Abstract
Background:
An estimated 50% of people living with HIV (PLHIV) globally are unaware of their status. Among those who know their HIV status, many do not receive antiretroviral therapy (ART) in a timely manner, fail to remain engaged in care, or do not achieve sustained viral suppression. Barriers across the HIV care continuum prevent PLHIV from achieving the therapeutic and preventive effects of ART.
Methods:
A systematic literature search was conducted, and 6132 articles, including randomized controlled trials, observational studies with or without comparators, cross-sectional studies, and descriptive documents, met the inclusion criteria. Of these, 1047 articles were used to generate 36 recommendations to optimize the HIV care continuum for adults and adolescents.
Recommendations:
Recommendations are provided for interventions to optimize the HIV care environment; increase HIV testing and linkage to care, treatment coverage, retention in care, and viral suppression; and monitor the HIV care continuum.
Introduction
Modern antiretroviral therapy (ART) has dramatically changed the course of HIV disease. Today, a person diagnosed with HIV at the age of 20 years if started promptly on ART is expected to live a normal life span, with a highly preserved quality of life. 1 Antiretroviral therapy is also highly effective in preventing the sexual, 2 parenteral, 3 and vertical 4 transmission of HIV. These medical breakthroughs fostered the development of a treatment as prevention (TasP) strategy, 5,6 whose aim is to provide early access to HIV testing and treatment to prevent progression to AIDS and premature death as well as HIV transmission. 7
The United Nations (UN) has formally embraced TasP as it proposed a new global target for HIV treatment referred to as the 90-90-90 targets. 8 The new targets propose that by 2020, 90% of all people living with HIV (PLHIV) should know their HIV serostatus, 90% of people who know their HIV-positive status should be receiving sustained ART, and 90% of people on ART should achieve sustained viral suppression. 8 Modeling suggests that meeting the 90-90-90 targets by 2020 may decrease AIDS incidence, AIDS-related deaths, and new HIV infections by 90% from 2010 levels by 2030. 8
According to the Joint United Nations Programme on HIV/AIDS (UNAIDS), an estimated 15 million PLHIV were on ART by mid-2015—about 50% of the estimated number of diagnosed PLHIV—and less than 25% of those on ART were virologically suppressed. 9,10 The number of PLHIV who are diagnosed, on ART, and achieving viral suppression reflect 3 distinct points of patient engagement along the HIV care continuum, also referred to as the cascade of care, 11 with viral suppression as the ultimate clinical goal to improve individual PLHIV health outcomes and reduce HIV acquisition and transmission, thus conferring community and public health benefits.
Efforts are needed to optimize the HIV care continuum in order to attain the 90-90-90 targets. Key to these efforts will be to overcome and/or eliminate legal, social, environmental, and structural barriers that prevent PLHIV from accessing and utilizing HIV services. 12 The International Association of Providers of AIDS Care (IAPAC), through a multidisciplinary panel of international advisors, developed the first comprehensive, evidence-based guidelines for optimizing the HIV care continuum, with an aim to increase HIV testing coverage, linkage to care, treatment coverage, engagement and retention in care, and viral suppression for adults and adolescents (persons aged 10-19 years).
Guidelines Focus and Target Audience
These guidelines focus on clinical, behavioral, social, and structural interventions to optimize the HIV care continuum for adults and adolescents, including enhancing the care environment, addressing contextual barriers to scaling up services, and measuring and monitoring the HIV care continuum.
The target audience for these guidelines includes care providers, program managers, policymakers, affected communities, organizations, and health systems involved with implementing HIV programs and/or delivering HIV care.
Guidelines Methodology, Process, and Structure
A systematic search of the US Centers for Disease Control and Prevention (CDC) Research Project Database was conducted to identify randomized controlled trials (RCTs), observational studies with or without comparators, as well as cross-sectional and descriptive studies from January 2002 to July 2013. The CDC Research Project Database draws literature from bibliographic databases such as MEDLINE, CINAHL, PsycINFO, and EMBASE. Additional scientific input included hand reviews of RCTs, observational studies, and modeling papers focused on the HIV care continuum. A total of 6132 articles met the criteria (6020 within the review’s time frame augmented by an additional 121 more recent articles reporting on studies deemed to be of sufficient scientific merit and programmatic importance to be included in the evidence base). Data were extracted and coded by 6 independent reviewers using standardized data extraction forms.
Based on the advisory panel’s analyses of the available data, an evidence base comprising 1047 articles was used to support the development of evidence-based recommendations by advisory panel members. The evidence base is comprised of 131 RCTs, 171 observational studies, and 745 other data sources, including guidelines, systematic reviews, and descriptive articles. Recommendations were graded based on the quality of evidence, and this informed the strength given to each recommendation. Table 1 summarizes the grading scales used. Additional details regarding the methodology used to formulate the recommendations are available as Supplemental Appendix 1.
Grading Scales for Strength of Recommendation and Quality of the Body of Evidence.
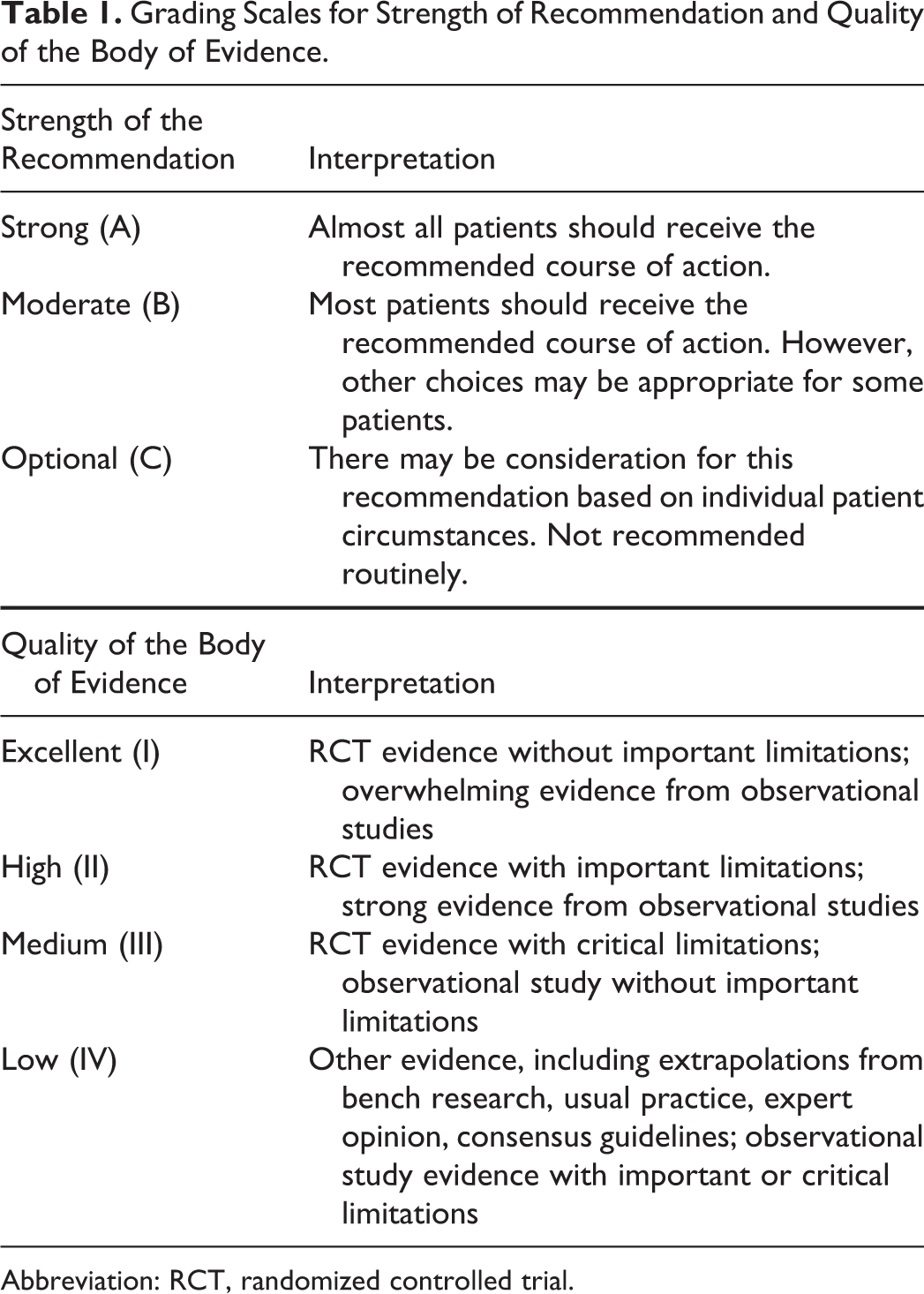
Abbreviation: RCT, randomized controlled trial.
The guidelines are structured in 6 sections, each containing graded recommendation statements. Table 2 lists the 36 recommendation statements. Supporting statements are provided for each recommendation. The supporting statements include selected articles from within the evidence base that provide context to and/or explain the basis for the recommendations.
Evidence-Based Recommendations for Optimizing the HIV Care Continuum.

Abbreviations: ARV, antiretroviral; ART, antiretroviral therapy; MSM, men who have sex with men; PLHIV, people living with HIV; PEP, postexposure prophylaxis; PrEP, preexposure prophylaxis; QD, once-daily; SMS, short message services.
In most instances, the evidence supporting the recommendations is generalizable across populations and settings. Nonetheless, the advisory panel notes that in relation to some recommendations, there is a challenge of context because implementation of a recommendation may be uniquely demonstrated in one setting but may not transfer well in another. In such instances, there is a need to measure both performance and outcomes as recommendations are implemented to ensure that there are not unique features related to the population or the setting that could affect the outcomes.
Table 3 provides context to and describes recommended standards for measurement of the HIV care continuum. While outside the scope of these guidelines, the advisory panel also developed guidance on a series of issues specific to key populations that are disproportionally affected by HIV/AIDS, which is available as Table 4. Finally, the advisory panel identified gaps in the available data regarding the HIV care continuum, which are the basis for future research recommendations listed in Table 5.
Recommended Standards for Measurement of the HIV Care Continuum.
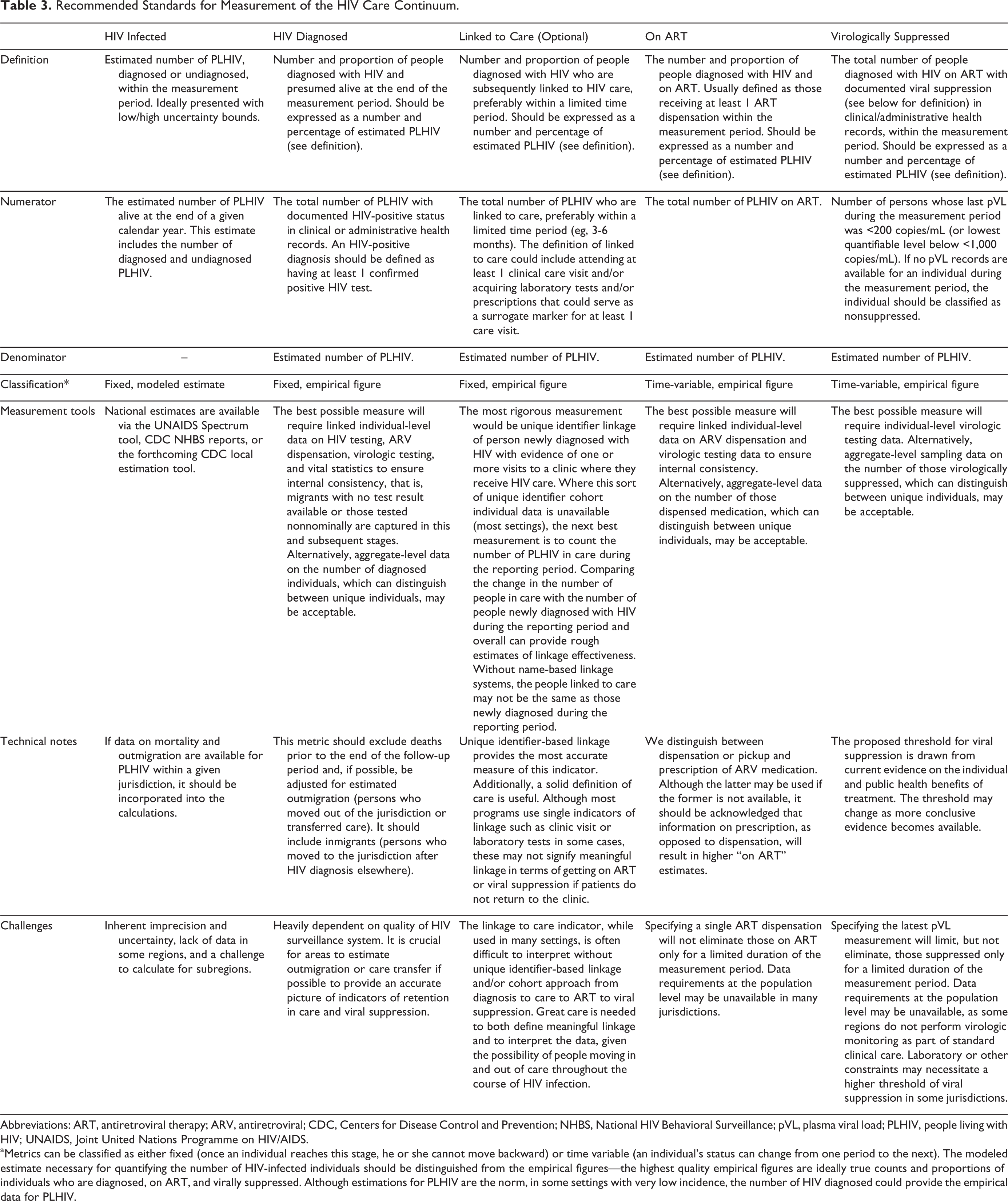
Abbreviations: ART, antiretroviral therapy; ARV, antiretroviral; CDC, Centers for Disease Control and Prevention; NHBS, National HIV Behavioral Surveillance; pVL, plasma viral load; PLHIV, people living with HIV; UNAIDS, Joint United Nations Programme on HIV/AIDS.
aMetrics can be classified as either fixed (once an individual reaches this stage, he or she cannot move backward) or time variable (an individual’s status can change from one period to the next). The modeled estimate necessary for quantifying the number of HIV-infected individuals should be distinguished from the empirical figures—the highest quality empirical figures are ideally true counts and proportions of individuals who are diagnosed, on ART, and virally suppressed. Although estimations for PLHIV are the norm, in some settings with very low incidence, the number of HIV diagnosed could provide the empirical data for PLHIV.
Guidance on Issues Related to Optimizing the HIV Care Continuum for Select Key Populations.

Abbreviations: ART, antiretroviral therapy; DAART, directly administered antiretroviral therapy; HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus; HPV, human papilloma virus; MSM, men who have sex with men; STI, sexually transmitted infection.
Recommendations for Future Research on Optimizing the HIV Care Continuum.

Abbreviations: ART, antiretroviral therapy; ARV, antiretroviral; LGBT, lesbian, gay, bisexual, and transgender; MSM, men who have sex with men; PEP, postexposure prophylaxis; PLHIV, people living with HIV; PMTCT, prevention of mother-to-child transmission; PrEP, preexposure prophylaxis; QD, once-daily; STI, sexually transmitted infection; TasP, treatment as prevention.
Guidelines
Optimizing the HIV Care Environment
In many settings, optimizing the HIV care environment may be the most important action to ensure that there are meaningful increases in the number of people who are tested for HIV, linked to care, started on ART if diagnosed to be HIV positive, and assisted to achieve and maintain long-term viral suppression.
13
–15
Overcoming the legal, social, environmental, and structural barriers that limit access to the full range of services across the HIV care continuum requires multistakeholder engagement, diversified and inclusive strategies, and innovative approaches. Addressing laws that criminalize the conduct of key populations and supporting interventions that reduce HIV-related stigma and discrimination are also critically important. People living with HIV also require support through peer counseling, education, and navigation mechanisms, and their self-management skills reinforced by strengthening HIV literacy across the continuum of care.
Many countries have enacted legal measures to make substance use and certain sexual behaviors among consenting adults illegal and have imposed mandatory HIV testing requirements and/or forced rehabilitation for men who have sex with men (MSM), transgender individuals, substance users, and sex workers. Sexual minorities and sex workers often face significant vulnerability to HIV as a result of high-risk behavior (eg, substance use), poor coverage by HIV and health care services, social marginalization, and continuing stigma and discrimination.
16
–19
Criminalizing the conduct of sexual minorities exacerbates the stigma and discrimination experienced by these individuals, which may result in increased HIV transmission and acquisition risks and contribute to decreased access to and utilization of HIV services.
20
Numerous countries have enacted laws that criminalize behaviors associated with HIV exposure, many of which pose a low or negligible HIV transmission risk.
21
–23
No differences in behavior have been noted between settings that enact such laws and those that do not.
24
–28
Many of these laws do not take into account measures that reduce HIV transmissibility, including condom use,
29,30
and were enacted before the preventive benefit of ART
31
or antiretroviral (ARV)-based preexposure prophylaxis (PrEP)
32
was fully characterized. Most PLHIV who know their status take steps to prevent transmitting HIV to others.
33,34
HIV-specific laws thus primarily exacerbate HIV-related stigma and decrease HIV service uptake.
21,35
–37
HIV-related travel restrictions fail to protect public health, run contrary to human rights principles, and may result in deleterious effects on HIV prevention and care. Nonetheless, many countries continue to impose such restrictions for PLHIV. These restrictions increase the stigmatization of migrants and PLHIV, discourage HIV testing, and can exacerbate marginalization by restricting the rights and opportunities of PLHIV.
38,39
Additionally, in humanitarian settings, although refugees are typically eligible for HIV services including ART through human rights laws and agreements, the rights of internally displaced populations to care are often more precarious.
40
Action is needed to repeal laws restricting travel for migrants living with HIV and to ensure comprehensive access to health care for all PLHIV.
Stigma can negatively shape quality of life, affect mental health, and influence ART use and outcomes. Studies have shown that individuals on ART face greater stigma and that health care settings and the personal environment were areas where the most stigma was experienced.
41
–43
A number of interventions have been shown to reduce HIV/AIDS stigma, including peer-led or -based strategies, community and popular leader interventions,
44
health care provider interventions, and community-level biomedical approaches.
45
The use of standardized stigma measures is recommended to ensure better comparability across health care and epidemic settings.
46,47
The People Living with HIV Stigma Index developed by and for PLHIV is recommended as an instrument that can be adapted and used in different settings.
48
Clinical mental health disorders such as anxiety, depression, and traumatic stress need to be proactively identified and treated to reduce their severity, as they place individuals at an increased risk of acquiring HIV and of HIV treatment failure. Mental health disorders such as these can result in a poorer quality of life and negatively affect access to and use of HIV services. Interventions in clinical settings can be effective in addressing mental health disorders among PLHIV and improving health outcomes.
50,51
Connecting PLHIV to mental health services at critical points along the HIV care continuum can help identify and treat mental health issues that may arise due to a diagnosis of HIV. It is recommended that measures of mental health be appropriately tailored and comparable across populations in broad categories (eg, different dimensions of emotional well-being).
52,53
Numerous observational studies show a significant adverse effect of comorbid psychiatric conditions on treatment outcomes among substance users, with strong evidence of an adverse effect of depression on ART adherence and treatment outcomes.
54,55
A recent meta-analysis found that treatment for depression increases the odds of ART adherence by 83%.
49
Enabling PLHIV to manage their care has several positive health outcomes including improvement in physical and psychological health; improved social relationships with families, communities, and providers; improved health literacy and seeking out HIV support and care services; and improved behavior change such as improved ART adherence.
56
–61
An RCT conducted in San Francisco assessing the efficacy of a self-management intervention among HIV-positive adults with high levels of self-reported ART distress showed positive effects for ART adherence and health-seeking behaviors among those randomized to the intervention. Those in the intervention group had a 6% decrease per month in the odds of nonadherence compared to unchanged adherence among the control group, and further, those who received the intervention were more likely to seek out information and social support to cope with side effects.
60
Education about self-management and user-driven techniques can be accomplished through primary care visits, through peer or lay health worker–led interventions, and by the use of mobile phones or the Internet to self-monitor and manage health.
57,62
–66
When provided with appropriate training and oversight and when guided by national policies, lay health workers can effectively distribute ART, provide pretest education and testing, and support related administrative tasks at the facility and community levels. Task shifting has been shown to be cost-effective
67
and allows for significantly increased patient enrollment
68
while maintaining quality of care
69,70
and retention in care
71
as well as improving ART adherence.
72
Use of community-based outreach workers or peers of similar risk status/cultural backgrounds has been shown to improve the uptake of HIV testing within social and risk-based networks when provided appropriate preservice and in-service training and competent professionally based supervision.
73,74
Additionally, community-based HIV rapid testing attracts those who are unlikely to undergo routine HIV testing at a medical facility, such as people from key populations affected by HIV or people who are working during clinic hours.
44,75
Multidisease prevention campaigns have been used to increase participation and access to HIV testing by providing multiple health interventions that have a broader appeal.
76
Satisfaction with the services among clients was high in 1 study, with 92% reported being “very satisfied.”
77
Counseling by community-based counselors increased uptake in pre-ART care,
78
decreased virologic failure,
79
and improved ART adherence and CD4 count response.
80,81
Nurse-led interventions were linked to improved ART adherence,
82
a significantly higher rate of retention in care,
83
and a decreased number of patients lost to follow-up.
81,84
Models of community-based support and ART delivery to complement facility-based ART programs can be effective strategies for enhancing psychosocial support and improving access to and outcomes across the HIV care continuum. 71,85 –90 The involvement of patients in chronic disease management in high-income countries has often resulted in improved health outcomes and increased utilization of health services. There is also evidence from low-income, high HIV burden countries that people living with chronic diseases can be trained to effectively act as tutors or peer navigators for other patients, particularly in settings with a severe shortage of human resources for health. 91 –94 The cost-effectiveness and potential relevance of this approach in countries with a high HIV burden have been documented. 84 –87
Increasing HIV Testing Coverage and Linkage to Care
Optimizing the offer of HIV testing (“seek and test”) is the critical first step in addressing the HIV care continuum. This should be done within the context of a high-quality confidential diagnosis in the location that best suits the individual’s needs. A critical focus should be placed on posttest counseling and immediate linkage to care for those found to be HIV positive as well as immediate access to ART. 95 The Strategic Timing of AntiRetroviral Treatment (START) study recently showed a 53% reduction in serious morbidity and mortality from HIV due to early ART at CD4 counts of over 500 cells/mm3 compared to deferred initiation of ART at CD4 counts of 350 cells/mm3. 96 Health care systems should strive to make HIV screening widely available and accessible to all individuals regardless of gender, age, or perceived risk factors. 97 In addition, targeted HIV testing approaches are recommended both in concentrated and in generalized epidemics.
Linkage to medical care is a critical but often neglected step in the HIV care continuum. 98 It can be defined as the process that facilitates a newly diagnosed person’s optimal engagement in HIV care. Typically, linkage may consist of verbal or written referral to an HIV care facility by the counselor or the individual who provided the HIV test result. 99 In settings where HIV testing and care are provided at the same or a nearby facility, recently diagnosed persons may be escorted from the test site to the care site to ensure that this initial linkage is successful. Other programs use “navigators” to ensure linkage to services that may not be available at the same site and/or provided on the same day. 100,101
Linkage to care should enable a patient living with HIV to engage in care early, benefit from a broad package of care, and facilitate immediate access to ART. Prompt engagement in HIV care also optimizes individual and public health outcomes.
31
Linkage to care is attenuated by economic, geographic, transportation, and distance barriers, as well as stigma and discrimination,
102,103
all of which should be addressed in efforts to optimize the HIV care environment.
Routine HIV screening identifies significantly more undiagnosed HIV infections than a risk-based approach, and routine screening is generally cost-effective in populations that meet minimum thresholds of HIV prevalence of >0.1%.
97
Opt-out HIV screening and testing means that the patient is notified orally or in writing that an HIV test will be performed unless the patient declines. Opt-out testing is now routinely recommended in both national and international guidelines, with the provision of pretest information about the meanings of positive and negative test results made available within the cultural context in the languages of the populations encountered within the area in which testing is being offered.
97,104
In a retrospective cohort study, 2408 (73%) of 3303 HIV-infected women diagnosed during the study period had missed-opportunity medical visits, suggesting that a routine offer could have diagnosed them earlier.
105
Another retrospective cohort study indicated that the 1769 individuals diagnosed “late” with HIV (ie, who developed AIDS within 1 year of their diagnosis) made a total of 7988 visits to a health facility during 1997 to 2005 before their first reported positive HIV test.
106
Further, the diagnoses reported that nearly 8 in 10 of these visits were not likely to prompt HIV testing under a risk-based testing strategy. Opt-out HIV testing has been demonstrated to increase HIV testing coverage and aid in earlier detection of the virus.
107,108
In addition to general medical/primary care outpatient settings, this recommendation includes outpatient venues where individuals seek care for conditions associated with HIV, such as tuberculosis (TB), viral hepatitis, and sexually transmitted infection (STI).
One RCT,
109
a systematic review and meta-analysis,
110
and numerous observational studies examined various community-based testing approaches.
111
–117
In addition to routine HIV screening in facility-based settings, community-based HIV testing approaches have the potential to normalize testing within a community and among couples to significantly expand HIV testing and to reach those who do not access the health system. Project Accept (HPTN 043) was especially effective in reaching men
109
and also demonstrated a 4-fold increase in identifying previously undiagnosed HIV cases in comparison to the control group. Community-based HIV testing interventions
118
identified a larger proportion of people who had tested for the first time and who had CD4 counts above 350 cells/mm3 than standard facility-based HIV testing, though the positivity rate was lower. Overall acceptability of the approaches exceeded 80%.
118
Using varied modalities to reach people
119,120
has the potential to reach those who have never been tested before. Multidisease prevention community health campaigns that combine other public health interventions
121
with HIV testing and counseling, such as maternal–child health programs, can be effective
122
and cost-efficient.
123,124
Community-based HIV testing of individuals and couples should be coupled with follow-up by an HIV-positive patient navigator. Such an intervention can significantly contribute to linking and retaining HIV-positive persons to care in a timely manner.
125
HIV prevalence is often higher than anticipated or reported among employee populations in large institutional settings, particularly in communities where the undiagnosed HIV seroprevalence is >0.1%.
126
–128
Screening programs for individuals and for couples in such settings have the potential to identify undiagnosed individuals earlier than they might otherwise present (ie, if they waited until they were symptomatic) and get them early into care, so that they can optimize their own health by starting ART and protect their partners. However, stigma and confidentiality concerns remain significant barriers to acceptance of HIV testing in some workplace settings, especially in communities where stigma due to HIV is acute (eg, mining/trucking companies).
129
–131
In such communities, voluntary workplace testing should be conducted after strict implementation of confidentiality measures and assurances of fair treatment of those who test HIV positive. Despite high acceptance of the HIV test offer at employment sites, some studies have suggested that follow-up rates can be low and that HIV positivity can be a risk factor for nonattendance at follow-up.
129
These results point to the likely benefit of rapid test results in employee testing programs, as well as supportive and rapid linkage to care protocols. Although the near-term individual and public health benefits of knowing one’s HIV status may be clear, it is important for patients and providers to consider the context and prevent the negative aspects of stigma and criminalization. Thus, testing interventions in workplaces and institutional settings are advisable only in high-prevalence situations and where the dangerous consequences of stigma, employment impact, and criminalization can be managed.
HIV self-testing can happen in private with indirect support such as provision of referral information or can be supervised with moderate pretest and/or posttest support. The modality can include nonrestricted access to HIV self-testing through various sites and locations (eg, over the counter, vending machines, pharmacies, and the Internet) or through community or health care facility distribution. Three cross-sectional feasibility studies
132
–134
examined the acceptability and uptake of HIV self-testing in different settings for different populations. All the studies reviewed demonstrated that the testing modality is well accepted in both high-prevalence settings and concentrated epidemics, and across different populations,
133
–135
with high accuracy after moderate direction on how to use the self-test which should include instruction on confirming preliminary positive results and linkage to care if confirmed positive. HIV self-testing was also cost-effective in resource-limited settings.
136
Ensuring high-quality products for self-testing through regulatory processes and policies concerning medical devices and regulating the legal use of the devices are essential for scale-up of quality HIV self-testing.
Use of epidemiological data to guide HIV testing has been a key component of HIV testing practices since the beginning of the epidemic. Use of network analysis has been instrumental in understanding the transmission dynamics of HIV among MSM.
137
Offering HIV testing to members of networks identified to be at risk for further HIV transmission is recommended. A systematic review of studies of partner notification and referral for HIV testing found that this strategy identified an HIV seropositivity rate of 1% to 8% among partners of an index case.
138
Use of network analysis has led to enhanced HIV testing among vulnerable populations, including black MSM.
139,140
Network-based peer recruitment for HIV testing led to improved HIV testing rates within networks (sexual and in injection drug users) in the United States, with a subsequent HIV seroprevalence rate of 5% among those tested.
141
Public health interventions for the follow-up of persons newly diagnosed with HIV, such as partner services and short-term case management, are strongly recommended. Partners and other contacts who may be infected with HIV should be notified and offered testing. All newly diagnosed individuals should be helped to link to HIV primary care. Voluntary partner notification should be considered, being mindful of stigma concerns and criminalization-related issues in jurisdictions where this is relevant. A systematic review
138
found that 67% of the partners identified were notified, with 63% undergoing HIV testing with a mean HIV diagnosis rate of 20%. Another study found that provider notification was higher than self-notification (51% versus 24% for self-notification).
142
Partner- or couple-based HIV testing and counseling can be more supportive and can facilitate safe disclosure, and it is therefore recommended that health care providers consider initiating such discussions with women and with men and their partners. Knowing that a partner has taken an HIV test and knowing a partner’s HIV status are associated with HIV testing among pregnant women.
143
HIV-positive pregnant women whose partners have an unknown HIV serostatus are more likely to be lost to follow-up.
144
A study involving 1928 patients in New York City showed that initiation of HIV care was more likely to occur when HIV testing was conducted at a clinical site with access to HIV care.
145
In Uganda, outpatients referred to HIV testing (74%) were more likely to attend an HIV clinic within 6 months than those receiving HIV testing and counseling on-site as inpatients.
146
In another study conducted in a hospital emergency department, on-site linkage teams were able to connect more than 90% of newly diagnosed and out-of-care HIV-infected patients to HIV care services.
147
However, in other hospital settings, suboptimal ART eligibility assessments and referral systems created unnecessary delays and attrition in the pre-ART clinical management period.
148
The use of ART and PrEP reduces the transmission risk to 0.5% among adherent HIV-uninfected individuals who are serodiscordant couples.
149
HIV serodiscordance in couples is common, and approximately 45% of HIV-infected individuals have an HIV-negative partner.
150
The option of early ART in combination with PrEP while the index member of the couple has a detectable viral load should be considered among HIV-serodiscordant monogamous couples, as a means to virtually eliminating the likelihood of HIV transmission. In an open-label trial of daily PrEP in high-risk MSM, an 86% reduction in the risk of HIV infection was observed in the PrEP treatment group compared to no PrEP, and the number needed to treat to prevent a single infection was 13.
32
It bears noting that PrEP is one of many prevention options for people who are not infected with HIV. Condoms, like most other HIV prevention interventions, are effective when used correctly and consistently and have been shown to reduce heterosexual HIV transmission by 80%.
151
Within the context of PrEP, condoms protect against STIs not prevented by PrEP, thus regular monitoring and counseling regarding condom use are advised given recent data from PrEP use in regular clinical practice in San Francisco indicating a 40% drop-off rate in condom use among PrEP users.
152
Voluntary male medical circumcision has been proven to be effective in 3 RCTs which demonstrated around a 60% reduction in transmission risk for circumcised heterosexual men.
153
–155
Postexposure prophylaxis, by blocking HIV infection, is recommended for people who had occupational exposure such as needle stick (after careful evaluation of the circumstances) and after sexual assault.
156
For persons who inject drugs, opioid substitution treatment and needle and syringe programs are recommended as part of an evidence-based harm reduction package.
157
–160
Case management is a collaborative process of assessment, planning, facilitation, care coordination, evaluation, and advocacy for options and services to meet a patient’s health needs and achieve health, quality, cost-effectiveness, and outcomes. Case managers have been successfully used to strengthen patient outcomes throughout the HIV care continuum including early linkage to care, retention in care, and sustained ART adherence. 161 –163 Additionally, case managers have been demonstrated to be useful in reaching hard-to-reach and key populations. 162 Patient navigators are individuals who assist individual patients to navigate through the continuum of care as it pertains to the patient’s specific disease, ensuring that barriers to that care are resolved and that each stage of care is as seamless as possible. The benefit provided by patient navigators in relation to linkage to care is also well documented. 98,164,165 Newly diagnosed HIV-positive persons have been more successfully linked to care when supported or encouraged by a patient navigator. 166 When the patient navigator is also a person living with HIV, the potential magnitude of benefit may be even higher. 103
Increasing HIV Treatment Coverage
Increasing early access to ART is associated with decreased AIDS-related morbidity and mortality, as well as reduced risk of HIV transmission. 167 Final results of the HPTN 052 clinical trial showed a 72% risk reduction in HIV transmission among 1763 serodiscordant couples with immediate initiation of ART. 168 Importantly, no genetically linked HIV transmissions occurred when the index member of the couple had an undetectable viral load. Preliminary results from the PARTNERS study of 1100 serodiscordant couples, 40% of them were MSM, found that there had been no HIV transmission within couples who were not using condoms from a partner with an undetectable viral load, albeit with large confidence intervals around this estimate. 169
Recently, final results of 2 major RCTs provided strong evidence of the benefit of early ART initiation. The TEMPRANO study showed that initiating ART at the 500 cells/mm3 threshold in lower-income settings with a high prevalence of TB and bacterial infections reduced the risk of serious illness, including TB and death, by 44% when compared to starting ART according to 2013 WHO guidance.
170
Similarly, as noted previously, the START study showed that among 4685 patients in 35 nations, the risk of progression to AIDS, other serious illnesses, or death was reduced by 53% among people who initiated treatment immediately.
96
The START study results are especially important because the magnitude of the health benefit conferred by ART was similar in high- and low-income countries.
The benefits of ART, such as prevention of illness, death, and transmission, extend to persons irrespective of CD4 count, including those with recent or long-standing HIV infection and/or those with normal to severely depleted CD4 counts.
95,110
Antiretroviral therapy initiation should not be delayed in individuals with acute opportunistic infections, including TB or Pneumocystis pneumonia, with the notable exception of cryptococcal meningitis and other opportunistic infections associated with increased intracranial pressure.
171
HIV is the strongest known risk factor for developing TB, and ART is the strongest TB prevention intervention for PLHIV.
110
In one large randomized trial, initiation of ART during TB therapy resulted in a 56% reduction in death, and this result was observed in all CD4 count strata.
172
Antiretroviral therapy should be provided as soon as possible after a diagnosis of HIV infection and no later than 8 weeks after initiating TB treatment. For those individuals with severe immunosuppression and TB, ART should be offered within 2 weeks of initiating TB treatment. The use of ART to prevent HIV transmission to sexual partners as well as from mother to child (through Option B+) may be an added incentive for starting immediate life-long ART.
173
In resource-limited settings, programmatic prioritization of vulnerable groups of people should be considered to ensure that they get access to treatment. CD4 count monitoring is optional for individuals who are on stable ART and who have been virologically suppressed for more than 2 years.
174,175
If available, assessment of CD4 count is recommended to direct prevention of opportunistic infections. However, the initiation of ART for prophylaxis against opportunistic infections should not be denied or deferred if CD4 count testing is not available.
Antiretroviral therapy is proven to prevent HIV-related morbidity, mortality, and transmission.
7
Immediate initiation of ART maximizes the opportunity to achieve these objectives. The thoughtful selection of ART and the maintenance of patients on first-line treatment are therefore central to care for PLHIV. A number of highly effective, well-tolerated, QD-dosed, first-line ARV regimen options are available, including nonnucleoside reverse transcriptase (NNRTI)-, ritonavir (RTV)-boosted protease inhibitor (PI)-, and integrase inhibitor-based regimens. For example, recent RCTs using integrase inhibitor-based ARV regimens for first-line ART have demonstrated statistical superiority or comparability to other commonly recommended regimens.
176
–179
Study superiority was driven by improved tolerability and lower adverse event profiles of these integrase inhibitor-based ARV regimens. Whenever possible, switching ARV regimens following evidence-based strategies to address adverse effects, toxicity, or advances in medications (eg, increased efficacy) should be considered.
180,181
Fixed-dose combinations are preferred to aid with ART adherence and decrease risks during medication stock-outs.
182
Patients taking QD-dosed regimens have higher rates of adherence than those taking twice-daily dosed regimens.
182
Although challenging within the context of HIV care delivered via a public health approach in resource-limited settings, care should nevertheless be taken to individualize treatments based on transmitted drug resistance, comorbid illness, and/or lifestyle considerations.
Suppression of viral replication is the key therapeutic goal as it is the driver of individual and public health outcomes. Measuring plasma HIV-1 RNA levels through a viral load test is the preferred laboratory tool for HIV disease prognosis and to guide HIV treatment strategies. Viral load testing should be used after ART initiation as a means to monitor ART response.
183
However, lack of availability of viral load testing should not delay the initiation of ART. A patient’s HIV-1 RNA level should reach below the limit of quantitation within 6 months of ART initiation. Among individuals who are on stable ART with a CD4 count >350 cells/mm3 and who have been virologically suppressed for 2 years, viral load monitoring can be performed every 6 to 12 months.
183,184
CD4 counts can be used to help determine eligibility for opportunistic infection prophylaxis but are not effective for assessing ART effectiveness since CD4 count and viral load levels can be discordant.
Transmitted or treatment-emergent HIV drug resistance may limit the response to ART. Transmitted drug resistance is well documented in both resource-rich and resource-limited settings.
185,186
Transmitted drug-resistant HIV is associated with reduced rates of viral suppression to first-line treatments
187
and occurs in approximately 5% to 15% of treatment-naive individuals.
188,189
For this reason, where available, HIV drug resistance testing is recommended at entry into care or prior to ART initiation to help guide ARV regimen selection. Treatment-emergent resistance is a major determinant of virologic failure.
190,191
The use of resistance testing in this setting has been shown to improve clinical response to subsequent lines of ART.
192
Resistance testing for an individual is recommended in contexts where there is availability of second- and third-line medicines.
193
In settings where most people do not have access to ART and/or there is limited access to viral load testing, it is advisable to expand access to these interventions while preparing for the medium- and long-term goal of improving access to resistance testing and alternative ART regimens for individual patients before and while they are on ART.
172
In resource-limited settings where individual pre-ART resistance testing is not routinely available, regular population-based surveillance of transmitted resistance in the country is recommended in order to provide information for policy and programmatic considerations. Population-based resistance surveillance can provide important information for decision making about the selection of initial and subsequent ART regimens, the population-level efficacy of ART regimens, as well as PrEP and PEP.
173,194
Community-located ART distribution is a cost-effective service delivery model whose rates of attrition and mortality are similar to those at the facility level. 195,196 A retrospective cohort study in the Bronx, New York, found that HIV services incorporated into community-located primary care setting improved the distribution and delivery of HIV treatment, demonstrated comparable treatment outcomes when compared with a hospital-based specialty center, and achieved slightly lower prevalence of AIDS (46% versus 54%, respectively) in the community-located program. 196 Studies have demonstrated the efficacy of community-based approaches to expanding other HIV services, including testing. 197 –199 Community-based ART distribution may also be more effective in helping to retain patients. In Mozambique, Médecins Sans Frontières (MSF) developed a community-located ART adherence and distribution program, now adopted as national policy, which resulted in 91.8% adult retention and 94% youth retention rates at 4 years and facilitated the increased involvement of affected communities in HIV services. 200 Patients receiving specialized ART counseling at community-based pharmacies in multiple US states 201 had significantly higher ART adherence rates than those using traditional pharmacies. 202 A cohort study in California demonstrated that pharmacist-provided therapy management services were associated with higher ART adherence rates. 203 A pharmacist-managed adherence clinic in Colorado was shown to significantly increase ART adherence rates. 202 In Uganda, ART distribution at community-based sites was found to be associated with significantly higher rates of retention in care and lower mortality rates. 88,204
Increasing Retention in Care, ART Adherence, and Viral Suppression
A number of barriers to HIV treatment engagement are common across countries, even when the availability of local resources may differ widely. Yet, the need to efficiently keep people engaged in their care is more critical than ever, as resources are ultimately limited in every setting, and growing numbers of PLHIV require lifelong care and treatment to attain the individual, public, and community health benefits of early ART.
Significant attrition from HIV care programs has been documented in many settings. Monitoring and optimizing retention in care should be a priority of programs if control of the HIV epidemic and optimal individual health outcomes are to be achieved. Moreover, as life expectancy for PLHIV increases, the impact of successful engagement and retention in care is magnified.
Ultimately, adherence to virologically active ART has been shown to be a major predictor of suppression of HIV replication,
205
–207
emergence of drug resistance,
208
–210
disease progression,
211
and AIDS-related death.
212
–214
The dramatic dose–response pattern between adherence and viral suppression, as well as the reasonable rates of suppression achieved at moderate levels of adherence, supports the recommendation to treat all eligible PLHIV and encourage maximum adherence with each patient.
11,215,216
Tracking the retention of clients in HIV care is important for the purposes of both clinical and health services and could be considered a part of quality-of-care indicators at both clinic- and surveillance-system levels.
217
Retention measures used and the desired frequency of visits may vary according to jurisdictions and programs, but in order to facilitate effective monitoring, efforts should be made to harmonize measures with existing national and international recommendations. Gaps in care, visit adherence, and intervals of time between visits, in addition to the use of data including medical records and surveillance systems, are well-tested measures that can be integrated into local standards of HIV care.
218
–221
For example, an integrated public health surveillance approach and an electronic health record system in Louisiana were successfully used to provide alerts to medical providers when their clients had been out of HIV care for longer than 12 months.
222
Identifying optimal thresholds of being “out of care” or lost to follow-up remains important, and a 12-week delay in visit, based on pharmacy registry data in South Africa, was considered optimal.
223
Pharmacy tracking data can also include failure to refill medication prescriptions
224,225
and interruptions in laboratory monitoring
226
as warning signs of loss to follow-up (LTFU).
Adherence to ART is a reliable predictor of adherence to clinic visits and can be tracked using clinical, immunological, as well as pharmacy and clinic visit measures. Viral load suppression is the most reliable indicator of ART adherence. Viral load surveillance data can be successfully used to monitor engagement and retention in care and improve linkage and retention efforts in local communities.
217
Patient self-report is the most widely used method for evaluating medication adherence,
227
particularly because physician estimates of their patients’ ART adherence is not an effective measure, and evidence shows a poor correlation between physician estimates and patient self-report.
228
–230
However, a literature review indicated that pharmacy refill data may be a better measure than self-reported adherence.
231
The availability and reach of mobile communication have increased worldwide, including in sub-Saharan Africa, where two-thirds of PLHIV reside. A systematic review found that technology-based approaches supporting self-care among PLHIV may improve adherence to ART, especially when these approaches utilized multiple components and facilitated communication with health care providers.
232
Once-a-week text message or SMS reminders may be the optimal periodicity.
233
There is evidence
233
–236
that weekly 2-way SMS improves adherence and virological outcomes in HIV-infected adults in Kenya. Evidence from 2 RCTs
237,238
shows that there is no significant impact of using an alarm on poor adherence or viral failure, except in PLHIV with memory impairment. In fact, appointment reminders alone were found to be useful in a small RCT
239
but only among nonhomeless, nondepressed clients in a much larger RCT.
240
Evidence from several RCTs
237,241
–243
found that PLHIV who had received targeted adherence counseling were significantly more likely to have optimal adherence and virologic suppression.
227,244,245
Educating patients on the importance of keeping clinic appointments has demonstrated positive effects on retention in care and ART adherence.
246,247
A study assessing the efficacy of an intervention using targeted messages enforcing the importance of regular clinic visits using posters, messages, and brochures in clinics demonstrated a 7% improvement in patients keeping 2 consecutive clinic visits in the intervention year compared to the preintervention year.
247
The use of pillbox organizers has also been associated with improved ART adherence and virologic suppression in patients from difficult-to-treat populations. A study using marginal structural models to estimate the pillbox organizer effect on adherence, viral suppression, and disease progression among a homeless HIV-positive population in the United States found that pillbox organizers were associated with improved adherence, reduction in viral load, and decreased risk of progression to clinical AIDS.
248
Evidence from a meta-analysis of RCTs (N = 12) indicated that directly administered ART did not offer any benefit over self-administered ART.
249
Ten RCTs reported on the primary outcome of virologic suppression for directly observed versus self-administered ART,
249
each reinforcing that this approach is not recommended for supporting ART adherence in a general HIV-infected patient population. Community outreach-based directly administered ART through methadone maintenance treatment programs has been found to be effective in improving ART adherence and viral suppression among both PWID and released prisoners.
250
–253
Its effects decay postintervention, thereby reducing its efficacy in the long term.
254
–256
Efficacy is improved markedly when patients are linked to other supportive services.
257
HIV care continuum data from various jurisdictions show that a sizable percentage of those with HIV infection are not engaged in care following an HIV diagnosis, miss clinic appointments, or are lost to follow-up after establishing HIV outpatient care. 15,258 –260 In the Antiretroviral Treatment and Access Study (ARTAS) comparing strength-based case management sessions with passive referral to care for patients with recently diagnosed HIV infection, a significantly larger proportion of the case-managed participants visited an HIV clinician at least once within 6 months (78% versus 60%) and at least twice within 12 months (64% versus 49%) using trained linkage case managers to help clients identify their internal strengths and assets to facilitate successful linkage to HIV care. 98 The provision of transportation to the clinic and mental health and drug counseling services to PLHIV who were in need of those services was associated with a 20% improvement in clinic retention. 261,262
The Retention in Care (RIC) Study conducted with patients at high risk for inadequate retention demonstrated that enhanced personal contact between patients and a trained provider improved retention in care compared to standard contact at the participating clinics. 263,264 This study demonstrated that a low-cost intervention to incorporate retention in care into the clinic culture proved effective in improving visit adherence, particularly among new patients and those with detectable viral loads. 247,264 In addition, a loss to follow-up intervention found that the use of case workers to locate and assist patients to reengage in care was successful in reengaging more than half of those lost to follow-up over an almost 3-year period. 265
Adolescents
Adolescents are often overlooked in HIV testing and are reluctant to seek out HIV services. Increased awareness among care providers and policymakers is critical. Provider-initiated testing and the provision of adolescent-friendly testing services outside health facilities are recommended, as is the potential for self-testing.
266
A number of countries require parental consent for HIV testing, which can be a significant barrier to testing.
266
However, nations should consider lowering the age of consent to 12 to 14 years or recognize the evolving capacities of the child in order to give adolescents clear access to HIV counseling and testing. National policies should also acknowledge the holistic nature of HIV counseling and testing and ensure that consent laws do not restrict access to condoms, treatment autonomy, and HIV prevention interventions such as PrEP and medical male circumcision.
Youth have repeatedly articulated that engagement with health services should be local, integrated, quick, confidential, nonprejudicial, “hassle-free,” and free (or inexpensive).
267
Utilizing venues and activities where adolescents routinely gather offers opportunities for youth-friendly interventions, including HIV testing. By leveraging the commonality of adolescence and group norms, comprehensive services can be offered with subsequent reduction in stigma, a sense of shared experiences, peer support, and health system efficiencies. Health care delivery, especially related to sexual and reproductive health, through school-based services, should be maximized, provided these can be youth-friendly.
268
Clinics should strengthen the existing system for youth and target youth who are already being screened for or diagnosed with STIs or provided reproductive health care. Service delivery should include the following components: a youth-friendly atmosphere, flexible hours, separate waiting areas for adolescents, alternative service delivery settings, and the assurance of confidentiality, including the option for self-testing.
266
Results from an observational study with simulated patients demonstrated that adolescent-friendly clinics improve the accessibility of HIV testing by ensuring that youth are not turned away without a test.
269
Disclosure of HIV diagnosis can open the dialogue with youth about HIV and dispel myths about HIV infection, which often leads to better adherence to ART programs. Disclosure is also important for retention in care as young adolescents get to understand why they have to meet regularly with health care professionals.
270
–272
Disclosure to adolescents should take into account their stage of development and other factors. If disclosing to minor adolescents, such disclosure should include parental notification. Support for older adolescents, whose results should be given confidentially, to facilitate their diagnosis disclosure to parents, family members, peers, and sexual partners is also essential for coping and the ability to engage in taking medications and in prevention activities.
270,273,274
Transition programs are needed to effectively transition HIV-positive adolescents from pediatric HIV care to more independent adult HIV care. A well-managed transition can lead to improved adherence to both care and treatment. 275 Adolescents on ART may present with neurocognitive and mental health challenges, may be socially stigmatized, and may face difficulty in having successful sexual relationships. 276 These issues often evolve into major obstacles when transitioning from adolescent to adult HIV care, posing a significant obstacle to achieving and maintaining viral suppression.
Metrics for and Monitoring of the HIV Care Continuum
Measuring the HIV care continuum from diagnosis to viral suppression has become critical to evaluating the success of HIV responses. 8,11,277 However, monitoring the continuum is challenged by a lack of clarity regarding methodology and incomplete and/or fragmented data infrastructure in many settings. There have been very few estimates covering the full spectrum of stages of the continuum, produced for a complete jurisdiction, and covering an extended time frame. 278 –280 There is a lack of consensus regarding the metrics to be included in estimating the HIV care continuum, and measurement methodologies and standards of published continua are often not well described. 280 –283 This lack of consensus compromises the ability to interpret and compare continuum results within jurisdictions over time and across geographic settings.
In order to better align with the proposed UN 90-90-90 targets, a simplified, but comprehensive, 4-stage continuum of HIV care is proposed to capture (1) the proportion of PLHIV who have received their diagnosis (the first 90), (2) the proportion of PLHIV who are linked to care, (3) the proportion of PLHIV who are on ART, and (4) the proportion of PLHIV who are virally suppressed. The estimated number of PLHIV is the common denominator for all indicators. It is important to consider that the 90-90-90 targets use a floating denominator and that achieving 90-90-90 translates into a 90% tested-81% on ART-73% virally suppressed HIV care continuum.
As described in Table 3, the HIV care continuum should be (1) representative of the complete geographic area in question, (2) internally consistent, and (3) longitudinally monitored. Representativeness requires that the complete population of PLHIV in the geographic setting be accounted for. Internal consistency requires that the numerator from each continuum stage is also represented in its denominator. For example, all those classified as “diagnosed” are captured within the estimated prevalent population (“estimated HIV infected”), those classified as “on ART” are also classified as diagnosed, and those classified as “suppressed” are also classified as on ART. Further, the number of people estimated to have HIV infection is the overall denominator, and numerators of the subsequent stages use this single denominator to derive proportions.
The goal is to use metrics that are practical, feasible, and as simple as possible to enable accurate monitoring of the HIV care continuum. Programs may need to develop multiple monitoring approaches beyond the focused recommendations mentioned subsequently to assure a comprehensive quality response. Nevertheless, comprehensive and transparent reporting of the measurement methodology for each step of the continuum is imperative for internal decision making and external comparison. Incomplete reporting may result in suboptimal program assessment and suboptimal resource allocation decisions. If the recommended continuum is not followed, then a clear explanation of the methodology that is used should be included to alert end users and to avoid inappropriate comparison of results with continua that use the recommended approach.
For the sake of comparability, the use of a common method to establish the denominator for the HIV care continuum is critical to unbiased evaluation of the progress and impact of program implementation. There are a number of estimation methods. For example, the Spectrum tool
284
is available and can be applied to jurisdictions with a minimal set of data elements. National and local governments may opt to use other estimation methodologies to estimate the overall number of PLHIV. It is important to standardize, validate, and publish the methodology for review. In some settings with comprehensive HIV testing, the number of people diagnosed with HIV may serve as an accurate surrogate for the number of PLHIV. The estimations for key population denominators require different methodologies, and countries are encouraged to monitor the continuum among these vulnerable and often neglected communities. Continua should be measured annually, and all proposed metrics should be expressed as period rather than point prevalence figures.
The estimated number of PLHIV in a given geographic setting during a given calendar year is taken as the denominator. No exclusions are made pertaining to the status or risk group of the individual. It is expressed as both a count and a proportion of the total population within the geographic setting. This indicator serves as the denominator for all the remaining steps in the HIV care continuum and is crucial for the population-based framework of the continuum. Although it is the most challenging step in the continuum to estimate, it is key for monitoring the success of HIV case-finding efforts. To prioritize and monitor efforts to increase HIV diagnosis, an estimate of the undiagnosed fraction is required. This measure is the cornerstone of the entire continuum framework and is required to assess the relative priority or success of efforts to improve case finding and access to HIV care and treatment. In some settings, the estimated number of PLHIV is nearly the same as new diagnoses—in these settings, if the epidemiologic experts agree, the new diagnoses can be used to estimate the number of PLHIV.
The estimated number of PLHIV (diagnosed and undiagnosed) serves as the first data element and the overall denominator for the HIV care continuum. Not standardizing the denominator can lead to bias toward using smaller denominators to inflate reported success with implementing the continuum and also prohibits the comparison of program success both within the program and with other programs.
The numerator for the first step in the continuum should be the total number of people diagnosed with HIV. The indicator should be expressed both as a count and as the proportion of the estimated number of PLHIV within the geographic setting.
The second step in the continuum is the number and proportion of PLHIV who are linked to care. Although linkage to care is a critical step toward starting ART and ultimately achieving viral suppression, this indicator is considered optional. Linkage to care is useful to monitor to ensure that people who are diagnosed with HIV have access to ART as soon as feasible for their health and to prevent HIV transmission. In some settings, linkage to care is equivalent to starting ART, whereas in others, there may be considerable delay while waiting to access ART and achieve viral suppression. In those settings, linkage proportions may be high without achieving on ART and “viral suppression” targets. The definition of “linkage to care” has a wide variety of definitions, however, it is important to standardize its measurement within programs so that it includes everyone newly diagnosed with HIV and uses a reliable measure of whether they have accessed care. Common definitions of linkage to care include at least 1 CD4 count or other laboratory value, attendance of at least 1 clinic appointment, and/or pickup of ARV prescriptions among others.
The third step in the continuum is the total number of PLHIV on ART. It is defined as the number receiving ART at least once within the calendar year. The indicator should be expressed as both the number of PLHIV on ART and as a proportion of the number of PLHIV within the geographic setting. This is a key element of the continuum and determines the potential for sustained viral suppression, which is the ultimate objective of the continuum. The number of PLHIV picking up medications can be determined from providers or others as a surrogate measure and does not require clinical evaluation and/or other tests.
The numerator for the fourth step in the HIV care continuum is the total number of PLHIV with documented viral suppression within a given geographic setting (viral suppression is defined as at least 1 measurement of the lowest quantifiable level [eg, HIV RNA <200, 500, or 1000 copies/mL] during the measurement period). This indicator should be expressed as both a count and a proportion of the number of PLHIV within the geographic setting. Viral suppression among PLHIV is the single most important common goal of HIV care and prevention and the key element of the continuum.
In addition to the annual determination of the basic continuum measures, there is also considerable value in taking a longitudinal cohort approach to follow individuals living with HIV infection. Cohort methods allow for monitoring and evaluation of HIV service utilization and outcomes among individuals over time. Specifically, longitudinal cohort analyses from the time of diagnosis to ART initiation and viral suppression (including ART discontinuation and reentry, death, loss to follow-up, and other outcomes) can inform efforts to identify interventions at various stages of the HIV care continuum, the patient populations most in need of intervention, and those most likely to benefit. 285 Although the longitudinal cohort approach may appear to be complex, it has been successfully implemented in a number of diverse settings, including directly observed treatment, short-course–based TB control, noncommunicable chronic diseases, and also HIV in some settings, including in British Columbia, Canada, Malawi, Rwanda, and Switzerland. 279,286 –288
Attrition at different stages along the HIV care continuum is heterogeneous. It comprises individuals who dropout of HIV treatment for the first time, experience multiple treatment discontinuations, as well as those lost to HIV care entirely (ie, discontinued regular physician follow-up and diagnostic testing). The sometimes cyclical process of engagement, disengagement, and reengagement in HIV care has been referred to as continuum “churn” and has been cited as a crucial component of HIV monitoring. Persistence with ART, or sustained engagement (as opposed to adherence that measures the number of prescribed doses of ART that were ingested), is of particular interest, as the ultimate goal of sustained viral suppression cannot be achieved without such an engagement.
Key Populations
A number of distinct populations continue to be disproportionately affected by the HIV pandemic, including, but not limited to, women, adolescents, MSM, transgender individuals, sex workers, substance users, migrants and unstably housed people, and incarcerated populations. 289 –295 A growing body of evidence indicates that these populations also contend with an array of challenges associated with engagement or retention in the HIV care continuum. It bears noting that racial and ethnic disparities can both exacerbate the challenges faced by key populations and result in poorer overall health outcomes. 296 –298
Challenges vary across key populations, but there are some common challenges among these populations, including pervasive stigma and discrimination, 299,300 violence, 301,302 mistrust of medical providers or systems, 303 unmet basic needs of daily living (eg, food and shelter), 291,304 lack of access to culturally appropriate services, 305 underaddressed comorbidities, 306,307 and suboptimal access to evidence-based interventions due to these not being offered or brought to an appropriate level of scale. 289,308 Population-specific programs and policies that address these challenges are urgently needed. Further, although a growing body of evidence indicates that peer-delivered programs often help extend the reach, coverage, acceptability, and effectiveness of a range of programs, 309 –311 these approaches remain underutilized.
A number of key populations are also subjected to human rights abuses, as well as policies and laws that criminalize them and their behavior, and thereby undermine engagement in the HIV care continuum. Further, migrant, displaced, and mobile populations often face laws that prohibit their access to universal HIV treatment and care, and some are threatened with deportation upon HIV diagnosis. 312 Therefore, there is an urgent need for structural reform of a range of laws and policies in order to promote full engagement in the continuum. 290,313 Policy reforms that seek to ensure adequate access to evidence-based programs are also needed, including those that may be deemed controversial in some settings, such as harm reduction, opioid substitution therapy, and needle–syringe and condom distribution programs. 289,314
Although the development of evidence-based recommendations specific to key populations is beyond the scope of these guidelines, guidance on a series of issues specific to women, adolescents, MSM, transgender individuals, sex workers, substance users, migrant and unstably housed populations, and incarcerated populations is provided in Table 4.
Footnotes
Acknowledgments
The Chairs thank IAPAC for serving as the international advisory panel’s secretariat and the following individuals who were instrumental to the development of these guidelines: José M. Zuniga, PhD, MPH, Imane Sidibé, MSc, and Sindhu Ravishankar, MPhil (IAPAC; guidelines conception, and technical support, respectively); Cynthia Lyles, PhD, and Linda Koenig, PhD, (Centers for Disease Control and Prevention; providing access to and assisting with review of CDC Prevention Synthesis Project database for systematic review); Kathryn E. Buggraf, MPH; Anne E. Byers, MPH; Raissa Dickinson MPH; D. Dennis Flores, MSN, ACRN; Natalie M. Leblanc, BSN, MPH; Petra Lolic, MPH; Jacquelyn Malasky, MPH; Avani Patel, MPH; Kenisha M. Peters, MPH; Cathy Puskas, MSc; Cho Hee Shrader, MPH; Sara Wuite, MPH; Qian Zhang, MPH (systematic review); and Bandana Malhotra, MBBS, DVD (medical editing).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Development of the guidelines was jointly sponsored by IAPAC and the US National Institutes of Health (NIH), Office of AIDS Research (OAR).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
