fluticasone, her cortisol had normalized at 18.5 mcg/dL and ACTH had normalized at 24 pg/mL.
Case 6
A 62-year-old woman (CD4 count 687 cells/mm3) with chronic hepatitis C, asthma, allergic rhinitis, osteoarthritis with prior knee replacement, gastroesophageal reflux disease, and depression was taking inhaled budesonide 180 mcg/spray, 2 puffs daily, as part of her asthma regimen. She was started on zidovudine (ZDV)/3TC and LPV/r for HIV, and then 13 months later, her budesonide dose was increased to 2 puffs twice daily due to an asthma flare that required hospitalization.
Five months later, she began to complain of worsening fatigue, depression, anorexia, diarrhea, and weight loss. Six months after doubling her budesonide dose, her cortisol was less than 1.0 mcg/dL and ACTH was less than 5.0 pg/mL, so budesonide was changed to beclomethasone, and prednisone was started at 7.5 mg every morning and 2.5 mg every evening. Pituitary magnetic resonance imaging was normal. She returned to clinic 1 month later, reporting that her energy had improved and she had gained 10 pounds.
Six months later, she self-discontinued prednisone and her random cortisol level was less than 1 mcg/dL so prednisone was restarted at 5 mg daily. Nineteen months after being diagnosed with AI, she exhibited clinical recovery, and prednisone was stopped.
Discussion
These cases underscore the significant risk of interactions between RTV and exogenous corticosteroids. Although numerous cases of Cushing syndrome and a small number of cases of AI have been reported in the literature, the cases presented here are noteworthy because they demonstrate the severity and prolonged effect of adrenal axis suppression that can occur with both inhaled and injectable steroids when combined with RTV (Table 1). In each of these cases, suppression of adrenal function was confirmed with biochemical tests and treatment involved discontinuation of the offending agent plus corticosteroid replacement therapy.
Summary and Management of Adrenal Insufficiency Cases that Resulted from Ritonavir–Corticosteroid Interactions.
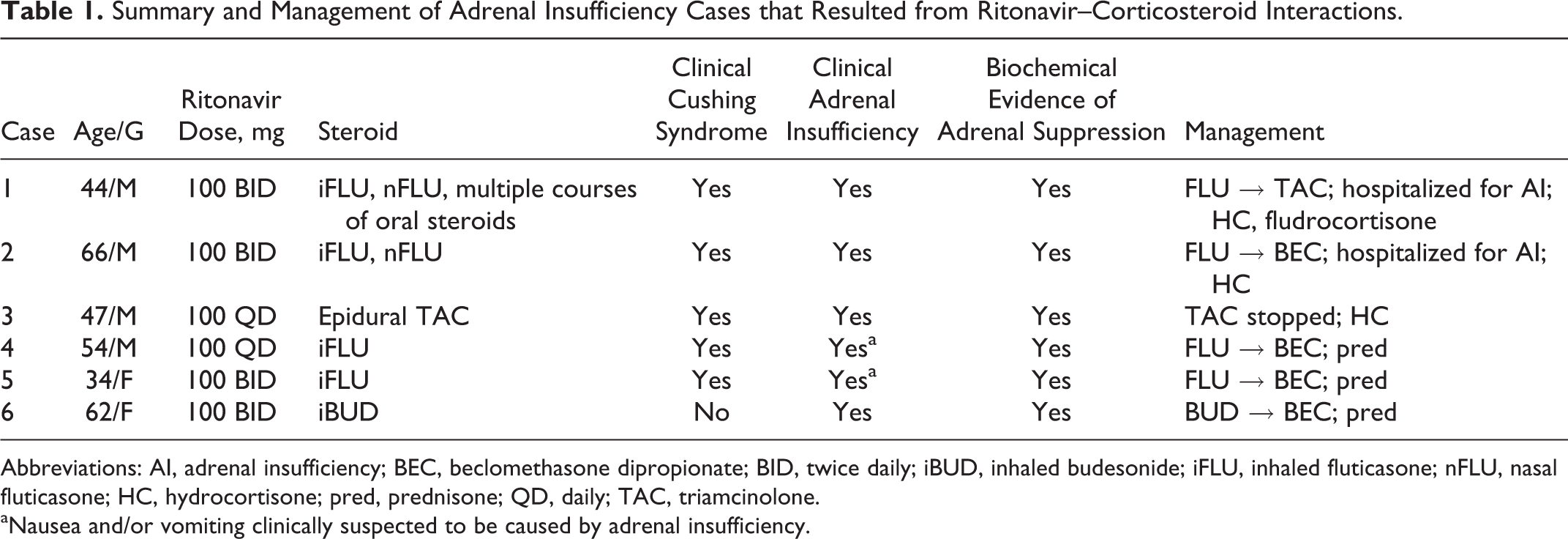
Abbreviations: AI, adrenal insufficiency; BEC, beclomethasone dipropionate; BID, twice daily; iBUD, inhaled budesonide; iFLU, inhaled fluticasone; nFLU, nasal fluticasone; HC, hydrocortisone; pred, prednisone; QD, daily; TAC, triamcinolone.
aNausea and/or vomiting clinically suspected to be caused by adrenal insufficiency.
A 2013 literature search on adverse effects of PIs combined with inhaled and/or intranasal corticosteroids identified 51 published cases. 4 Of note, the PI in 48 (94%) cases was RTV (the rest involved either another PI or the PI was not reported), and the corticosteroid in 46 (91%) cases included inhaled or intranasal fluticasone. The other 5 (9%) cases involved inhaled budesonide, with 1 case of inhaled budesonide plus intranasal fluticasone. Because fluticasone is the most potent of the inhaled corticosteroids and the most dependent on CYP3A4 for metabolism, the risk of complications when combined with RTV is higher than that with other inhaled or intranasal corticosteroids. In the review, 11 (24%) of the 46 cases in which the management was reported required corticosteroid replacement therapy. This suggests that approximately a quarter of cases in which clinically apparent adverse reactions occur secondary to interactions between PIs and corticosteroids will require corticosteroid replacement therapy for secondary AI.
Consistent with our experience, a 2005 report documented 6 cases of Cushing syndrome with osteoporosis caused by RTV plus inhaled fluticasone. All 6 patients developed laboratory evidence of secondary adrenal failure upon withdrawal of fluticasone, and 4 of the 6 patients developed symptoms of AI. Three patients required corticosteroid replacement therapy. 5 Reports such as this highlight that many patients who take RTV with exogenous corticosteroids will develop Cushing syndrome with biochemical evidence of adrenal suppression, and a certain subset will develop symptomatic AI, requiring replacement therapy. The factors that determine which patients will need replacement therapy have not been delineated. This is an area in which further study and prospective trials are needed. Clinicians should avoid the combination of RTV with potent corticosteroids of any form and should be cautious when stopping the medications if Cushing syndrome has developed.
It is important to note that while most reported cases of Cushing syndrome and AI from RTV with exogenous corticosteroids have been caused by inhaled fluticasone, other inhaled steroids and other steroid preparations can also be problematic. For example, cases of AI have been reported following administration of RTV with intranasal fluticasone 6 and with inhaled budesonide, 7 as in the final case presented here. Despite the lower potency of inhaled budesonide compared with fluticasone, coadministration of RTV still requires caution and close monitoring. Other corticosteroid formulations can cause issues as well. For instance, one report documents a case of Cushing syndrome and secondary adrenal suppression due to an interaction between RTV and corticosteroid eye drops, and another describes Cushing syndrome following administration of orbital floor triamcinolone in a patient taking RTV-boosted LPV. 2,8 Furthermore, while most complications develop in a subacute time frame, they can develop rapidly. For instance, a 2007 case report documented onset of Cushing syndrome in a teenager within 2 weeks of initiating inhaled fluticasone/salmeterol while taking RTV. After the withdrawal of the offending agents, the patient required hospitalization and corticosteroid replacement therapy. 9 The duration between initiation of RTV with corticosteroids to onset of symptoms of Cushing or AI is highly variable, as in the cases we report here, but previous reviews have reported a mean time to onset of symptoms of approximately 7 months. 4 The variability in time to presentation may depend on the potency, prescribed dose, and patient adherence to corticosteroid therapy. It should also be noted that while the literature has focused on complications from RTV combined with corticosteroids, cobicistat (which is included in certain fixed-dose combination medications) is also a nonselective CYP3A4 inhibitor and could lead to the same complications if used in conjunction with corticosteroid medications. In addition, PIs besides RTV may raise corticosteroid levels and cause complications, such as in a report of Cushing syndrome secondary to inhaled fluticasone and indinavir. 10
Multiple reports of secondary adrenal failure after epidural or intra-articular steroid injections in patients taking RTV have been described, as in case 3 reported here. This underscores the importance of awareness of this possible complication in any HIV-positive patient being considered for a corticosteroid injection. For example, one report describes a serious reaction in which a 44-year-old HIV-infected man receiving RTV who underwent an intra-articular injection of triamcinolone acetonide for osteoarthritis of the hip then required admission to the intensive care unit for hyperosmolar hyperglycemic state. Seven weeks after the injection, he had evidence of secondary AI with suppressed cortisol and ACTH levels. 11 Similarly, another report describes 3 cases of HIV-infected patients taking RTV who received single intra-articular triamcinolone acetonide injections then developed iatrogenic Cushing syndrome and suppression of their hypothalamic–pituitary axis. 12 The first of these patients required corticosteroid replacement therapy in the form of hydrocortisone 20 mg/d for 8 months prior to adrenal recovery. The second in this series required 10 mg/d of hydrocortisone for 4 months. The third patient in this series required no replacement therapy and experienced spontaneous resolution of the HPA axis suppression after 5 months. A retrospective review of complications occurring after corticosteroid injections in individuals on PIs found an 11% event frequency for complications, with the biggest risk factor being 2 or more corticosteroid injections within 6 months, though it is notable that adverse events have been reported after 1 injection in some patients. 13
There are no formal guidelines for management of patients who develop Cushing syndrome or AI secondary to PI and corticosteroid use. Recognition of the syndrome is the first challenge, as symptoms may initially be mistaken for HIV or ARV-associated lipodystrophy. Once complications are recognized, management involves either changing the PI to a different ARV medication or changing or discontinuing the corticosteroid. For example, changing inhaled fluticasone or budesonide to an alternate inhaled steroid that is less potent and less dependent on CYP3A4 metabolism has been successful in some patients (safer alternatives include inhaled triamcinolone or beclomethasone). 5 However, as documented in this report, some patients will develop symptoms of adrenal suppression after RTV or exogenous steroids are withdrawn. Therefore, we recommend close clinical monitoring of patients after any change of therapy that is done due to evidence of cortisol excess. Given the potential protean manifestations of AI, we advise that patients be well educated on the signs and symptoms that may occur and be advised to seek medical care immediately if they experience any of these. For those who develop clinical evidence of AI (such as weakness, fatigue, nausea, vomiting, or diarrhea), we recommend checking a cosyntropin-stimulation test to confirm adrenal suppression and we recommend corticosteroid replacement therapy. Whether empiric steroids at physiologic doses should be prescribed to those who develop Cushing syndrome and are at risk for AI but do not yet have symptoms is unclear and is a topic that should be explored in future studies.
If corticosteroid replacement is required, there are no unique guidelines for prescribing replacement glucocorticoid therapy for HIV-infected patients. As the cases presented here demonstrate, replacement therapy can be administered in the form of prednisone or hydrocortisone. For adults, one review recommends hydrocortisone at a total daily dose of 15 to 30 mg, divided 2 or 3 times daily, with one-half to two-thirds of the dose in the morning in order to mimic normal circadian cortisol secretion. 14 Prednisolone or prednisone at 5 to 7.5 mg daily is also an option. Cortisone acetate may be an option but availability is limited. 15 For pediatric patients, one source recommends daily hydrocortisone at physiologic dose of 8 to 10 mg/m2/d until morning cortisol normalizes. 16 Clinicians should also be aware of the risk of adrenal crisis in these patients, as they may require supraphysiologic steroid replacement doses during times of acute illness. 15
Determining when it is safe to stop corticosteroid replacement therapy is difficult. An attempt should be made to taper the corticosteroid medication slowly by monitoring for symptoms, recognizing that it can take a year or longer for return of normal function. 17 While some authors have suggested measuring serum cortisol every 4 to 6 weeks to determine whether continued steroid replacement is needed, 4 we do not advise this strategy. Measuring blood cortisol or ACTH levels while the patient is on active glucocorticoid replacement is not advisable since a variety of steroid preparations may be detected by the laboratory cortisol assay and exogenous glucocorticoids, even at presumed physiological doses, may suppress the hypothalamic–pituitary–adrenal axis. To obtain a valid laboratory assessment of the axis, it is necessary to discontinue glucocorticoid administration for a period of time that depends upon both the half-life and the cross-reactivity of the preparation being administered. For example, Sacre and colleagues performed standard 250-mcg ACTH stimulation tests 24 hours after the most recent dose of prednisone in 60 patients who were receiving long-term prednisone therapy for systemic inflammatory disorders. 17 These investigators found that among 29 patients who discontinued prednisone after demonstrating a normal serum cortisol response to ACTH, none had to be restarted on a glucocorticoid for symptoms or signs of AI. We suggest a similar strategy: perform a standard ACTH stimulation test 24 hours after discontinuing hydrocortisone or cortisone acetate therapy; 24 to 48 hours after discontinuing prednisone, prednisolone, or methylprednisolone therapy; or 72 hours after discontinuing dexamethasone therapy. If the serum cortisol value 60 minutes after ACTH administration is greater than 18 mcg/dL, the patient’s glucocorticoid therapy may be discontinued with routine clinical follow-up. If the value is less than 18 mcg/dL, clinical follow-up should be frequent for the first 3 to 4 months after glucocorticoid discontinuation, with a low threshold for restarting glucocorticoid therapy if any symptoms suggesting recurrence of AI develop.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
