Abstract
Objective:
To estimate health care costs associated with medical events identified as antiretroviral therapy (ART)-attributable adverse events (AEs).
Methods:
During September 2006 to June 2012, adults with ≥1 HIV International Classification of Diseases, Ninth Revision, Clinical Modification diagnosis code (042/V08), ≥1 claim for ART prescription (March 2007-June 2011; index date), and continuous health plan enrollment for ≥6 months pre- and ≥12 months postindex were included (IMS’ PharMetrics Plus Health Plan Claims Database). Patients with events of interest/ART claim during preindex period or with pregnancy/hepatitis C virus diagnosis/hepatitis B virus/cancer/tuberculosis during the study period were excluded. Postindex medical events were defined as first diagnosis code of event with ART claim ≤60 days prior to start of the event.
Results:
Differences in median total all-cause health care costs observed for diabetes/insulin resistance management (US$14 547 median all-cause health care costs during time periods identified as diabetes/insulin resistance medical events versus US$11 237 without diabetes/insulin resistance events; P = .0021), lipid disorders (US$12 825 versus US$10 033; P = .0004), and renal disorders (US$1389 versus US$0; P < .0001).
Discussion/Conclusion:
Health care costs of ART AEs should be key consideration for payers/providers in HIV management.
Introduction
HIV remains a serious health problem in the United States, with an estimated 50000 new HIV infections occurring each year. 1 In 2010, the estimated rate of diagnoses of HIV infection in the United States was 16.1/100000 population (47500 newly infected people). 2,3 Currently, there is no cure for HIV, but antiretroviral therapy (ART) has reduced the morbidity and mortality associated with HIV. 4 –6
Recommended first-line HIV treatment includes a combination of ART with 2 nucleoside reverse transcriptase inhibitors (NRTIs) plus 1 drug from another class of either nonnucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (PIs), integrase strand transfer inhibitors (INSTIs), or CCR5 antagonists, which have been shown to be effective in controlling the virus. However, HIV treatments also have the risk of adverse events (AEs) that can impact patients’ adherence to treatments, 7 thereby discontinuing treatment. 8 One study of health-related quality of life found that patients who experienced a serious AE had significantly lower scores on the Medical Outcomes Study HIV Health Survey physical and mental health summary scores, mean EuroQol (EQ)-5D, and mean Health Utility Index 3 measures compared to patients who did not experience a serious AE. 9 Al-Dakkak et al 7 performed a meta-analysis of the impact on HIV ART-related AEs and found that patients who experienced AEs had significantly lower adherence than those who did not experience treatment-related AEs. A US cohort study reported that 24% of patients discontinued their highly active antiretroviral therapy due to AEs. 10
Moreover, AEs can add significant cost to managing HIV. According to the Centers for Disease Control and Prevention, lifetime medical care for those in the United States, who become infected with HIV is estimated to cost US$20 billion each year. 11 Antiretroviral therapy AEs, in particular, can have a substantial impact on patients’ lifetime health care costs. A recent US study estimated the mean annual total expenditures per person for HIV care as US$19912 (interquartile range US$11 045-US$22 626). 12 The study did not isolate medication AEs and their contribution to those overall costs. The consideration of the health care costs of AEs is paramount in understanding the total impact on the costs of managing HIV as well as the value of ART. Yet, there is limited useful data in the literature regarding the economic burden of HIV ART AEs. The goal of this study was to leverage real-world data to estimate the economic burden of medical events (health events for which medical involvement was sought by the patient to help manage) that has been recognized to be associated with commonly used HIV third-agent medications (ie, NNRTIs, PIs, and INSTIs; comparing the costs incurred during the time periods identified in which there was medical involvement for managing events of interest and the costs incurred during time periods identified in which there was no management of events of interest).
Methods
Data Source
Data were derived from IMS Health’s PharMetrics Plus Health Plan Claims Database, which is composed of payer-sourced adjudicated claims for more than 150 million unique patients across the United States, with approximately 60 million active patients in 2012. Data are available from 2005 onward, with a typical 3- to 4-month lag due to claims adjudication.
In addition to inpatient and outpatient diagnoses and procedures and retail and mail order prescription records, PharMetrics Plus has detailed information on the pharmacy and medical benefit (copayment/coinsurance amount, deductible, in-network versus out of network), the inpatient stay (admitting versus other diagnoses, admission type and source, and discharge status), and provider details (specialty, zip code, attending, referring, rendering, prescribing, and primary care provider). Amounts charged by providers and amounts allowed and paid by health plans are available for all services rendered as well as dates of service for all claims. Other data elements include demographic variables (age, gender, and geographic region), product type (eg, Health Maintenance Organization [HMO] and Preferred Provider Organization [PPO]), payer type (eg, commercial and self-pay), and start and stop dates of health plan enrollment. All data are Health Insurance Portability and Accountability Act (HIPAA) compliant to protect patient privacy.
Patient Selection
This was a retrospective case–control database study, with all patients having at least 1 outpatient claim indicating the initiation of any US Food and Drug Administration–approved HIV drugs (ie, NNRTIs, NRTIs, PIs, fusion inhibitors, C-C chemokine receptor type 5 [CCR5] antagonist, or INSTIs; Table 1) between March 1, 2007, and June 30, 2011 (selection window). The date of the first HIV medication prescription during the selection window was defined as the index date. To be included in the cohort, patients had to be at least 18 years of age in the year in which their index date fell, had to have at least 1 clinical diagnosis for HIV (International Classification of Diseases, Ninth Revision [ICD-9] diagnosis code 042, V08) at any point in the patient’s study period (6 months preindex and/or 12 months postindex, a total of 18 months), and be continuously enrolled during the study period. Patients were excluded if they had one of the events of interest in both the pre- and the postindex periods, one or more claims for an HIV medication in the 6 months prior to their index date, or evidence of hepatitis B virus or hepatitis C virus, pregnancy, cancer, or tuberculosis in either the 6-month preindex or 12-month postindex periods (Figure 1).

Patient attrition (applied sequentially).
HIV Drugs of Interest.

Abbreviations: GPI Code, Generic Product Identifier; HCPC, Healthcare Common Procedure Coding; CCR5, C-C chemokine receptor type 5.
*A wildcard meaning any digit could be in its place (0–9).
Events of Interest
Events of interest were selected based on the Department of Health and Human Services HIV Treatment Guidelines list of common and/or severe third-agent (NNRTIs, PIs, and INSTIs) ART-associated AEs. 13 In order to assess the health care costs associated with specific events (Table 2), we constructed “episodes” for 8 events of interest (depression, dizziness, diarrhea, hepatic disorders, nausea/vomiting, rash, renal disorders, and somnolence/sleep effects) that were considered acute and tracked claims of 2 other events of interest (diabetes/insulin resistance and lipid disorders) that were considered chronic, during which health care resource utilization and costs were measured until the end of the 12-month postindex period. The first eligible mention (via ICD-9 diagnosis code) of an event in the 12-month postindex period was recorded for each patient. To better establish a link between the event and HIV medication, in order for events to be included in the analysis, patients were required to have filled a prescription for their index HIV medication within 60 days prior to the first mention of the event of interest (ie, first claim with the relevant International Classification of Diseases, Ninth Revision, Clinical Modification [ICD-9-CM] diagnosis code for an event of interest).
Selected Events of Interest and Corresponding ICD-9-CM Codes.
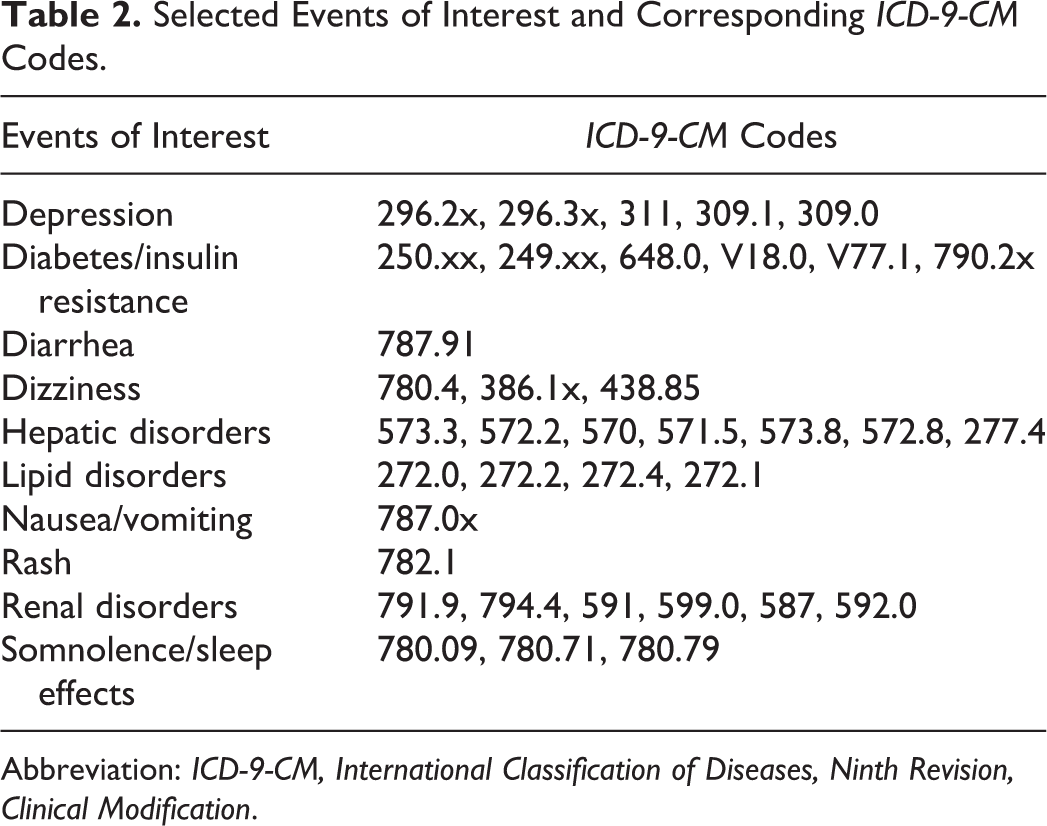
Abbreviation: ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification.
Episodes (n = 8 acute and n = 2 chronic) were propensity score matched (1:1) to nonepisodes (from patients without the event of interest). The nonepisodes had a start date that matched the start date of the event of interest. The episodes were also propensity matched on patient age (category), gender, pay type, doctor specialty (category), region, Charlson Comorbidity Index (CCI; category), preindex comorbidities, preindex medicine usage, and preindex total cost (category). All unsuccessfully matched episodes were excluded from the analyses.
The length of an episode was determined based on the service date of the first claim (denoted by the relevant ICD-9 diagnosis code of the event of interest) and the service date of the last observed medical or pharmacy claim (denoted by the relevant ICD-9 diagnosis code of the event of interest or event of interest-associated prescription claim and drug classes typically used to treat such health events). A new event was considered to have occurred if the time interval between 2 successive events of interest–associated claims was greater than 90 days. If the gap between 2 such claims was less than 90 days, the costs for that event were considered to have occurred within 1 episode. One patient may have had multiple events of interest and, therefore, for each event type, may have had multiple episodes. If there was not enough data to view the entire length of an event by the end of the postindex period, then that event was not counted. For example, if an event of interest was identified but the data were unable to allow the identification of a 90-day gap, its cost was not assessed/captured. The proportion of events censored/lost due to this requirement was reported (Table 3).
Events-of-Interest Episode Descriptive Statistics.
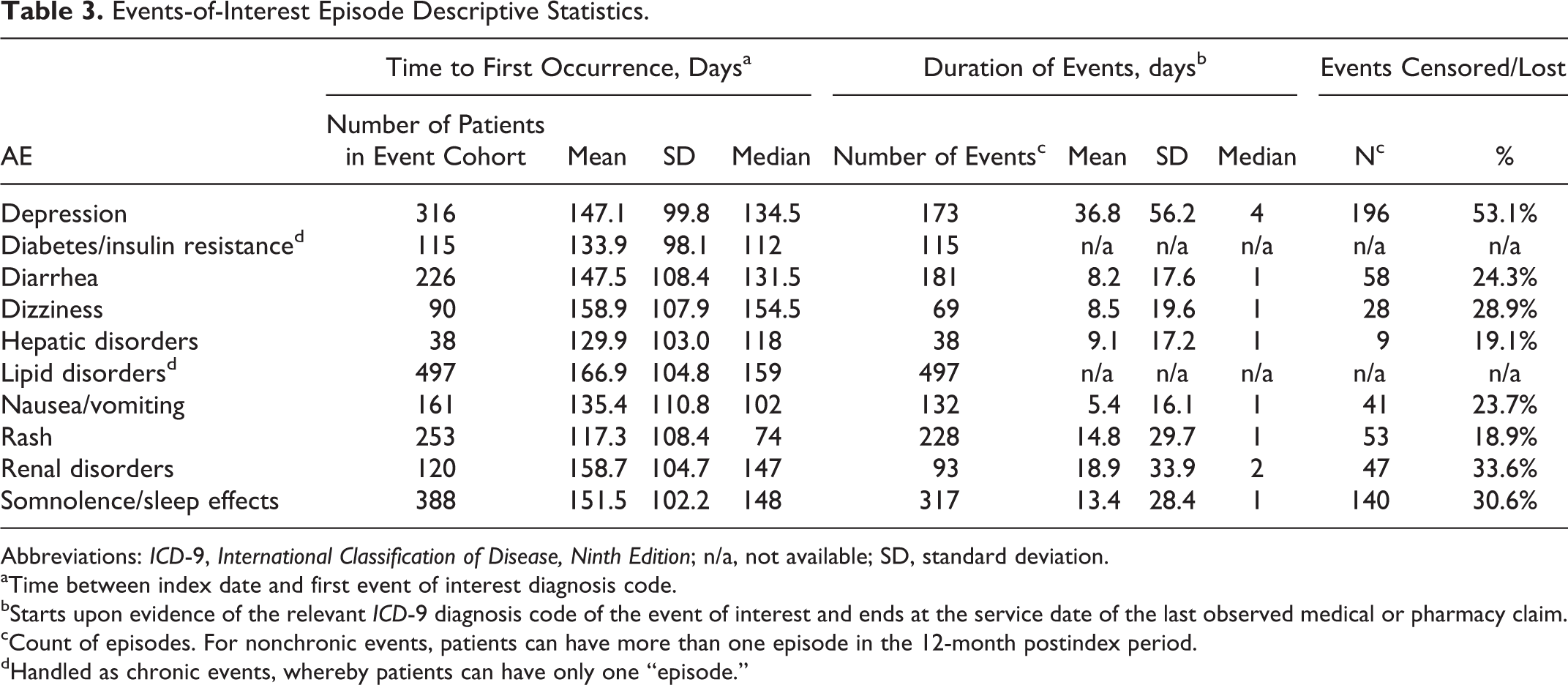
Abbreviations: ICD-9, International Classification of Disease, Ninth Edition; n/a, not available; SD, standard deviation.
aTime between index date and first event of interest diagnosis code.
bStarts upon evidence of the relevant ICD-9 diagnosis code of the event of interest and ends at the service date of the last observed medical or pharmacy claim.
cCount of episodes. For nonchronic events, patients can have more than one episode in the 12-month postindex period.
dHandled as chronic events, whereby patients can have only one “episode.”
Since diabetes and lipid disorders were considered chronic, the 90-day gap rule mentioned earlier was not applied to these events. The duration of the “episode” for these events was calculated as the time from the first claim to the end of the postindex period, and all costs were collected over that period regardless of gaps between successive AE claims. Therefore, the health care costs for diabetes and lipid disorders estimate the costs incurred within the 12 months following HIV treatment initiation.
Health Care Costs
Pharmacy costs for events were assessed during the entire time period of the event (see previous section) and included costs of ART as well as medications associated with the treatment of the AE itself (see previous section). Inpatient costs were based upon hospitalizations, with the respective ICD-9 diagnosis code reported in the claim. Similarly, ICD-9 diagnosis codes were used to calculate outpatient costs that comprised emergency department visits, physician office visits, laboratory and pathology claims, radiology examinations, and surgical and ancillary services.
Statistical Analysis
Demographic and clinical characteristics of study patients and episodes were described using frequency and percentage distributions for categorical variables and descriptive statistics (mean, standard deviation [SD], and median) for continuous and count variables. Patient demographics and AE episodes were measured as of the index date or preindex period, unless otherwise specified. For unmatched cohorts, P values using Pearson chi-square for categorical variables and Wilcoxon rank-sum for continuous variables were generated. For matched cohorts, P values using McNemar test for categorical variables and the Wilcoxon signed-rank test for continuous variables were generated to evaluate the differences in outcomes. A P value of <.05 was considered statistically significant. A generalized estimating equation model was developed for each AE episode type to describe aggregate episode-based differences in the average all-cause costs, with a gamma distribution and log-link function. All statistical analyses were conducted using SAS version 9.2 (Cary, North Carolina, USA).
Results
Demographic and Clinical Characteristics
A total of 2346 patients contributed to the overall matched population and were 41.0 years of age (SD = 9.6) on average, with the overwhelming majority (80.9%) being male. About half of the population (50.4%) resided in the southern region of the United States and 77.3% of the overall population underwent their care through a preferred provider organization. Hypertension (9.3%), vascular disease (7.3%), and anemia (6.4%) were the most common preindex comorbidities, with the mean CCI score of the overall population being 4.7 (SD = 2.6). The most commonly used drugs in the preindex period were hypertensive medications (9.2%), antidepressives (8.0%), and antianxiety medications (6.9%).
In the preindex period, the overall population had an average of US$4932.60 in total health care costs (median = US$1282.70; SD = US$16 724.50), with just a small portion of that comprising pharmacy costs (mean = US$324.30; median = US$56.20; SD = US$942.00; Table 4).
Patient Demographic and Clinical Characteristics (Postpropensity Match).

Abbreviations: COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; SD, standard deviation; USD, US dollars.
aCalculated using McNemar’s test for categorical variables and the Wilcoxon signed-rank test for continuous variables.
bPrescribing physician specialty is imputed from billing specialty of the physician claim prior to index Rx.
Adverse Event Episodes
Lipid disorders, somnolence/sleep effects, depression, and rash were the most common medical events of interest with 497, 388, 316, and 253 episodes, respectively. The average time from initiation of HIV therapy to an event of interest was shortest for rash (117.3 days, SD = 108.4) and longest for lipid disorders (166.9 days, SD = 104.8). The average duration of an event was longest for depression (36.8 days) and shortest for nausea/vomiting (5.4 days; Table 3).
Health Care Costs
Diabetes incurred the greatest average difference in health care resource use and costs among the events of interest, with an average of 34.4 more services (including pharmacy, emergency department, physician office visits, laboratory and pathology, radiology, surgical services, ancillary services, and hospital admissions) than those without diabetes and average costs totaling US$5361 more than those without diabetes (Table 5). Similarly, the greatest difference in median total all-cause health care costs was reported between those episodes with evidence of diabetes/insulin resistance when compared to those without evidence of diabetes/insulin resistance (US$14 547 versus US$11 237; P = .0021). The next largest difference in median all-cause health care costs was between patients with evidence of lipid disorders compared to those without (US$12 825 versus US$10 033; P = .0004). Similarly, episodes of a renal disorder had greater median total all-cause health care costs compared to those who did not (US$1389 versus US$0; P < .0001). The smallest increment in median total all-cause health care costs was seen between patients with and without episodes of hepatic disorders (US$212 versus US$0; P = .0005), somnolence/sleep effects (US$357 versus US$0; P < .0001), and rash (US$390 versus US$0; P < .0001; Table 6).
Differences in Average All-Cause Health Care Resource Utilization between Matched Episodes for Patients with and without Events of Interest.
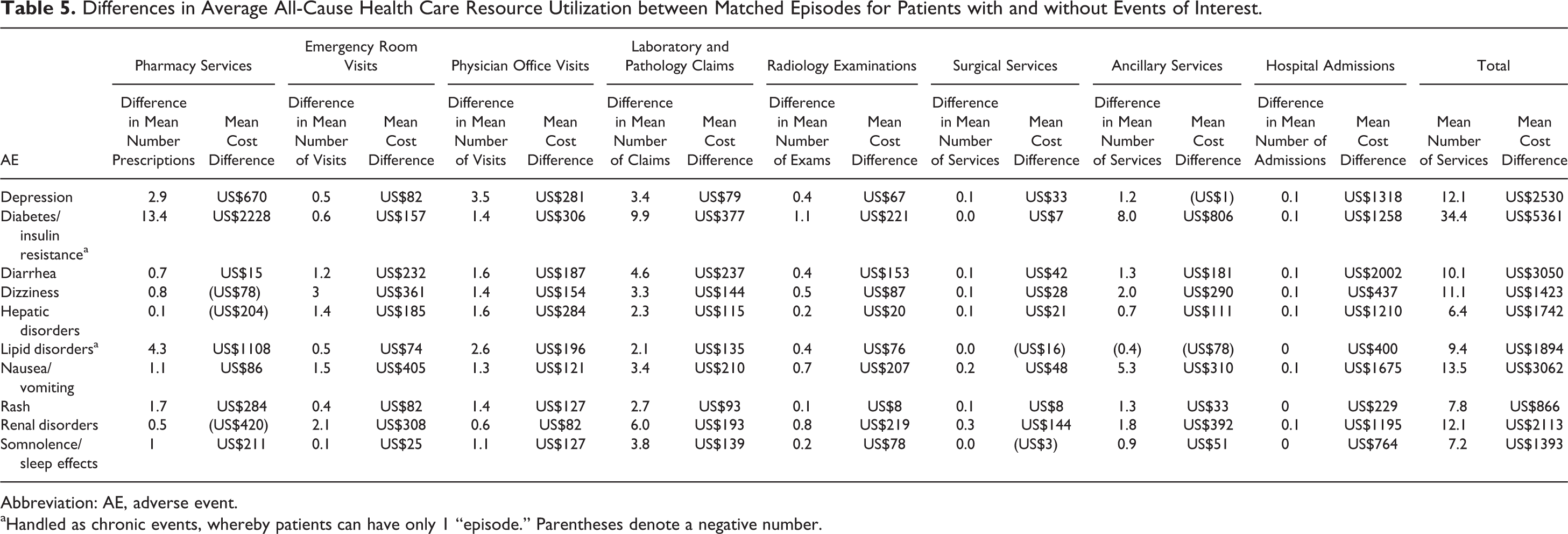
Abbreviation: AE, adverse event.
aHandled as chronic events, whereby patients can have only 1 “episode.” Parentheses denote a negative number.
Differences in Median Total All-Cause Health Care Costs among Matched Event Episodes.

Abbreviation: AE, adevrse event.
aWilcoxon signed-rank test was used to compare differences in median total all-cause health care costs.
bHandled as chronic events, whereby patients can have only 1 “episode”.
Pharmacy costs were the greatest cost component of the average total all-cause health care costs for patients with the following episodes: lipid disorders (81% cost contribution), diabetes/insulin resistance (74%), and rash (56%). Inpatient costs were the main contributor to hepatic disorders (54% cost contribution), diarrhea (54%), and nausea/vomiting (50%) episodes, while outpatient costs contributed most to dizziness (56% cost contribution) and renal disorder (43%) episodes (Figure 2).

Leading cost component contributor (average) to event-of-interest episode.
Regression Models
Episodes with evidence of nausea/vomiting had 29.4 times greater all-cause costs compared to those without evidence of nausea/vomiting (Exponential parameter estimate [Exp PE] = 29.400; P < .0001). Dizziness episodes had 22.9 times greater all-cause costs compared to those without evidence of dizziness (Exp PE = 22.895; P < .0001). Lipid disorders (Exp PE = 1.173; P = .0031) and diabetes/insulin resistance (Exp PE = 1.295; P = .0003) events both had greater all-cause costs compared to those without such events, but due to their chronic nature and longer follow-up periods their relative cost impacts were the smallest of the studied events of interest (Figure 3).

Generalized estimating equation—episode-based change in all-cause total health care costs (exponentiated parameter estimate shown; Ref grp: episodes without the event of interest).
Discussion
Our study suggests that there are significant health care costs in the United States for managing ART-related AEs. HIV exerts a significant economic burden from both a direct medical cost and productivity perspective, making research in this area of increasing interest to multiple stakeholders. 14 Reducing the incidence of treatment-related AEs will benefit patients, payers, and other stakeholders in the cost-constrained health care environment (eg, accountable care organizations and medical homes).
Prior evidence on the health care costs of specific HIV treatment-associated AEs in a real-world setting is limited. Bozzette et al 15 examined the average monthly expenditures for the care of HIV-infected patients (following patients over 36 months), but their findings were based on patient self-reported information and did not quantify expenditures by specific AEs. An international study estimated the cost of identification, reporting, treatment, and follow-up of AEs in a large HIV perinatal trial; however, this study took place in Uganda and within a clinical trial environment, making its findings difficult to compare to ours. 16 Llibre-Codina et al 17 quantified the impact of toxicity related to NRTIs on the total medical cost in HIV-1-infected patients via a pharmacoeconomic model from data obtained by a prospective, observational multicenter study in Spain. The study reported €2223 in direct and indirect costs related to AE management, primarily for the treatment of lipoatrophy, lipodystrophy, and peripheral neuropathy, the 3 AEs that were not explored in our study. A Spanish retrospective study estimated the hospital resources used to manage AEs for patients treated with fixed dose combinations compared to patients treated with combinations of separately administered antiretroviral medications. The cost of an AE during the study period was €230.26 (2011 currency value) lower than many of our study’s AE findings. However, the data were reported as a composite, not by individual AE, and were limited to the hospital setting, whereas many AEs are treated in the outpatient setting of care. 18
A recent retrospective study based upon Medicaid administrative health care claims from 15 states focused on 2 specific products of interest—atazanavir (ATV) and darunavir (DRV). Johnston et al 19 utilized an evaluation period that began on the index date (defined as the first observed prescription claim for ATV or DRV) until censoring at the earliest occurrence of either 6 months postindex or the addition of a different HIV agent. Gastrointestinal AEs had a total health care cost of US$38 ± US$387 (mean + SD) per patient per month, while diabetes and rash had costs of US$96 ± US$1253 and US$6 ± US$80, respectively. These values are lower than reported in our study, however, their calculation was not per AE episode but per patient, accounting for exposure time. Simpson et al 20 conducted a study in a commercially insured population, in which AE episodes were constructed based on medical claims with associated AE diagnosis and pharmacy claims. The reported mean total health care costs per AE episode were higher than what we found in our study, however, this is likely due to the longer AE episode duration employed. Simpson et al 20 recorded duration as whichever occurred later, the service date of last observed related medical/pharmacy claim, or the index date plus the average length of episode (based on literature review), while our approach utilized AE-associated health care claims to determine episode duration.
To aid in the assessment of the value of ART, the health care costs of AEs can be included in cost-effectiveness models. Colombo et al 21 included AE management costs in a cost-effectiveness model developed to assess the value of various ARTs in Italy, however, the model assumed a mean additional cost per year for all AEs combined. Aragao et al 22 obtained the cost of AEs from the literature in order to enhance a microsimulation discrete events model for the cost-effectiveness analysis of HIV treatment. To support Health Technology Assessments (HTA), real-world data quantifying the economic burden of AEs may play a greater role in the future development of cost-effectiveness models and related pharmacoeconomic analyses.
In conclusion, our study has highlighted the impact that ART-related AEs may have on health care utilization and costs. Clinicians, payers, patients, and other stakeholders may consider the economic burden of AEs when discussing HIV treatments.
Limitations
The results presented here must be viewed in light of some limitations associated with this study using administrative claims data. The correspondence between pharmacy submission of claims and patients’ receipt and consumption of the medication was assumed and not directly measured. Misclassification of an event of interest or HIV diagnosis may have occurred through incorrect listing in the medical claim or if the medical claim was incomplete. Additionally, events of interest were constructed based upon statistical algorithms and could not be confirmed to be actual HIV medication AEs. Although we required the start of an event to occur within 60 days of an HIV medication claim, these events of interest may have occurred for other reasons such as aging and changing guidelines. Furthermore, there is the potential for underreporting costs (based upon both procedure and diagnosis codes as well as differential recording of diagnosis codes across AEs), resulting in a potential misrepresentation of the associated direct medical costs.
The claims data set used does not include uninsured patients and those covered only by Medicare (Part B), and the source population consisted primarily of commercially insured patients in the United States, therefore, the results are most generalizable to similar commercially insured patients and may not be generalizable to other populations. The database does not provide information on systemic factors that could affect care, including plan limits on medication use and only includes the claims for services covered by/paid for by a patient’s payer. Due to the large and diverse nature of the plans in the database, however, these factors should not have impacted our study results.
Footnotes
Authors’ Note
MD, CM, SS and EM contributed to the study design and interpretation of results. MD, CM, and MM were responsible for the acquisition of data and its analysis. All authors contributed to the development of all sections of the article.
Acknowledgments
The authors thank Michael Harbour, MD for his input on the ICD-9 codes used for identifying the adverse events that were included in the analysis. This study was sponsored by Merck Sharp & Dohme Corp (Whitehouse Station, NJ, USA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EM is an employee of Merck Sharp & Dohme Corp. SS is employed as a contractor of Merck Sharp & Dohme Corp. MD, CM, and MM are employees of IMS Health who were paid consultants to Merck Sharp & Dohme Corp in connection with the conduct of the study and development of this manuscript. The authors have indicated that they have no other conflicts of interest regarding the content of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
