Abstract
We carried out a retrospective, multicenter study of a cohort of 298 asymptomatic HIV-infected patients who switched from a regimen based on 2 nucleoside reverse transcriptase inhibitors + protease inhibitor (PI)/nonnucleoside reverse transcriptase inhibitor or ritonavir-boosted PI monotherapy to emtricitabine/rilpivirine/tenofovir disoproxil fumarate (FTC/RPV/TDF) to analyze lipid changes. At 24 weeks, 284 (95.3%) patients were still taking the same regimen, maintaining similar CD4 counts as at baseline (651 versus 672 cells/mm3, P = .08), and 98.9% of them with an undetectable viral load. Eight of the other 14 patients were lost to follow up and 6 (2.0%) ceased the new regimen: 3 due to adverse effects, 2 due to virologic failure, and 1 due to abandonment. The mean levels of fasting total cholesterol (TC), low-density lipoprotein cholesterol, high-density lipoprotein cholesterol (HDL-C), and triglycerides fell at 12 and 24 weeks, with no changes detected in the TC to HDL-C ratio.
Keywords
Introduction
Many therapeutic regimens are currently available to suppress the viral load in patients with HIV, but maintaining a continuous lasting response requires much effort due to adverse effects, long-term toxicity, and the complex treatment regimens involved. 1 Antiretroviral therapy (ART) can induce an increase in lipids levels and metabolic disorders, and there is a well-known relationship between hyperlipidemia and cardiovascular risk (CVR) in the general population. 2,3 On the other hand, the close association between lack of adherence to ART and therapeutic failure has led to recommendations to improve adherence with antiretroviral combinations that are much easier to administer and much preferred by patients. 4,5 Rilpivirine (RPV), a second-generation nonnucleoside reverse transcriptase inhibitor (NNRTI), has demonstrated a better lipid profile compared with efavirenz (EFV) in naive patients. 6,7 A recent randomized study has also shown an improvement in lipid profile after switching from a ritonavir-boosted protease inhibitor (PI/r) regimen to the coformulated emtricitabine/rilpivirine/tenofovir (FTC/RPV/TDF) single tablet. 8 Moreover, this coformulated single tablet improves adherence and has proven its efficacy and safety. 8 –10 Therefore, switching to RPV may be convenient in many patients, maintaining a good immune and virological control. 6 –9 The aim of this study was to analyze lipid and CVR changes in HIV-infected patients at 6 months after switching to FTC/RPV/TDF and to assess the efficacy and safety of the switch.
Patients and Methods
A cohort of 6970 HIV-infected patients was followed prospectively in 12 hospitals from Andalusia, southern Spain, from February 2013 to October 2013. In this open-label, multicenter, noncontrolled study, we retrospectively analyzed the asymptomatic patients from this cohort who switched from a regimen based on 2 nucleoside reverse transcriptase inhibitors (NRTIs) plus a PI/r/NNRTI or PI/r monotherapy to FTC/RPV/TDF. All the patients had to have had an undetectable viral load (<50 copies/mL) for at least the last 3 months prior to switching. Patients with previous virologic failures on ART including TDF and/or FTC/lamivudine (3TC), with genotype tests showing resistance to components of FTC/RPV/TDF, or who changed the third drug of the ART during the study period were excluded. Changes were analyzed at 12 and 24 weeks in the lipid profile, including total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides, and the TC to HDL-C ratio, and the CVR using the Framingham equation. We also assessed the percentage of patients with an undetectable viral load, CD4 responses of the new regimen at 24 weeks, and the percentage of patients who stopped FTC/RPV/TDF due to adverse events.
Institutional review board/ethics committee approval was obtained for this study protocol, for the analysis of anonymous routine clinical data of patients without written informed consent with a view to scientific publication. The study was sponsored by the Grupo de Estudio de SIDA (GESIDA) of the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC) and registered as the GESIDA 8114 Study.
Information for each patient was included in a database for statistical analysis. A descriptive analysis of the variables was done with point estimation and 95% confidence intervals. Continuous variables are shown as the mean plus standard deviation or median depending on the distribution. Categorical variables are shown as frequencies or percentages. The comparison between continuous data at baseline and 24 weeks was done by the Student t test for paired samples, and the comparison between categorical data was done by the chi-square or Mann-Whitney tests. The statistical program used was SPSS version 17.0 (SPSS, Chicago, IL, USA).
Results
Seven of the 305 patients who fulfilled the inclusion criteria were excluded due to lack of data. The study therefore comprised 298 cases. The median age was 44.7 years, and 81.2% were male. The clinical characteristics of the patients at the time of switch are shown in Table 1. Of the 298 patients, 233 (78.2%) switched from a regimen based on 2 NRTI plus NNRTI, with the coformulated single-tablet FTC/EFV/TDF being the prior regimen in 90.5% of these. The most frequent reasons for switching were central nervous system (CNS) adverse events (30.9%), convenience (27.5%), and metabolic disorders (23.2%).
Baseline Characteristics of the Study Patients.a,b

Abbreviations: ART, antiretroviral therapy; NRTIs, nucleoside reverse transcriptase inhibitors; NNRTIs, nonnucleoside reverse transcriptase inhibitors; PIs/r, ritonavir-boosted protease inhibitors; CNS, central nervous system; IQR, interquartile range; FTC, emtricitabine; RPV, rilpivirine; TDF, tenofovir disoproxil fumarate.
aN = 298.
bThe quantitative variables are expressed as median and IQR and the qualitative variables as n (%).
cFixed dose emtricitabine/tenofovir 275 and fixed dose abacavir/lamivudine 12.
dEfavirenz 224, etravirine 12, and nevirapine 7.
eAtazanavir 16, darunavir 11, lopinavir 11, fosamprenavir 8, and saquinavir 4.
fDarunavir 6, lopinavir 1, and atazanavir 1.
gRaltegravir 5, trizivir 1, PI/r plus maraviroc 1.
At 24 weeks after the switch to FTC/RPV/TDF, 284 (95.3%) patients were still taking the same regimen, maintaining similar CD4 counts as at baseline (651 versus 672 cells/mm3, P = .08), and 281 (98.9%) of them with an undetectable viral load. Eight of the other 14 patients were lost to follow up and 6 (2.0%) ceased the new regimen: 3 due to adverse effects, 2 due to virologic failure, and 1 due to withdrawal (Table 2). The mean levels of fasting TC, LDL-C, HDL-C, and triglycerides fell at 12 and 24 weeks (Student t test for paired samples, P < .05 for all comparisons), with no changes detected in the TC to HDL-C ratio. In the 69 patients switching to FTC/RPV/TDF due to metabolic disorders, all the lipid components, including the TC to HDL-C ratio, fell at 24 weeks (Student t test for paired samples, P < .05 for all comparisons). Mean decreases in TC, LDL-C, and triglycerides at 24 weeks were more pronounced in these 69 patients compared with those switching to FTC/RPV/TDF due to other reason (Student t test for independent samples, P < .05 for all comparisons). Changes in fasting lipids from baseline to weeks 12 and 24 are shown in Figure 1. Categorical analyses of fasting lipids demonstrated similar trends (Figure 2). The CVR also decreased (8.7 versus 7.5%, P = .0001). Mean transaminase levels were similar at baseline and at 24 weeks (ALT 36 versus 39 IU/L, P = .1; AST 30 versus 32 IU/L, P = .5). A small increase in mean creatinine level (0.84 versus 0.88 mg/dL, P = .0001) and a decrease in fasting blood mean glucose level (98 versus 94 mg/dL, P = .0001) were noted.
Characteristics of the Patients 24 Weeks after Switching to FTC/RPV/TDF.

aTwo nephrotoxicity, 1 rash.
bEmergence of minor resistance mutations (K70E, M230I) against efavirenz and nevirapine in 1 patient, and no mutations in the other one.

Changes in fasting lipids from baseline to weeks 12 and 24. TC indicates total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides.
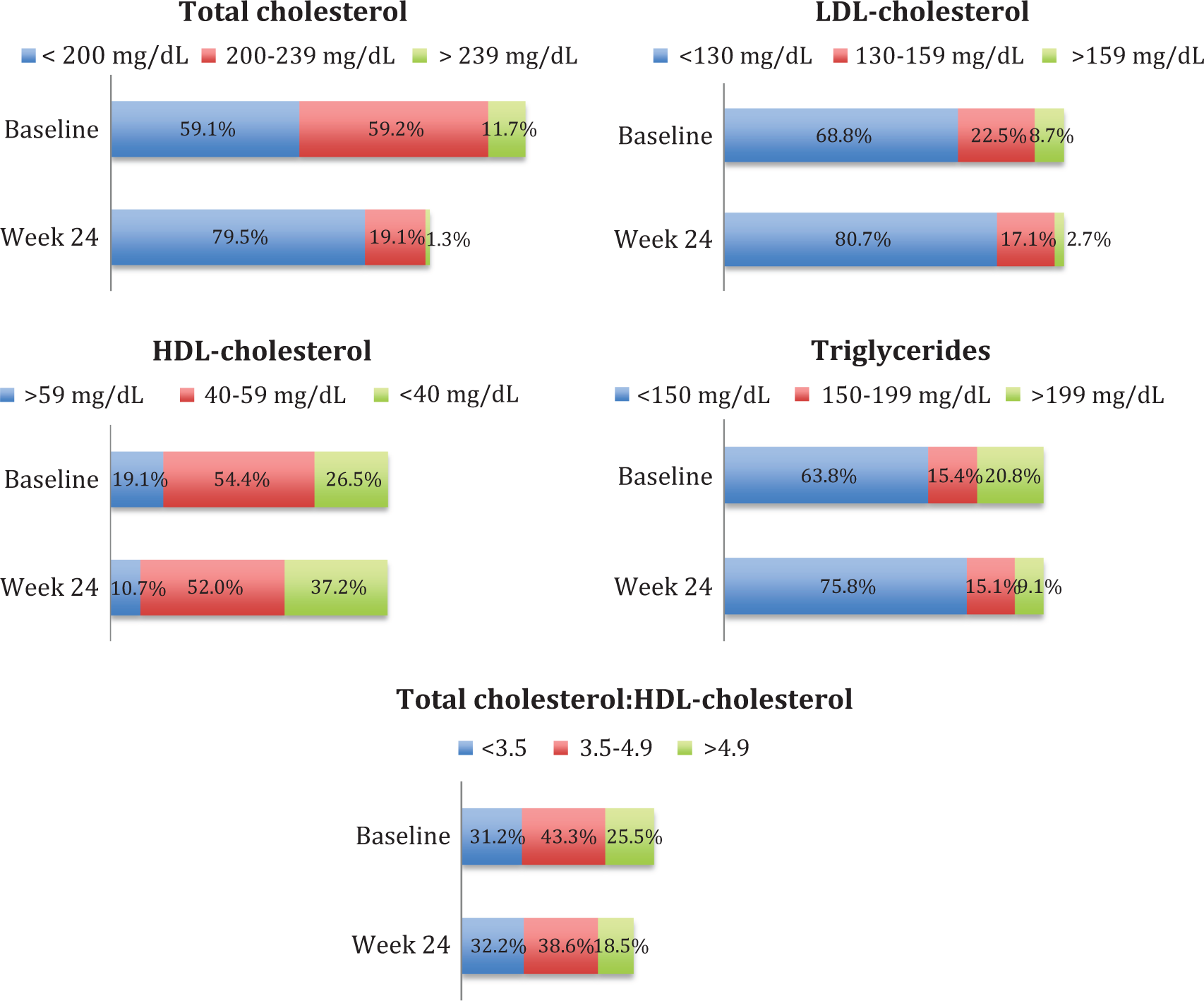
Categorical summary of fasting lipids at baseline and week 24 for the overall study population. P < .05 for the comparison between baseline and week 24 in all categories.
Discussion
This study shows that switching from an ART regimen based on NRTI plus NNRTI or PI/r to FTC/RPV/TDF in stable patients improves the lipid profile and CVR, while maintaining a good immunovirological control. Most participants switched to FTC/RPV/TDF from a regimen based on NNRTI, mainly the coformulated single-tablet FTC/EFV/TDF, which has been the gold standard for naive patients over recent years. 11 Given that CNS adverse effects are the main toxicity of EFV, 11 it is not surprising that this intolerance was the prime reason for switching. Receiving ART as a once-daily single-tablet regimen is associated with both clinical and economic benefits in terms of adherence, quality of life, and pharmacoeconomic evaluation. 5,12 In line with this, convenience was the second reason for switching to FTC/RPV/TDF in this cohort. Metabolic disorders, mainly hyperlipidemia, and an increased CVR have been widely associated with PI/r, and also with EFV, and have thus emerged as a set of important considerations when selecting antiretroviral drugs. 13,14 Rilpivirine has shown a better lipid profile compared with EFV in naive patients 6,7 and compared with PI/r in just one prior switch study. 8 We observed an improvement in lipid components and the CVR as early as 12 weeks after switching to FTC/RPV/TDF. HIV treatment guidelines that address CVR management recommend evaluating and managing serum lipids according to the established National Cholesterol Educational Panel goals; for patients on ART, in addition to lifestyle changes and lipid-lowering therapy, modifications in the antiretroviral regimen can be an important component of overall CVR reduction. 14 –16
Only 6 patients discontinued FTC/RPV/TDF, 3 due to adverse events, 2 due to virologic failure, and 1 due to withdrawal it. Although after discontinuation, EFV has an extended inductive effect on cytochrome P450 that, after switching, could reduce RPV exposures, this strategy was safe, as has been reported. 17
This study has certain limitations. First, the absence of a control group prevents us determining the evolution of the lipid profile and the effectiveness and safety in patients who would have continued with the prior regimen. The second limitation concerns the retrospective design of the study.
In summary, at 24 weeks after switching from a regimen based on NRTI plus NNRTI or PI/r to FTC/RPV/TDF, the lipid profile and the CVR improved, while maintaining good immunovirological control. Therefore, this strategy seems convenient for many patients.
Footnotes
Acknowledgment
The authors would like to thank the study participants as well as the investigators.
Authors’ Note
R Palacios, I. A. Pérez, C. M. González-Doménech, J. Santos (Hospital Virgen de la Victoria, Málaga), M. Mayorga, F. Castaño (Hospital Carlos Haya, Málaga), F. Lozano (Hospital de Valme, Sevilla), L. Apellido, A. Rivero (Hospital Reina Sofía, Córdoba), A. Del Arco, J. De la Torre, J. Olalla, J. L. Prada (Hospital Costa del Sol, Marbella), M. Yllescas, H. Esteban, and E. Aznar (Fundación SEIMC-GESIDA, Madrid) are the members of the GeSida 8114 Study Group.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. A. R, A. A, F. L, and J. S are on the scientific advisory board and speakers’ bureau for Gilead Sciences and have received grants and travel expenses for conferences from Gilead Sciences. R. P is on the speakers’ bureau for Gilead Sciences and has received grants and travel expenses for conferences from Gilead Sciences. M. M and I. P have no personal disclosures. This study was partially presented at the HIV Drug Therapy Congress, Glasgow, 2014.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by Gilead Sciences. This study has been supported in part by the RD12/0017/0017 project (Plan Nacional R+D+I) and cofinanced by Instituto de Salud Carlos III-Subdirección General de Evaluación y el Fondo Europeo de Desarrollo Regional.
