Abstract
Background:
Hypersensitivity reaction to antiretroviral treatment (ART) poses potential threats in maintenance of treatment. Lamivudine (3TC), is rare to cause rash. We are reporting 23 cases of 3TC-induced rash.
Methods:
An observational study conducted in the antiretroviral treatment center of a tertiary care hospital of North India from Feb 2009–Dec 2013 to record 3TC-induced rash. These were then recommended to start ART without 3TC and were followed up at 1-, 2-, and at 4-week intervals to monitor the toxicity, if any, with alternate therapy.
Results:
We observed 3TC-induced skin rash in 23 HIV-infected individuals (0.7%), out of 3213 HIV-infected individuals initiated on first line ART (zidovudine [ZDV]/tenofovir [TDF] + 3TC +nevirapine [NVP]/efavirenz [EFV] during the study period of 5 years [Feb 2009–Dec 2013]). The mean age of these 23 individuals was 37.5 ± 12.8 (17-60) years. Lamivudine rash was more common in women than men (F = 19, M = 4), with an overall mean age of 37.5 ± 12.8 (17-60) years. It was generalized, erythematous, maculopapular eruptions associated with intense itching with no associated mucosal involvement. Lamivudine was substituted with TDF in 19, didanosine (ddl) in 3 and abacavir (ABC) in 1 individual. Mean duration of follow-up is 11.1 ± 12.8 (3-42) months. CD4 count was repeated at 3 months and showed significant improvement (
Conclusion:
Lamivudine-induced rash was found at a frequency of 0.7%. The correct and early recognition that the rash is due to 3TC, would save unnecessary substitution to a different class of drugs.
Introduction
Highly active antiretroviral therapy (HAART) has shown a significantly reduced morbidity and mortality in HIV-infected patients. 1 The HAART regimens consist of a combination of at least 3 antiretroviral (ARV) drugs drawn from various classes, which include nucleoside reverse transcriptase inhibitors (NRTIs) or nucleotide reverse transcriptase inhibitors (NtRTIs), nonnucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (PIs), integrase inhibitors, and fusion inhibitors. Two or more NRTIs typically form the backbone of HAART regimens. 2 These drugs have considerable activity against this retrovirus. To attain viral suppression to undetectable levels, nearly 100% adherence is required. 3 The development of drug toxicity limits the adherence of the HIV-infected individuals. 4 Cutaneous drug reactions in HIV-infected individuals result in substantial morbidity. Within the first year of treatment, drug side effects are common reasons for the discontinuation of HAART among HIV-infected individuals. Drug toxicity is presumably thought to be the result of immune dysregulation, altered drug metabolism, and/or polypharmacy. 5 HIV-infected patients have a higher risk of developing cutaneous reactions than the general population. 6 These adverse events have significant impact on patients current and future care options. The overall size of the problem is substantial, and drug hypersensitivity complicated 3% to 20% of all prescriptions in one large series by Coopman et al. 7 It has also been reported that the incidence of cutaneous diseases including drug reactions increases as the immune function deteriorates. All licensed NNRTIs, abacavir (ABC; NRTI), and the PI amprenavir (APV) are common ARV drugs reporting to cause hypersensitivity reaction. Lamivudine (3TC) is an integral component of all regimens in the national guidelines. It is the drug considered to be safest and least toxic, is nonteratogenic, and is also effective against hepatitis B virus (HBV). 8,9 This drug is also preferred in patients with risk for hepatotoxicity of antiretroviral therapy (ART). Clearance of 3TC is dependent on weight and renal function and is not influenced by gender, disease stage, CD4 count, or race. 10 This nucleoside analogue is commonly used in multidrug therapy of HIV-1 disease, as it not only potentiates the antiviral effects of other reverse transcriptase inhibitors, but it is also relatively nontoxic. Even in the presence of point mutation resulting in M184V, 3TC exerts antiviral activity and is therefore given in second-line combination too. Here we report series of 23 individuals reviewed in State AIDS Clinical Expert Panel (SACEP) meeting of Centre of excellence in HIV care at a tertiary care center in North India, who developed hypersensitivity reaction in the form of skin rash due to 3TC when initiated on HAART.
Materials and Methods
This retrospective, observational study was conducted in the Centre of excellence in HIV care, Department of Internal Medicine, Postgraduate Institute of Medical Education & Research, Chandigarh, India. The study period extended from January 2009 to December 2013. HIV-infected individuals eligible for ARV treatment per the national guidelines were initiated with first-line drugs, which included zidovudine (ZDV)/stavudine (d4T)/tenofovir (TDF) + 3TC + nevirapine (NVP)/efavirenz (EFV) after ART preparedness, counseling sessions, and a consent for ART initiation. 11 These drugs were given as fixed drug combinations. Lead-in periods of 14 days were given when initiated with NVP-based ART. Patients were advised strict adherence to ARV drugs and to report in the center if signs/symptoms of toxicity appear. Individuals who developed grade 4 toxicity with NVP-based combination or drug rash with EFV-based combination were advised to stop treatment. These were later restarted after SACEP review with boosted PI-based ARV drugs after their rash resolved completely. The revised regimen consisted of ZDV/d4T/TDF + 3TC + atazanavir (ATV)/lopinavir (LPV) boosted with ritonavir (RTV). Individuals who again developed hypersensitivity reaction with this combination were reviewed in SACEP once again. The opinion of National AIDS clinical expert physician was further taken for these patients. Lamivudine was later suspected to cause toxicity, as it was found in fixed dose combination in both first and alternative treatment lines with ZDV/d4T/TDF. Photodocumentation of the rash of the individuals reporting hypersensitivity reaction was done in the prestructured proforma after obtaining informed consent. Demographic details, any history of alternative therapy taken along with HAART, past history of ARV drug exposure, history of coinfection, opportunistic infection, and substance abuse were recorded. These individuals were then advised to start ARV drugs without 3TC and were followed up at intervals 1, 2, and 4 weeks, so as to monitor the toxicity in the form of rash or any other side effect. Baseline investigations in the form of hemogram, biochemistry that included liver and renal function tests, and lipid profile were done per standard National AIDS Control Organization (NACO) ARV treatment guideline protocol. The study has been approved by the Institutional Ethical Committee.
Results
During the study period of 5 years, 3213 (male: 2127, female: 1081, and transgender: 5) HIV-infected patients were initiated with first-line ARV drug combination as recommended by national ART guidelines. One hundred and four (3.2%; male = 52 and female = 52) developed hypersensitivity reaction in the form of rash with NVP. Fifty-one (1.6%; male = 19, female = 31, and transgender = 1) had rash with EFV. Twenty-three patients (0.7%; female = 19 and male = 4) were observed to have 3TC-induced skin rash. Table 1 shows the demographic details of patients diagnosed with 3TC hypersensitivity reaction. Rash was more common in women than in men (female = 19 and male = 4), with an overall mean age of 37.5 ± 12.8 (n = 23; range: 17-60) years. Mean duration of HIV seropositivity was 42.3 ± 41.8 (range: 4-184) months. Mode of HIV acquisition was heterosexual in 21, and history of blood transfusion and perinatal route of acquisition was seen in one individual each. None was coinfected with HBV or hepatitis C virus. There was no history of past or recent opportunistic infection. None reported history of past or recent substance abuse. None of the individuals presented with history of past/concomitant alternative medicine intake. One woman reported history of monotherapy (indinavir [IDV] 800 3 times daily) taken for 3 years before initiation of HAART. Mean baseline CD4 count before HAART initiation was 278 ± 99 cells/mm3 (n = 23, range: 81-566). Mean duration of development of toxicity in the form of rash when initiated with first-line HAART (ZDV/d4T/TDF + 3TC + NVP) was 26.5 ± 11.1 (n = 22, range; 10-53) days. There was associated transaminitis and Stevens Johnson syndrome in 4 women. Antiretroviral therapy was stopped, and after the rash resolved completely, NVP was replaced with EFV (n = 19) or ATV/r or LPV/r-based regimen (n = 4), with continuation of nucleoside reverse transcriptase backbone (ZDV/d4T/TDF + 3TC). Subsequent development of rash when suspected with 3TC occurred in average 4.6 ± 5.1 days (4 hours-17 days). This hypersensitivity reaction was generally in the form of generalized, pruritic erythematous, maculopapular eruptions. There was no associated mucosal involvement this time in any of the patient. The recommended algorithm by NACO for reintroduction of drugs in case of suspected drug toxicity was followed (Figure 1). Figure 2 shows the drug rash observed. These individuals were then recommended to be given combination ART without 3TC and were followed up. A diagnosis of probable 3TC rash was established, as there was no further recurrence of skin rash with alternate therapy. Of the 23 patients, 3TC was substituted with TDF in 19, didanosine (ddI) in 3, and ABC in 1 individual (Table 1). Mean duration of follow-up on HAART without 3TC was 11.1 ± 12.8 (3-42) months. Mean CD4 count at 3 months of alternate therapy was 370 ± 152 (153-758) cell/mm3, showing significant improvement (
Demographic Details of HIV-Infected Individuals Diagnosed with Lamivudine Rash.
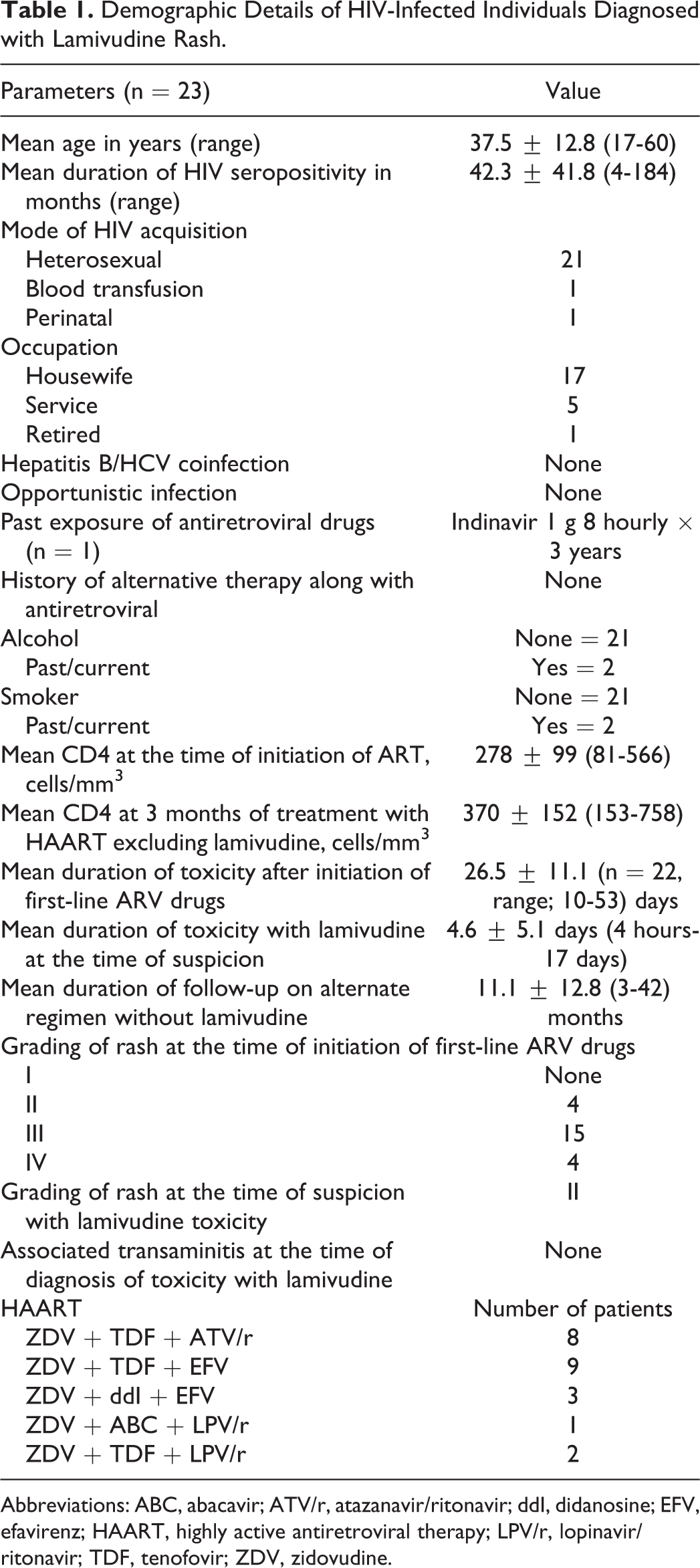
Abbreviations: ABC, abacavir; ATV/r, atazanavir/ritonavir; ddI, didanosine; EFV, efavirenz; HAART, highly active antiretroviral therapy; LPV/r, lopinavir/ritonavir; TDF, tenofovir; ZDV, zidovudine.

Algorithm for ruling out drug toxicity. Adapted from NACO Medical Officer Refresher Training Module: Drug Toxicity Case Studies. ARV, indicates antiretroviral; ART, antiretroviral therapy; ATV, atazanavir; CPT, cotrimoxazole; EFV, efavirenz; NVP, nevirapine; r, ritonavir; TDF, tenofovir; ZDV, zidovudine; 3TC, lamivudine.

Discussion
Of all ARV drugs, NNRTIs are often complicated by cutaneous drug rash, however, these seldom occur with NRTIs. 12 The incidence of occurrence of hypersensitivity reaction is 100 times more common in HIV-infected population than in general population. 7 Nevirapine is known to cause hypersensitivity rash in 15% individuals and associated transaminitis in 9% of individuals but clinical hepatitis in 1% to 2% of individuals. 13 Women are more likely than men to develop NVP rash. 14 To our best knowledge, there was not much literature reported on toxicity with 3TC except for few case reports. 15 -17
We found 23 HIV-infected individuals with 3TC-induced hypersensitivity reaction in our center, amounting to a frequency of 0.7%. The hypersensitivity reaction was of grade 1 to 2. 18 There was female preponderance. None of our patient was coinfected with HBV. Modak and Guha reported severe skin rash with 3TC in 4 HIV-infected individuals from Kolkata of whom 3 were women. 16 None of our patients suspected with 3TC rash had mucosal involvement. In 1996, Kainer and Mijch 15 had reported anaphylactoid reaction associated with 3TC. Another case report of 3TC-induced contact dermatitis has been reported by a health care professional when given 3TC along with ZDV and IDV as postexposure prophylaxis by Smith et al. 17
Generally, NNRTIs are suspected as the culprit agent when an HIV-infected individual is initiated with first-line ART (ZDV/TDF/d4T + 3TC+ NFV/EFV) and hypersensitivity reaction appears. Following recovery, in cases of grade 1 to 2 rash 18 with NFV, EFV is often used, and if it is grade 3 rash with NVP, EFV is given under close medical supervision. In grade 4 rash such as Stevens-Johnson Syndrome (SJS)/toxic epidermal necrolysis (TEN), NNRTIs are never reintroduced and alternate therapy which included PIs (ATV/r or LPV/r) along with continuation of NRTI backbone (ZDV/TDF/d4T + 3TC) is given. 2,18 Lamivudine is the drug that is present in fixed dose combination in first-line, alternate first-line, and second-line ART in national program. 2,18
Our study reported appearance of erythematous, maculopapular, pruritic, and confluent urticarial rash, typically manifesting after 10 to 53 days of initiation of first-line ARV medications. This hypersensitivity reaction was observed in average of 26.5 days of starting ART, and reexposure to ARV drug combination containing 3TC (in alternate therapy) triggered this reaction in an average of 4.6 days. A higher index of suspicion for 3TC toxicity was kept, and all drugs were stopped immediately. The reaction responded to antihistamines in all the individuals. They were later provided with the ART drug combination which included 2 NRTIs other than 3TC along with NNRTI/PI, and this in due course confirmed hypersensitivity to 3TC, since there was no recurrence of rash.
Rechallenge is contraindicated with hypersensitivity to ABC and NVP mucosal involvement, and grades 3 to 4 rash. 19 -21 Desensitization would be inappropriate for ARV drug hypersensitivity, since it would necessitate a period of subtherapeutic drug concentration and so favor the development of drug resistance. 21 In most of the individuals, the hypersensitivity reaction resolved either spontaneously or with antihistamines or oral steroids. Apprehension of developing toxicity with ARV drugs is noted among individuals whenever a regimen is modified. This apprehension further affects adherence to treatment. Three patients out of fear of drug toxicity, when restarted with ART, initially took only half of the prescribed dose.
All our 23 patients were rechallenged with 3TC after the first occurrence of rash, and rechallenge of this drug was done as 3TC-induced rash was not suspected and possibility of NNRTI/PI-induced rash was initially considered. Later a higher index of suspicion of 3TC was kept when patients presented with rash after reinitiation of alternate regimen. With repeated episodes of rash, patients were unwilling to continue the ARV combinations even when the rash was of less severity. The drugs were therefore stopped immediately. This might have been the reason for less severity of rash (maximum of grade 2 intensity) when rechallenged with 3TC (available in fixed dose combination with ZDV/d4T/TDF). We could not offer emtricitabine, another NRTI known to have equivalent efficacy as of 3TC, 22 since this drug is not available in the program, and whether there is cross-hypersensitivity to this drug is not known. Lamivudine is an integral component of HAART, and the induced M184V mutation when given with ZDV is known to reduce HIV viral fitness; increase susceptibility to ZDV, d4T, and TDF; delay accumulation of thymidine analogue mutations; and may partially reverse their effects. 23 The HAART regimens without 3TC could lead to early virological failure, and regimens with 3TC are considered to be superior to those without it. 24 It is also known in the literature that discontinuation of 3TC in patients with chronic HBV may cause flare-up of hepatitis. 25 Long-term effect of such substitution is not known. It also minimizes future drug options in case of failure. We suggest genetic studies and resistance testing in larger cohorts of patients in multicentric settings before initiating ART.
Footnotes
Acknowledgments
The authors would like to acknowledge NACO for providing antiretroviral drugs to the patients.
Authors’ Note
Ravinder Kaur Sachdeva conceptualized and wrote the manuscript and is a treating physician. Aman Sharma is a treating physician and edited the manuscript. Dipankar De is a treating dermatologist. Jasjit Malhi contributed to the supply of ART drugs to the patients and also edited the manuscript. Bharat Bhushan Rewari contributed to establishing the final diagnosis of the patients. Surjit Singh is a treating physician. Subhash Varma was involved with final editing of the manuscript and approved the work. All the participants enrolled in the study have been provided antiretroviral treatment free of cost by NACO.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
