Abstract
In this article, we sought to understand the perceptions and practice of providers on anal cancer screening in HIV-infected patients. Providers in an academic outpatient HIV practice were surveyed. Data were analyzed to determine the acceptability and perceptions of providers on anal Papanicolaou tests. Survey response rate was 55.3% (60.7% among male and 47.4% among female providers). One-third of the providers had received screening requests from patients. Female providers had higher self-rated comfort with anal Papanicolaou tests, with a mean score of 7.1 (95% confidence interval [CI] 4.7-9.5) compared to 3.6 (95% CI 1.5-5.7) for male providers, P = .02. Sixty-seven percent of male providers and 37.5% of female providers would like to refer their patients for screening rather than perform the test themselves. Only 54.2% of our providers have ever performed anal cytology examination. Our survey revealed that not all providers were comfortable performing anal cancer screening for their patients.
Introduction
The incidence rates of anal cancer in the United States have doubled from 0.8/100 000 person-years of follow-up in 1975 to 1.7/100 000 in 2010 (see Figure 1). Over the past 10 years, the incidence rates have increased by 2.2% each year, and death rates have increased by 3.7% per year from 2001 to 2011. 1 Multiple factors may be contributing to this rise in incidence of anal cancer; however, most experts believe that infection with the HIV increases the risk of developing this cancer, especially in men who have sex with men (MSM). Prior to the HIV epidemic, the incidence rates of anal cancer in men who reported receptive anal intercourse were estimated to be 35/100 000 person-years. This is similar to the incidence of cervical cancer prior to the use of generalized cervical cancer screening. 2 Studies in the post–antiretroviral therapy (ART) era have revealed that ART may not have a significant impact on the incidence of anal cancer in HIV-positive individuals. 3 Palefsky et al, in a review of the high incidence of human papillomavirus (HPV)-related cancers among HIV-positive patients, confirmed earlier concerns that ART would actually lead to a paradoxical increase in the incidence of these cancers, as HIV-positive individuals live longer and join the aging US population. 4 An analysis of data from the Swiss HIV Cohort revealed a standardized incidence ratio of 33.4 (95% confidence interval [CI] 10.5-78.6), comparing HIV-positive MSM to HIV-negative men. This study also demonstrated that among all non-AIDS-defining cancers, anal cancer had the highest relative increase in incidence among all HIV-infected patients irrespective of gender or HIV risk category and the second highest standardized risk ratio for women. 5 Silverberg et al 6 , using NA-ACCORD (North American AIDS Collaboration on Research and Design) data, reported anal cancer incident rate of 131/100 000 person years among HIV-positive men who have sex with men (MSMs). This study reports an incident rate ratio of 80.5, comparing HIV-positive MSMs with the HIV uninfected general population.
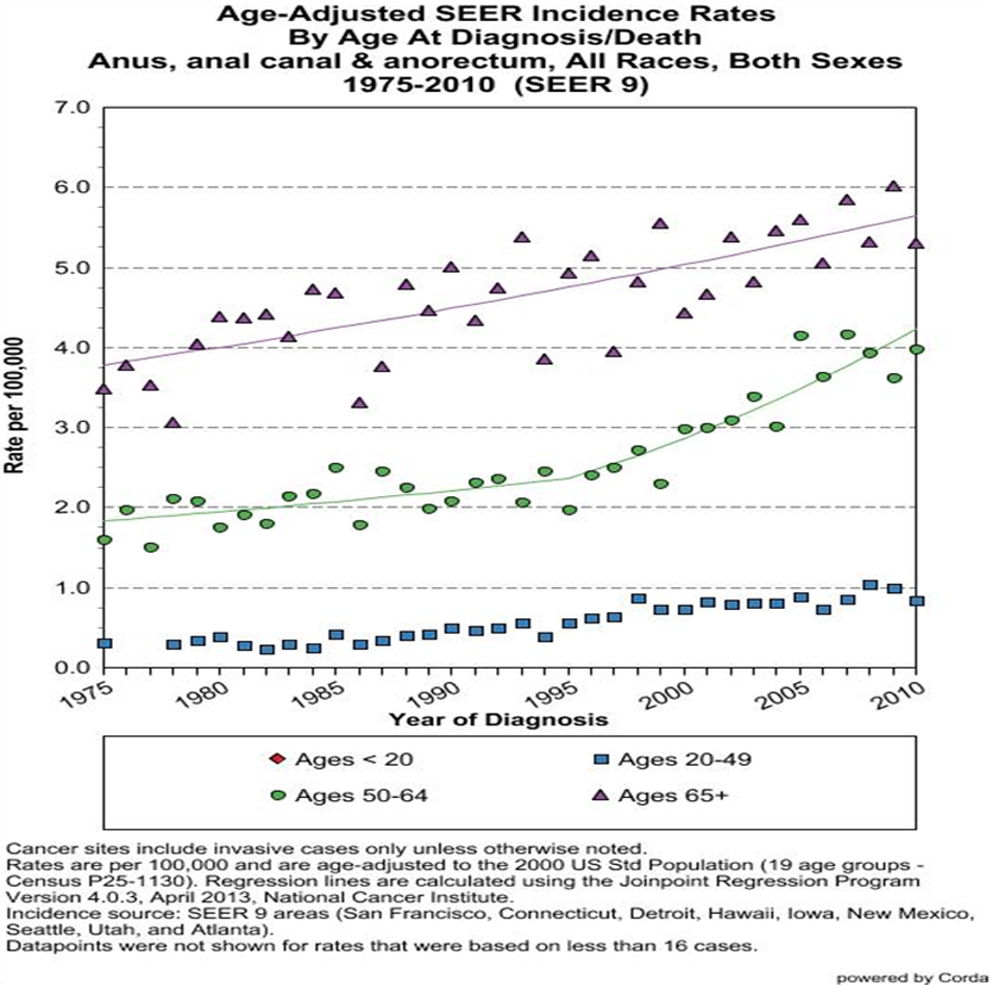
Trends in anal cancer incidence rates by age-groups from 1975 to 2010.
While there are currently no screening guidelines for anal cancer in the general population based on randomized controlled trials, recommendations based on expert opinion address anal cancer screening with anal cytology and high-resolution anoscopy (HRA) in specific populations. The New York State Department of Health recommends screening HIV-positive MSMs and HIV-infected women with a history of HPV-related lesions with anal cytology and HRA. 7 The HIV Medicine Association and the Infectious Diseases Society of America also suggest screening with anal cytology in similar populations without making any mention of HRA. 8 Center for Disease Control and Prevention Guidelines state that anal cytology screening may be considered in MSM, but note that there are limited data on efficacy of treatment modalities for high-grade squamous intraepithelial lesions. 9 The US Preventive Services Task Force however does not recommend routine screening for anal cancers in any population.
Prior research suggests that in Baltimore, Maryland, close to 62% of MSM in the MACS cohort study are aware of anal cancer screening in the community. 10 Other studies suggest that though MSMs accept anal cytology screening, some patients report that they perceived provider-related barriers to screening. In one focus group interview, a patient was quoted as saying “Physicians hate, hate to check your anus!” 11 Anecdotal evidence suggests that in our HIV clinics in Baltimore, Maryland, anal cancer screening practices vary widely. There are currently no institutional screening recommendations with regard to anal dysplasia/anal cancer screening. To better understand the attitudes and acceptability of screening for anal dysplasia and anal cancer in our practice, we surveyed all providers who provide care for HIV patients in our clinics.
Methods
In June 2013, we contacted all 47 active HIV providers in our practice by e-mail. Providers were asked to complete an online survey on their attitudes and practices with regard to anal cytology screening among their clinic patients. The texts for the e-mail message used to solicit participation of these providers and the survey questions are attached to this article in Appendices A and B, respectively. The survey tool was first piloted among a small sample of providers with attempts made to have the different provider types evaluate the clarity and internal consistency of the instrument.
Data from the survey were analyzed to determine the overall acceptability of the screening method to providers as well as to determine the perceptions of providers on anal Papanicolaou tests. Data analysis was done using STATA version 10 software. Chi-square tests were done to test for associations between different categorical variables, and student t test was used to test for associations between continuous variables. Analysis focused on determining the prior experience of providers in performing anal Papanicolaou tests as well as to determine whether provider characteristics had any correlation with their knowledge or perceptions with regard anal cytology screening.
Results
Provider and Respondent Characteristics
During the 3 weeks of the survey 47 providers were contacted. Twenty-six (55.3%) providers responded and completed the online questionnaire. Most (65.4%) respondents were males, and the response rate for males and females was 60.7% and 47.4%, respectively (P = .4). Providers who completed the survey were between 25 and 74 years of age, with 75% falling between ages 35 and 54 years. Two of the 26 respondents only provided answers to the demographic questions without answering other relevant survey questions, and both were excluded from further analysis. The mean age of respondents was 44.5 years, and female providers were relatively older compared to males (mean age: 48.2 ± standard deviation [SD] 12.46 vs 42.6 ± SD 7.04, P = .08). The respondents clinical experience in the care of HIV-infected patients ranged from 1 to 30 years with a mean of 12.5 years. Ninety-one percent of all respondents had more than 4 years of experience caring for HIV-infected patients. Female providers had on average more years of experience compared to male providers (17.0 years [95% CI: 8.83-25.17] vs 10.1 years [95% CI: 6.31-13.8], P value = .05).
Knowledge of the Procedure and Attitudes toward Screening Algorithms
Using a 0 to 5 scale for self-assessment of anal cytology-related knowledge, 66.7% of our respondents rated themselves ≥3 of 5 for knowledge. The mean score was 2.7, and perceived knowledge did not differ significantly by sex or age of the respondent. Female respondents however scored themselves on average higher than their male counterparts (mean score: 3.2 ± SD 1.49 vs 2.4 ± SD 1.63, P = .4). With regard to opinions on which population to screen, 52.2% of all respondents believed that all MSMs should be screened for anal cancer. Providers ≥45 years old were more likely to state that all HIV-infected patients should be screened compared to younger providers (who tended to recommend screening in specific population subgroup such as HIV-positive patients with genital warts; 57.1% of providers < 45 years vs 20% of providers ≥ 45 years, P value = .07). Younger providers were 5.3 times as likely to suggest screening in HIV-positive patients with anogenital warts compared to their older counterparts (P value = .07), and 50% of providers older than 45 years recommended screening for all HIV-infected patients versus 7.1% of providers aged 45 years and younger. Male providers were also 8 times more likely to recommend screening for HIV-positive patients with anogenital warts than female providers (P value = .04), and 56.3% of male providers recommended screening in this group compared to only 12.5% of females. Eighty-seven percent of all providers replied that they would be interested in attending a lecture on anal cancer screening. With regard to knowledge on HRA, most (60.8%) providers had heard about this procedure and were aware that it was used in the evaluation of anal cytological changes. Forty-three percent of respondents however mentioned that they knew little or nothing about this procedure. Among the 14 providers who responded that they knew about HRA, 9 believed that any trained provider should be able to perform HRA and did not believe this was a procedure that required a surgeon. One provider failed to provide an answer to this question.
Provider Practice and Perception of the Procedure
Of the 24 providers who provided responses to this questionnaire, 13 (54.2%) had performed anal cytology smear on their patients. Female providers were 11.7 times more likely to have performed the procedure compared to males (P = .02). Most providers reported that they were fairly comfortable performing the procedure, and 58.3% rated their comfort level with the procedure at a level of 5 and above on a 0 to 10 arbitrary comfort scale, with a mean comfort scale of 49 of 10 (95% CI 3.3-6.6). Provider self-rated comfort in performing anal Papanicolaou tests was higher for females compared to males, 7.1 (95% CI 4.7-9.5) versus 3.9 [95% CI 1.8-5.9], P = .02; see Figure 2). Just over a third (34.8%) of the providers reported that patients had requested to have screening in the past. Providers younger than 45 years and females were more likely to have received a request for an anal Papanicolaou tests compared to those 45 years and older, although differences were not statistically significant. Of all providers surveyed, 58.3% would prefer to refer their patients to a different provider for anal cytology screen, and 68.5% of male providers versus 37.5% of female providers would consider referral (P value = .2). By age, 50% of older providers and 64.3% of providers ≤45 years would consider referral (P value = .3). The majority of respondents believed that anal cytology screening is a service that would be important for our practice and recognized the current lack of screening as a gap in care. Only 1 person of the 23 providers who provided responses to the survey did not favor a formal screening program in our practice.

Comparing provider comfort level with the performance of anal cytology screens by sex.
Discussion
Our survey revealed that while anal cytology screens are performed by more than 54% of our providers, female providers were 11.7 times more likely to male providers to perform this screening test. This result is similar to prior studies on performance of cancer screening tests among physicians. Franks and Clancy 12 in a survey among female patients revealed that those with female providers were significantly more likely to undergo screening with cervical Papanicolaou tests and mammograms compared to women with male providers. A 1997 study using a Minnesota Health plan utilization data found a similar physician gender bias in performance of Papanicolaou tests and mammograms. 16 Other studies have found that female providers focus more on prevention and education in their practices. 13 –17 However, for prostate and colon cancer, no clear gender biases of the provider are described. Gender congruence of physicians and patients especially with regard to intimate procedures and issues may be responsible for this observed difference. There is currently no study in the literature on physician behavior regarding screening of MSMs. Forty percent of the providers in our practice are females and therefore if male providers are less comfortable with this procedure and are less likely to offer the screening for their patients, a significant proportion of our patients may not get screened. We are not aware of any prior published studies that have evaluated this provider gender bias in performance of anal cytology screens; however, Newman et al 11 in a focus group interview looking at barriers to anal cancer screening among gay men revealed that several patients felt their physicians were not very comfortable with this type of screening. Thus, further studies in this field are required.
In our practice, female providers had more years of experience caring for HIV-infected patients, 17.0 years, compared to 10.1 years for males. This may explain the relative comfort with this procedure among female providers as opposed to males. However, younger providers defined in this study as younger than 45 years of age were 10 times as likely to consider screening for both males and females with evidence of anogenital warts.
High-resolution anoscopy is currently used to evaluate abnormal anal cytology screening. This procedure has been considered as being analogous to cervical colposcopy and is based on similar pathophysiological mechanisms. In our survey, 47% of respondents reported they had little or no knowledge of this procedure. On the converse, 48% of all respondents are aware that HRA would be the ideal form of evaluation for an abnormal anal cytology test. However, several providers were not sure to whom to refer their patients for an HRA. This is unfortunately a common situation as HRA is a procedure performed by different provider types, 17 and there is not currently any provider offering HRA in the clinic setting in our practice. Doctors at the University of California at San Francisco have utilized the procedure to evaluate abnormal anal cytologies since the early 1990s. 18 The practice has however become more widespread in the past few years, and more and more providers are being trained in this procedure. 17 Due to the low sensitivity and specificity of anal cytologies, some experts believe HRAs should be the initial screening test in this population. 19 While limitations in the number of providers offering HRA are currently limiting the availability of the procedure, HRA is the gold standard for further evaluation of abnormal cytology screens. Hence, at least large health systems with significant number of HIV-positive patients must consider setting up a systematic screening program for their patients. Third-party payers reimburse HRA providers at various levels ranging from US$40 to US$250.
Limitations and Strengths
Our study has some limitations. First there was a relatively small sample size, we surveyed 55.3% of the 47 HIV providers in a large urban academic institution, and this may not be generalizable to all HIV providers. Survey does not have any data on nonrespondents, so the respondents may not be representative of the HIV providers within the practice. However, to our knowledge, this is the first study conducted to evaluate provider attitudes toward this important screening modality.
Conclusion
Anal cancer screening must be considered as an integral part of the health maintenance screening for all MSMs, especially those who are HIV positive. Physicians taking care of such patients must examine their comfort level as well as proficiency at performing this procedure. Providers who do not feel comfortable performing this procedure must refer to appropriate colleagues. Providers who provide screening must ensure that patients are well educated on the potential “benefits” and harms of the procedure prior to initiating screening.
Footnotes
Appendix A
Appendix B
Authors’ Note
All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. LAS conceptualized the study, designed the article idea, analyzed the study data, wrote the initial draft coordinated, and contributed to each revision. UKB was involved in the conception and design of the study, data interpretation, wrote part of the discussion and provided critical review and revision of the manuscript. DJR was involved in data interpretation and provided critical review and revision of the manuscript. BLG was involved in data interpretation and provided critical review and revision of the manuscript. MK was involved in the interpretation of the data and provided critical review and was involved in revisions to the manuscript. LF was involved in data interpretation and provided critical review and was involved in revisions to the manuscript. DES provided critical reviews and was involved in revisions to the manuscript. JW provided critical review and was involved in revisions to the manuscript. GT provided critical review and was involved in revisions to the manuscript. MS provided critical reviews and was involved in revisions to the manuscript. AA provided critical review and was involved in revisions to the manuscript. RRR oversaw the implementation of the project, data collection, provided critical reviews, and was involved in all the revisions to the manuscript. All authors reviewed all draft versions and approved the final version. The authors thank all the HIV providers of the University of Maryland Medical System who responded to the questionnaire.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
