Abstract
Non-acquired immunodeficiency syndrome-defining cancers (NADCs) are malignancies in persons living with human immunodeficiency virus (PLWHIV) and are not primarily due to the host’s immunodeficiency. There is renewed clinical interest in long-term morbidities in PLWHIV as well as malignancies that occur in this population. We herein describe a 36-year-old woman with a 2-year history of an anal wound and right breast mass. She had been diagnosed with HIV infection prior to the development of these lesions. Clinical and laboratory evaluations led to diagnoses of breast and anal cancers. Chemotherapy and antiretroviral therapy were begun, but the patient discontinued these treatments early and was lost to follow-up. NADCs will continue to be a major clinical issue as the global population ages. This presentation of two NADCs (breast and anal cancers) in a PLWHIV further highlights the burden of multiple malignancies on the depleted health of HIV-infected patients. Early identification and treatment of HIV upon patients’ presentation to cancer care sites and screening for NADCs at HIV/AIDS care sites are recommended for improved outcomes.
Keywords
Introduction
Non-acquired immunodeficiency syndrome-defining cancers (NADCs) are malignancies in people living with human immunodeficiency virus (PLWHIV) and are not primarily due to the host’s immunodeficiency. 1 NADCs are a growing cause of morbidity for PLWHIV. 2 Highly active antiretroviral therapy (HAART) has transformed HIV infection from a fatal disease to a chronic condition.3,4 The association between HIV and malignancies became evident early in the HIV epidemic. Kaposi sarcoma (KS) was the first described AIDS-defining cancer (ADC); others include cervical cancer and non-Hodgkin lymphoma.1,3 These three ADCs are due to HIV-induced T-cell reduction and consequent immunosuppression. 1 Since the introduction of HAART, the incidence of NADCs has increased among PLWHIV in both developed and developing countries. This rise is projected to persist in future, whereas the incidence of ADCs has decreased.2,3 There is renewed clinical interest in long-term morbidities as well as malignancies that occur in PLWHIV. 4 This population is at higher risk of cancers that affect the general population, including breast, colon, and prostate cancers. 1 The risk is especially high in people with a confirmed or suspected infectious etiology. 4 PLWHIV have a markedly increased risk of anal cancer and other human papilloma virus (HPV)-associated malignancies. 5 The impact of antiretroviral therapy (ART) on the incidence of anal cancer is somewhat unclear. The improved survival of HIV-infected persons in the HAART era has resulted in longer exposure to HPV infection, which may lead to an increased risk of HPV-associated cancers. By contrast, some reports indicate that HAART results in better control of co-infections and underlying immunosuppression. 4 Among women with HIV, breast cancer is the most common type of NADC. Although the oncologic relationship between HIV/AIDS and certain cancers is well understood, there is little evidence regarding breast cancer among PLWHIV. 6 Recent studies have shown that PLWHIV with certain NADCs have a poorer prognosis and shorter survival than people in the general population. 3 Understanding the evolving patterns of both ADCs and NADCs in PLWHIV will direct screening and appropriate treatment strategies with improved outcomes. 2 We herein report a rare case of an HIV-positive woman with two NADCs that, interestingly, had an infectious and non-infectious etiology.
Case report
A 36-year-old woman presented with a 2-year history of an anal wound and right breast mass. She had been in her usual state of health until 2 years previously, when she noticed an anal growth that exhibited an insidious onset. It started as a small boil that broke into a wound and later grew into a large nonhealing perianal wound. This was preceded by the appearance of itchy rashes on the buttocks. Breast swelling, also with an insidious onset and starting at about the same time as the anal ulcer, initially manifested as small masses that increased in size and became multilobulated. There was associated pain, which the patient described as sharp, constant, and non-radiating. There was also associated bloody nipple discharge and axillary swelling. The patient reported a history of weight loss. Menarche had occurred at 13 years of age. She was a primipara and did not use contraceptives. Her elder sister had been diagnosed with breast cancer earlier. Twenty months prior to this presentation, the patient had been diagnosed with HIV infection; she commenced treatment but then discontinued it after 2 months.
Physical examination revealed a woman in no obvious distress. She had no fever or icterus, was not pale, and had no pedal edema. Her pulse rate was 70 beats/minute, respiratory rate was 18 breaths/minute, temperature was 37°C, and blood pressure was 120/70 mmHg. Breast examination revealed a normal left breast and ipsilateral axilla. However, the entire right breast revealed features consistent with advanced breast cancer (T4d, N-2) (Figure 1(a), (b)). Chest and abdominal examinations were unremarkable. Rectal examination revealed an anal ulcer on the left side extending to the adjoining gluteus with a raised and everted edge. The adjoining bilateral gluteal skin showed rashes. No ipsilateral inguinal lymphadenopathy was present (T3N0) (Figure 2). Clinical diagnoses of advanced right breast cancer and a malignant left perianal ulcer were made.
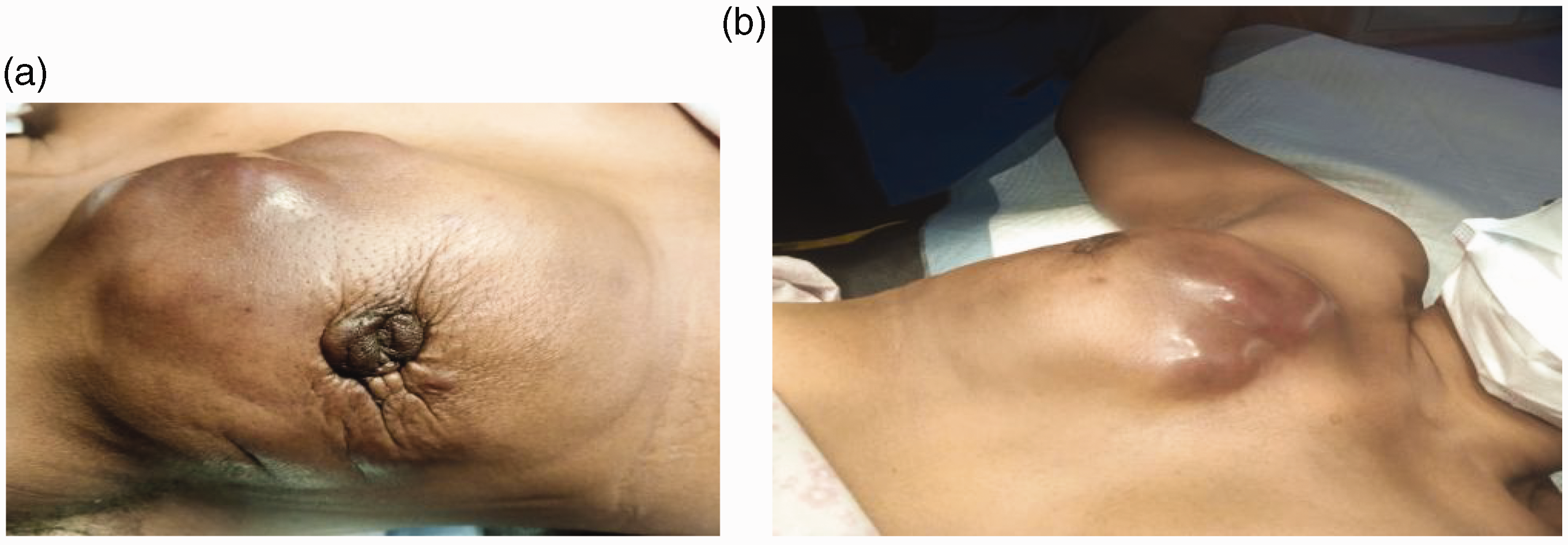
Images of right breast cancer. (a) Lateral view of right breast cancer showing features of inflammatory breast cancer and (b) medial view showing features of right inflammatory breast cancer.
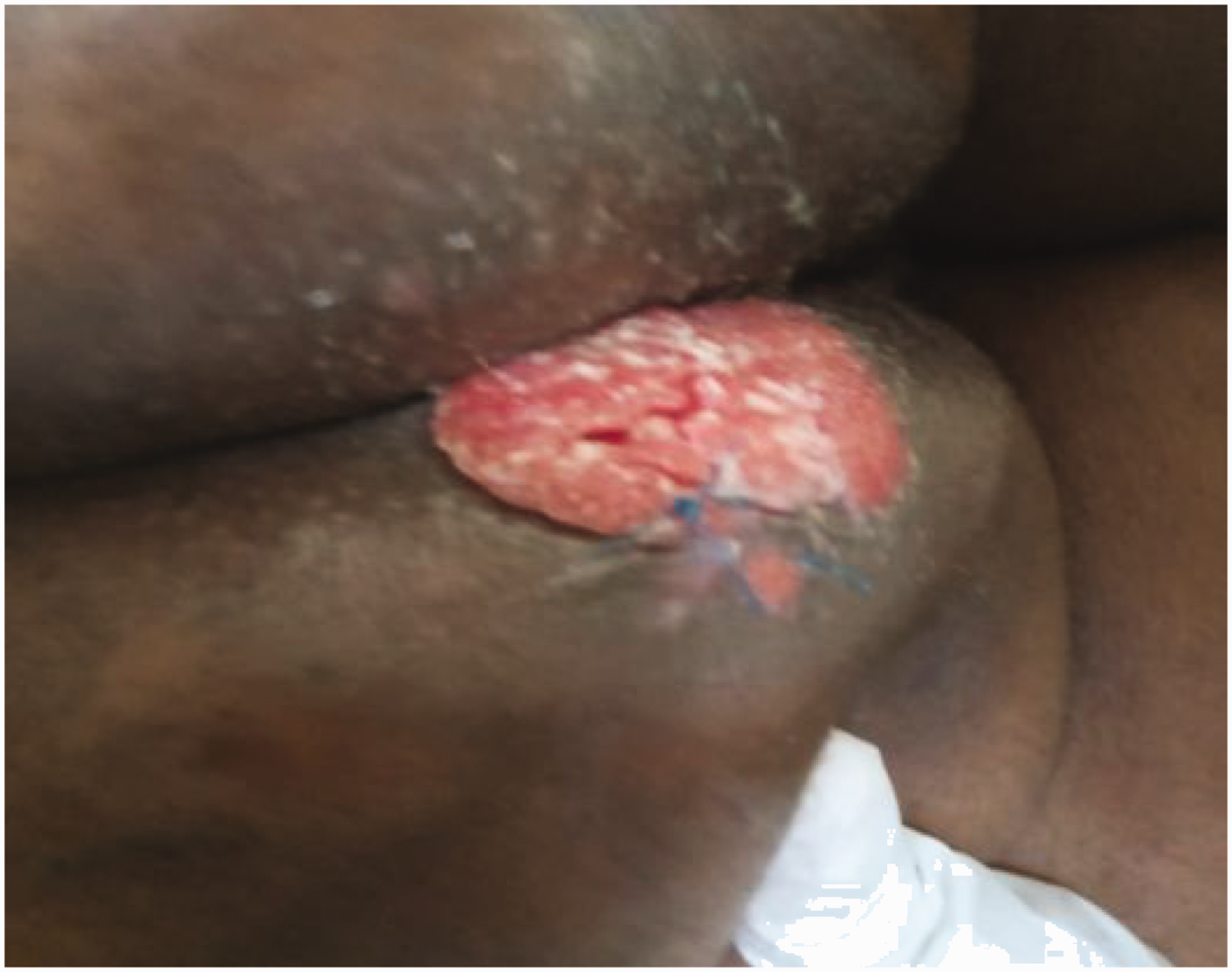
Anal and perianal malignant ulcer with adjoining dermatitis (squamous cell carcinoma).
Investigations included a full blood count with the following results: packed cell volume, 32%; white blood cell count, 6.0 × 103/µL; lymphocytes, 34%; neutrophils, 62%; eosinophils, 4%; monocytes, 0%; basophils, 0%; and platelets, 265 ×103/µL. HIV-1 and HIV-2 serology was positive, the CD4 count was 294 cells/mm3, and urinalysis results were normal. Tru-Cut biopsy of the breast mass and incisional biopsy of the perianal ulcer showed invasive ductal carcinoma and squamous cell carcinoma, respectively (Figures 3(a), (b) and 4(a), (b)). Immunohistochemical examination revealed triple-negative breast cancer. The definitive diagnoses were advanced right breast cancer and anal squamous cell carcinoma with HIV infection.

Invasive ductal carcinoma of the breast with inflammatory infiltrates. (a) Hematoxylin and eosin, ×40 and (b) hematoxylin and eosin, ×100.

Microscopic images of poorly differentiated squamous cell carcinoma. (a) Anal tissue showing malignant squamous epithelial cells in nests and sheets infiltrating the stroma (hematoxylin and eosin, ×40) and (b) anal tissue showing malignant squamous epithelial cells in nests and sheets with intense inflammatory infiltrates and hemolysis (hematoxylin and eosin, ×100).
The patient was counselled and advised to resume antiretroviral therapy (tenofovir, lamivudine, and dolutegravir). She received two doses of docetaxel and cyclophosphamide and was lost to follow-up.
The patient provided both verbal and written informed consent for the reporting of this case and all accompanying images. In addition, she provided written consent for treatment. The requirement for ethics approval was waived because this study was a report of a case managed by a clinical team at the University of Calabar Teaching Hospital and not an interventional study (i.e., no intervention or experimentation was conducted for the purpose of the study). The reporting of this study conforms to the CARE guidelines. 7
Discussion
The incidence of NADCs is increasing in HIV-infected persons, and the prevalence of these cancers ranges from 7% to 15% as a cause of morbidity in the HIV population. 8 Chiao et al. 2 reported lung, prostate, breast, colorectal, anal, and head and neck cancers as common NADCs in decreasing order of frequency. Their study excluded Hodgkin’s lymphoma. As the life expectancy and prevalence of women living with HIV has increased, there has been a three-fold increase in the incidence of certain NADCs in the United States. Some reports from Sub-Saharan Africa have shown similar trends, whereas others have indicated contrasting findings. For example, Botswana showed a decline in ADCs such as KS, and Tanzania showed an increase in NADCs. 6 In the largest African study conducted in South Africa, there was no overall increase in NADCs among PLWHIV compared with HIV-negative people. 9 This finding was consistent with a study in Nigeria. 10 However, the South African study revealed a higher risk of anal cancer and lower risk of breast, prostate, and lung cancer in PLWHIV than in HIV-negative people. 9 HIV-infected persons had a greater than nine-fold increased risk of infection-related NADC compared with HIV-negative persons, and this was largely influenced by differences in the risk of anal cancer. 8 In a study from Kenya, the most common NADC in PLWHIV was breast cancer (invasive ductal carcinoma), with an incidence of 18.8%, followed by lung and anal squamous cell carcinoma. 1 Some reports have suggested a decreased frequency of breast cancer in PLWHIV, whereas others have reported the contrary. 8 Mpunga et al. 11 found that conjunctival cancer, Hodgkin lymphoma, and anal malignancies were the most frequent. Anal cancer, once a rare entity usually found in elderly women, has increased in incidence, especially in PLWHIV. 12 During the past 20 years, the burden of anal cancer has increased by 60% in the United States and by more than three-fold in Africa. The rate of anal cancer has increased by 20 times in PLWHIV, with the highest increase (50 times) in HIV-positive men having sex with men.13,14 Among patients with multiple NADCs, concurrent breast cancer and anal cancer in PLWHIV is a rare presentation, and a literature search revealed no such previous reports.
Reports suggest that HIV-positive patients present with breast cancer at an earlier age than do HIV-negative patients. 8 In high-income populations, the median age at diagnosis of breast cancer is 61 years in HIV-negative patients but is 10 to 15 years younger than this in HIV-positive patients. However, breast cancers in sub-Saharan Africa generally present at a relatively younger age regardless of HIV status. 6 In our case, the patient was 36 years old at presentation but 34 years old at the onset of symptoms, which is consistent with the overall mean age at presentation of West African women between 35 and 45 years of age. 6 Furthermore, our patient had a history of breast cancer in her family (elder sister). Having a mother, sister, or daughter (first-degree relative) who has been diagnosed with breast cancer approximately doubles the risk of breast cancer. When multiple patients within a single pedigree develop cancer, the condition is termed familial cancer. When a strong genetic background is present, the condition is termed hereditary cancer. Approximately 5% to 10% of breast cancers are hereditary. 15 Breast cancer is more genetically relevant. Hereditary breast cancers present in younger age groups. Patients treated at Jackson Memorial Hospital in Miami were diagnosed at a median age of 44 years. 8 Anal cancer has historically been considered a rare cancer traditionally associated with elderly women. HIV-positive patients tend to develop anal cancer at a younger age. 12 Consistent with this, our patient presented at the age of 36 years. In a study in Nigeria, anal cancer accounted for 5.9% of anorectal lesions seen in HIV-positive patients. 16
Our patient presented with two NADCs representing malignancies in two major categories: anal squamous cell carcinoma and breast cancer of infective and non-infective etiologies, respectively. NADCs with an infective etiology occur at higher rates among PLWHIV than in the general population and are represented by cancers associated with HPV infection (cancers at anogenital sites). 3 Anal cancer commonly manifests as part of the phenotype of HIV infection. Infection-related NADCs might be largely due to HIV-induced immunodeficiency, which reduces the body’s ability to fight infections and allows for proliferation of oncogenic pathogens in PLWHIV. 3 This is in addition to the high-risk lifestyles prevalent among PLWHIV, which can increase the possibility of co-infection with oncogenic viruses because both HIV and HPV are sexually transmitted.2,3 HIV-induced immunosuppression might be a risk factor for some noninfectious cancers, such as breast cancer. It is believed to induce oncogenesis by disturbing the normal cell cycle, inducing oncogenic dysregulation, and increasing oxidation stress.3,8 Breast cancer develops because of the complex interplay among host, genetic, metabolic, immunologic, and environmental factors. Several malignancies have a well-defined association with viruses (e.g., KS and human herpes virus 8); by contrast, breast cancer has no established association with any viruses (including HIV), although several have been suggested. 6 A few studies have suggested an accelerating or detrimental effect of HIV on breast cancer. However, others have suggested a protective effect of HIV against breast cancer because HIV infection activates cell death pathways, which may lead to a decreased cancer burden in these patients. 8
In the context of HIV infection, breast cancer often presents as an advanced and markedly aggressive disease despite exhibiting favorable biologic prognostic indices compared with HIV-negative women.2,6 This was illustrated in our patient, who presented with an inflammatory tumor involving the entire right breast (Figure 1(a), (b)). However, in our setting, presentation with late-stage disease is common even without HIV infection. One study indicated that the average duration with HIV infection prior to the diagnosis of breast cancer was 8.8 years. 8 This duration in our patient was 2 years, having discontinued treatment after commencement of therapy following the diagnosis of HIV infection. However, the clinical importance of the length of time with HIV infection prior to the development of NADC has not been established. 8 Anal cancer in Tanzania is associated with a high HIV prevalence and locally advanced disease, 13 as shown in our patient with remarkable perianal involvement (Figure 2).
The clinical evaluation of PLWHIV includes a complete physical examination with special attention given to the oral cavity, skin, and lymph nodes. This should be performed in addition to the evaluation of comorbidities and co-infections. Basic laboratory evaluations include HIV tests, blood counts, and blood chemistry. The CD4+ cell count is a laboratory test that measures the number of CD4 T cells, with the reference range being 500 to 1500 cell/mm3. This test is used by clinicians to monitor the destruction of CD4 cells and measure the effectiveness of ART. Three types of HIV tests are available: antibody tests, antigen/antibody tests, and nucleic acid tests. Each test has a different time window. The clinical name for an HIV test is a nucleic acid test. This test can show whether the patient has HIV and how much virus is present in the blood (HIV viral load). It detects HIV sooner than other types of tests. More severe HIV infection has been associated with an increased risk of many NADCs. In population-based studies, a CD4 cell count of <200 cells/mm3 after 6 months of ART and long periods of low CD4 cell counts have been associated with increased standardized incidence ratios for NADCs. 5 The CD4 count of our patient was 294 cells/mm3, which is comparable with the count of 309 cells/mm3 recorded in Miami. 8 A study from the University of Maryland showed that the median CD4 count was 437.5 cells/mm3 in an Italian cohort, and 50% of South African patients with breast cancer and HIV had counts between 200 and 499 cells/mm3. In another study, CD4 counts among HIV-infected patients were lower in those with anal NADCs and Hodgkin’s lymphoma. The current CD4 count was the most significant predictive factor for all malignancies except anal cancers. 4 The relationship between the CD4 count and anal cancer is also unclear, but the above-mentioned study showed that longer exposure to a CD4 cell count of <200 cells/mm3 and a count of 200 to 350 cells/mm3 were significantly associated with anal cancer. 4
Regarding breast cancer characteristics, Ruiz and Davis 8 reported that 40% of patients had a triple-negative receptor status, which is correlated with more aggressive and less responsive types of breast cancer. Our patient had triple-negative breast cancer. Treatment involves a combination of surgery, chemotherapy, radiotherapy, and hormonal therapy, and HIV-infected patients also receive ART. Our patient received chemotherapy involving a combination of docetaxel and cyclophosphamide. The ART was a combination of tenofovir, lamivudine, and dolutegravir. In one cohort, 20% of patients underwent surgery as the only therapeutic intervention. 8 Our patient, who had advanced locoregional disease, was offered chemotherapy in addition to ART and not surgery. The reasons for this treatment recommendation were late presentation with advanced locoregional disease, a delayed diagnosis, and the inherent characteristics of HIV-related tumor biology. The average survival of 5.2 years aligns with findings from other studies, some of which reported even lower figures. 8
Chemoradiation is the key to disease control while preserving quality of life (avoiding colostomy) in patients with anal cancer. 13 Our patient presented with a stage II tumor with extensive perianal skin involvement. The treatment guideline for anal cancer in PLWHIV suggests intensity-modulated radiation therapy. Chemoradiation with cisplatin, fluorouracil, and cetuximab with an external beam of 45 to 54 Gy is recommended. 13 Immune restoration might decrease toxicity, and the addition of cetuximab reduces locoregional progression of the disease. Single-agent immunotherapies with pembrolizumab and durvalumab are safe in PLWHIV with a CD4 count of >100 cells/mm3. 17 ART-era data suggest that immune restoration might be associated with a decline in standard toxicity related to fluorouracil-based chemoradiation therapy. 2 Annual anal cytology screening for PLWHIV followed by high-resolution anoscopy for triage of abnormal cytology results is recommended.2,12 Despite the high 5-year survival rate among patients with early-stage anal cancer in Western countries, the survival rate dramatically decreases from 78% in patients with local disease to only 18% in those with metastatic disease. 13
The overall median survival of patients with NADCs is 1.6 years. 6 The future of prevention and treatment of NADCs for PLWHIV lies in immunotherapy. This necessitates increasing research capabilities and clinical research activities to support evidence-based prevention and treatment focused on NADCs.
Our patient discontinued treatment twice. This is a particular challenge in our setting and is associated with fear of stigmatization, cultural beliefs, and poverty, leading many individuals to seek care from alternate medical practitioners. Addressing these issues requires the provision of health education with an appropriate context and cultural sensitivity. 18
The HIV-infected patient described in this report presented with two NADCs: breast and anal cancers with non-infective and infective etiologies, respectively. The morbidity and mortality rates associated with NADCs have increased. 19 These tumors constitute a source of morbidity in association with HIV infection. Behavioral factors associated with high-risk lifestyles and poor compliance with treatment due to sociocultural beliefs, poverty, and fear of stigmatization contribute to poor outcomes in our setting. Large population-based studies have shown that PLWHIV with NADCs have poorer outcomes than those without NADCs. 2 The current approach to treatment is based on a combination of ART and antineoplastic therapy. Interventions such as immunotherapy will be needed to improve NADC outcomes. Understanding the changing patterns of both breast and anal NADCs will direct screening and treatment strategies to achieve optimal outcomes for PLWHIV. 2
Conclusion
NADCs will continue to be a major clinical issue as the global population ages. This is further compounded by the presentation of multiple NADCs (breast and anal cancers) in PLWHIV, placing a further burden on patients’ health. PLWHIV with a good performance status should be offered guideline-based cancer treatments. Oncologists should work as a multidisciplinary team with infectious disease physicians and pharmacists to manage potential drug interactions and side effects. Early identification of HIV infection upon presentation to cancer care sites and treatment with ART are important. Screening for NADCs at HIV care sites, increasing research capabilities, and performing further clinical research will improve outcomes.
