Abstract
Background:
HIV-related neuropathic pain (HIV-NeP) is common; however, the burden of HIV-NeP is not well-understood.
Methods:
The cross-sectional study aimed to characterize the HIV-NeP burden. A total of 103 patients with HIV-NeP recruited during routine office visits completed a questionnaire to assess patient-reported outcomes, including pain severity, health status, sleep, mood, and lost productivity. Physicians completed a 6-month retrospective chart review.
Results:
The sample was predominantly male and not employed for pay. A majority (75.7%) of patients experienced moderate or severe pain. Pain interference, general health, physical health, and depression were worse among patients with more severe pain (all Ps < .006). Most (87.4%) patients were prescribed at least 1 medication for NeP. HIV-related neuropathic pain was associated with 36.1% work impairment. Adjusted annualized costs increased with increasing pain severity (P < .0001).
Conclusion:
The impact of HIV-NeP on health status, physical function, and depression increases with severity, resulting in substantial clinical and economic burden.
Keywords
Introduction
In the United States, an estimated 1.3 million people are infected with HIV, with approximately 49 000 new infections annually. 1 With the advent and widespread use of highly active antiretroviral therapy (HAART), HIV has become a chronic condition, and the neurological complications of HIV infection have become more important to address. 2 –4 HIV-related neuropathic pain (HIV-NeP) is the most common neurologic complication of HIV infection and a major cause of morbidity in patients living with HIV; and AIDS. 5,6 Peripheral neuropathy is present in 48% of HIV-positive patients, 7 ranging from 1.2% to 69.4%. 8 HIV-related neuropathic pain may be caused by antiretroviral drug toxicity, opportunistic infections, or indirectly by the HIV virus. 9,10 The clinical presentation is similar regardless of the specific cause. 10 Although treatments are available with potentially less neurotoxicity due to reduced amounts of dose-limiting nucleoside reverse transcriptase inhibitors, such as stavudine (d4T), didanosine (ddI), and zalcitabine (ddC), HIV-NeP prevalence has not decreased in populations using these drugs. 11
In a recent US study, 55% of patients living with HIV reported pain. Of those, pain was frequent or almost constant in 67% and severe or very severe in 82%. 12 HIV-infected patients experiencing pain were significantly more likely to experience sleep disturbances, more severe fatigue, and higher levels of anxiety and depression than patients not experiencing pain. 12 The adverse effect of neuropathy from HIV/AIDS medications has been shown to negatively impact productivity in the United States. 13 Unrelieved pain is a common problem in patients living with HIV; 5,6,8,12 however, there is a paucity of published data detailing the burden associated with HIV-NeP. 12,13 The objective of this cross-sectional study was to characterize the burden of illness of HIV-NeP as it relates to patient-reported outcomes, health care resource utilization, and direct and indirect costs.
Methods
Study Design
This cross-sectional, observational study was conducted to determine patient and health care burden in HIV-NeP. Between October 2011 and May 2012, patients with HIV-NeP were recruited from 12 community-based physician practices across the United States. Practices were served by 5 neurologists, 3 infectious disease specialists, 2 general practitioners/primary care physicians, 1 pain specialist, and 1 endocrinologist. Each patient completed a self-administered questionnaire, and a 6-month retrospective review of the patient’s medical chart was also performed.
Inclusion Criteria
The 12 sites identified eligible patients when they presented for a routine medical appointment. Patients aged 18 years or older able to read and understand English were eligible to participate. Patients had to have been diagnosed with HIV-NeP and have been a patient at the practice for 6 or more months, and this allowed the capture of health care resource utilization data. For study inclusion, patients had to have had HIV and experienced symptoms of NeP for the 3 months before enrollment in the study with an HIV-NeP diagnosis confirmed by a neurologist using established diagnostic criteria. Neuropathies included distal symmetrical polyneuropathy (DSP), inflammatory demyelinating polyneuropathy, progressive polyradiculopathy, mononeuropathy multiplex, autonomic neuropathy, and diffuse infiltrative lymphocytosis syndrome.
Exclusion Criteria
Patients with medical/psychological conditions that would compromise participation or those patients who had participated in an investigational drug study in the previous 6 months were excluded.
Patients provided written informed consent, including permission to access their medical records. Central Institutional Review Board, Concordia Clinical Research (Cedar Knolls, New Jersey, USA), approved this study. Patients were recruited as part of a larger observational study of NeP burden. Data from the overall NeP sample, including the sample size and pain severity distribution of the HIV-NeP subgroup, have been published. 14,15
Data Collection
For each patient, a 6-month retrospective chart review was conducted by the physician or site coordinator to determine the date of HIV-NeP diagnosis, comorbid conditions, and HIV-NeP–related health care resource utilization.
Patients completed a self-administered, 90-item questionnaire to ascertain information about demographics and burden of HIV-NeP, including duration of symptoms, nonprescription treatments, related out-of-pocket costs in the past 4 weeks, and changes in employment status. The questionnaire also included validated patient-reported outcome measures commonly used in clinical trials of populations with NeP. Questions relating to pain severity (4 items) and pain interference with function (7 items) came from the Brief Pain Inventory–Short Form (BPI-SF). 16 The Hospital Anxiety and Depression Scale (HADS) measured both anxiety (7 items) and depression (7 items), with higher scores (range 0-21) representing poorer emotional well-being. 17,18 The impact of pain on health-related quality of life (HRQoL) was assessed using 2 separate measures: the Short Form-12v2 (SF-12v2) and EuroQol 5 dimensions, 3 levels (EQ-5D-3L). The SF-12v2 assessed physical and mental health over the past week. 19 Higher scores on the SF-12v2 (range 0-100) indicate better outcomes. 19 General health status and utility measures were evaluated using the EQ-5D-3L, with higher scores indicating better general health. 20 Sleep was assessed using the Medical Outcomes Study Sleep Scale (MOS-SS) consisting of 12 items, with the exception of sleep quantity, higher scores (range 0-100) represent more of the concept being measured. 21 Sleep quantity is the average number of hours of sleep per night in the past week. Productivity was measured with the Work Productivity and Activity Impairment-Specific Health Problem (WPAI-SHP) customized to assess overall work impairment (comprising absenteeism and presenteeism) and daily activity impairment specific to HIV-NeP. 22 The higher the scores on this 6-item measure, the greater the level of productivity impairment and decreased productivity. 22
Cost Analysis
Health care provider visits, prescription medications, portable transcutaneous electrical nerve stimulation (TENS) unit and supplies, office-based tests and procedures, emergency department (ED) visits, hospital outpatient visits, hospitalizations, and out-of-pocket costs to patients were included to calculate direct HIV-NeP–related medical costs. The 6-month costs associated with each were multiplied by 2 to calculate an annualized cost, and the 4-week out-of-pocket costs were multiplied by 13. Indirect costs included overall work impairment, disability, unemployment, early retirement, and reduced work schedule due to HIV-NeP.
To assign a per-unit cost (2012 US$) to all health care resource utilization, costing algorithms were developed. These excluded patient-reported out-of-pocket expenses because they were reported in dollar amounts. The Medicare Physician Fee Schedule for fiscal year (FY) 2012 was used to determine the unit costs for office visits, office-based procedures, and office-based tests. The Medicare Hospital Outpatient Prospective Payment System for FY 2012 was used to determine unit costs for hospital outpatient and ED visits. Hospitalizations resulting in admissions were mapped to diagnosis-related groups based on discharge diagnosis, procedures performed, and length of stay. The Medicare Hospital Inpatient Prospective Payment System for FY 2012 was used to determine unit costs for hospitalizations. The 2012 Red Book was used to determine unit cost of prescription medications based on discounted average wholesale price plus a dispensing fee. 23 The FY 2012 Medicare reimbursement schedule for durable medical equipment, prosthetics, orthotics, and supplies was used to determine the unit cost for use of a portable TENS unit and supplies.
For currently employed patients, indirect costs associated with work-related lost productivity were calculated using the Bureau of Labor Statistics average hourly wage values 24 multiplied by the overall work impairment score from the WPAI:HIV-NeP. 25 To calculate annualized costs, the weekly work-related lost productivity cost was multiplied by 52. For those patients who were unemployed, had retired early, or had a reduced workload due to HIV-NeP, indirect costs associated with lost productivity were calculated by multiplying the average hourly wage by time since change in employment status due to HIV-NeP (up to 52 weeks). For patients disabled due to HIV-NeP, the indirect costs for lost productivity were calculated using the average hourly wage plus the average monthly disability payment from the Social Security Administration applied to the time since patients were disabled due to HIV-NeP (up to 52 weeks). 26
Statistical Analyses
Summary statistics were used to describe the sample, means and standard deviations (SDs) were used for continuous variables, and frequency distributions were used for categorical variables. Scores on the BPI pain severity index were used to classify average pain severity: 0 to 3, mild pain; 4 to 6, moderate pain; and 7 to 10, severe pain. 27,28 Fisher exact test was used for categorical outcomes, and Kruskal-Wallis was used for continuous variables. Thus, while the means and SDs are presented for continuous variables by each of the pain severity groups, the P value presented is based on the ranks. Statistical significance was evaluated across pain severity groups at the .05 significance level. No adjustments for multiplicity were performed.
The association between pain severity and HIV-NeP costs within the HIV-NeP sample was examined using multiple linear regression models for cost components (direct and indirect) and total costs by pain severity. For the 3 adjusted models, forward stepwise regression was performed to select variables in the final model. The model for direct costs included adjustments for pain severity, ethnicity, time since diagnosis, and comorbidities (major depressive disorder, restless leg syndrome, cognitive dysfunction, and others). The model for indirect costs included adjustments for pain severity, age, ethnicity, employment status, time since diagnosis, and a comorbidity (chronic low back pain). The model for total costs included adjustments for pain severity, ethnicity, employment status, time since diagnosis, and comorbidities (chronic low back pain and restless legs syndrome). Analyses were performed using PC-SAS version 9.1.3 (SAS Institute, Cary, North Carolina, USA).
Results
Demographic and Clinical Characteristics
This study assessed burden of illness in 103 patients with HIV-NeP. Demographics and clinical characteristics were presented by pain severity based on the BPI pain severity scale (Table 1). Most patients experienced moderate (39.8%) or severe (35.9%) pain. Patients with mild pain made up the smallest proportion (23.3%) of the sample. One patient was excluded from the pain severity categorization because he or she did not complete all the BPI items.
Demographic and Clinical Characteristics of HIV-NeP by Average Pain Severity.a

Abbreviations: BPI-SF, Brief Pain Inventory–Short Form; GED, general educational development; HIV-NeP, HIV-related neuropathic pain; SD, standard deviation.
aMild, moderate, and severe classification was based on the BPI-SF average pain severity score. One patient did not respond to all required items needed to calculate a BPI-SF average pain severity score and thus was not included in any analysis by pain severity category. Distribution of pain severity in the HIV-NeP sample has appeared in previous publications. 14,15
bP values are from the Kruskal-Wallis test for continuous variables and the Fisher exact test for categorical variables, mild versus moderate versus severe.
cAmong patients with at least 1 comorbid condition.
The mean (SD) age of the overall HIV-NeP sample was 50.3 (9.6) years, and age increased with increasing pain severity (P < .05). The majority of patients in the overall sample were male (79.6%), and no significant differences were observed in the gender distribution by pain severity. Hispanics, who composed 18.4% of the overall study population, made up more than a quarter (29.7%) of the severe pain subgroup. The percentage of Hispanics increased with pain severity (P < .05). Patients with a high school education or less were more likely to have severe pain, while the opposite was true for patients with education beyond high school (P < .001). The percentage of disabled patients increased and the percentage of employed patients decreased with increasing pain severity.
The majority (84.5%) of patients had been diagnosed with HIV more than 2 years before the date of consent. The mean (SD) time since HIV-NeP diagnosis increased linearly as pain severity increased, though the difference was not significant. Based on available data, we estimate the vast majority of patients had DSP. Patients in the overall sample had an average of 2.8 (1.8) comorbidities. The most common was depressive symptoms, regardless of pain severity (Figure 1).
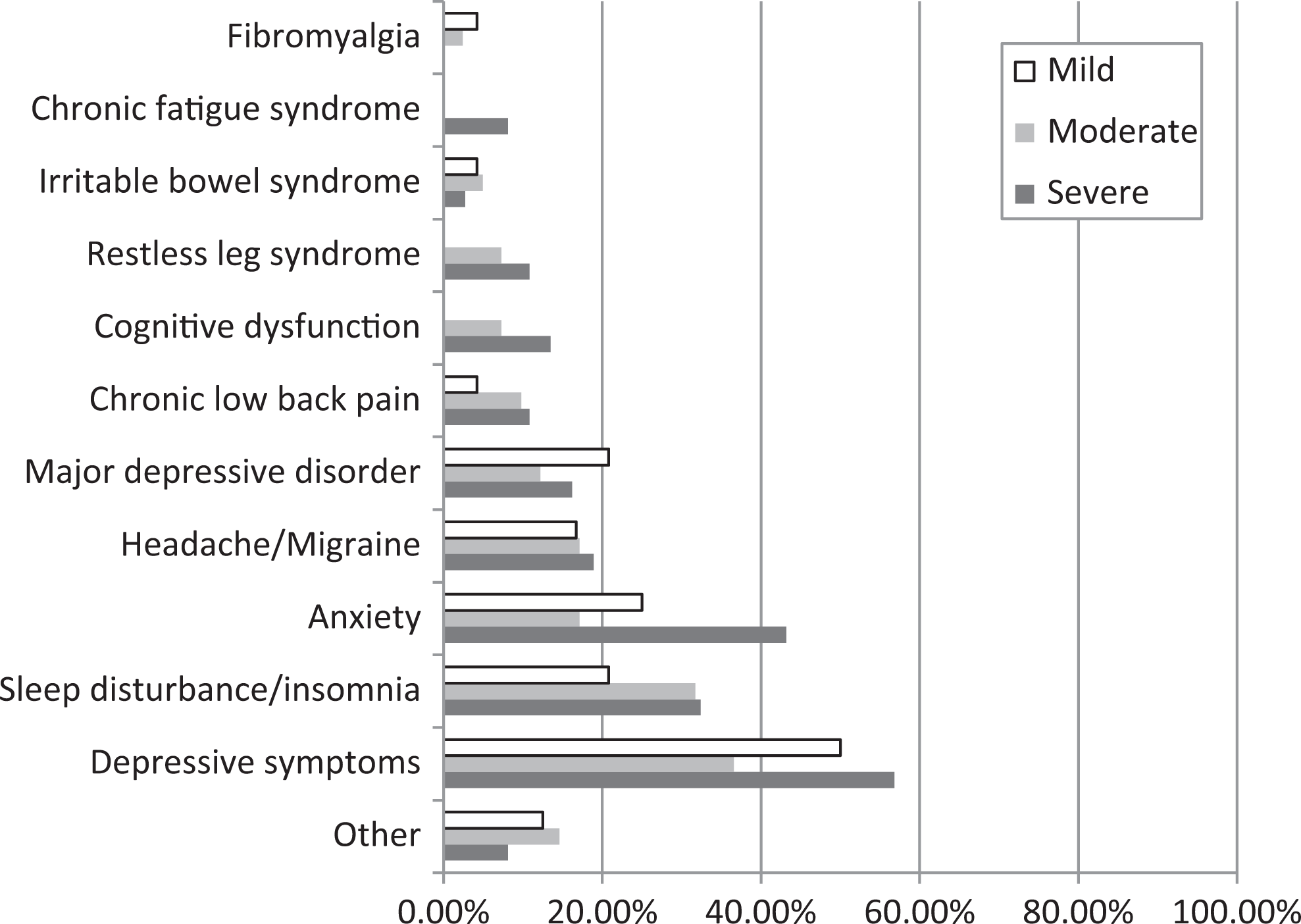
Frequency of comorbidities among patients with HIV-NeP by pain severity. A significant difference was observed across pain severity levels for anxiety (P = .0440). Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain.
Patient-Reported Outcomes
The overall mean (SD) pain severity score was 5.4 (2.5) on a 0 to 10 scale. Patient-reported outcomes, including pain severity, anxiety, and depression, by pain severity category are presented in Table 2. Across pain severity categories, a significant difference was observed for the HADS depression (P = .0050), with the highest mean score in the severe pain group (Table 2). Scores on the HADSs indicate that the majority of patients with HIV-NeP experienced some level of anxiety and depression.
Patient-Reported Outcomes: HIV-NeP by Average Pain Severity.a
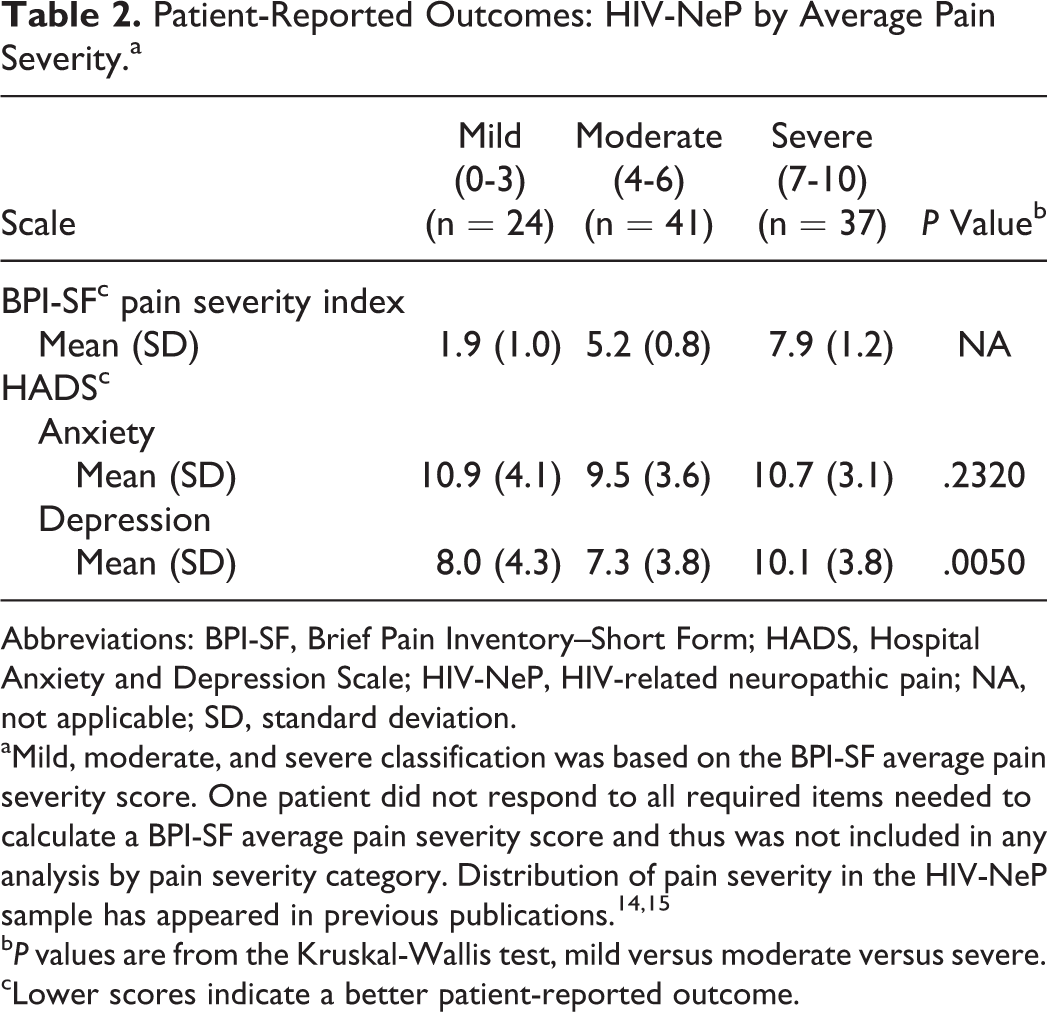
Abbreviations: BPI-SF, Brief Pain Inventory–Short Form; HADS, Hospital Anxiety and Depression Scale; HIV-NeP, HIV-related neuropathic pain; NA, not applicable; SD, standard deviation.
aMild, moderate, and severe classification was based on the BPI-SF average pain severity score. One patient did not respond to all required items needed to calculate a BPI-SF average pain severity score and thus was not included in any analysis by pain severity category. Distribution of pain severity in the HIV-NeP sample has appeared in previous publications. 14,15
bP values are from the Kruskal-Wallis test, mild versus moderate versus severe.
cLower scores indicate a better patient-reported outcome.
The mean (SD) scores for the pain interference index were higher among those with increasing pain severity (P < .0001). The components of the BPI pain interference index also increased with increasing pain severity (Figure 2; P < .0001).

Pain interference with function reported among patients with HIV-NeP by pain severity. The BPI-SF pain interference index scored on a 0 to 10 scale. A significant difference was observed across pain severity levels for BPI-SF pain interference index and all individual domains: all Ps < .0001. Note: Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain.
Mean SF-12v2 physical component summary (PCS) score, a measure of physical health status, decreased as pain severity increased (P < .0001). Mental health status, as measured by the SF-12v2 mental component summary (MCS) score, remained stable across pain severity groups. Mean PCS and MCS scores are presented in Figure 3. The mean health utility score was significantly worse among patients with greater pain severity (P < .0001; Figure 4).

Physical and mental health status among patients with HIV-NeP by pain severity. Summary scales are scored on a 0 to 100 scale (higher scores indicate better health status). A significant difference was observed across pain severity levels for the PCS: P < .0001; no significant difference for SF-12v2 MCS: P = .1154. Note: Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain; MCS, mental component summary; PCS, physical component summary; SF12v2, Short Form-12v2.

General health among patients with HIV-NeP by pain severity. EQ-5D-3L is scored on a −0.11 to 1.0 scale (higher scores indicate better health status). A significant difference was observed across pain severity levels for health state utilities: P < .0001. Note: Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; EQ-5D-3L, EuroQol 5 dimensions, 3 levels; HIV-NeP, HIV-related neuropathic pain.
Sleep, assessed by the MOS-SS, differed significantly across pain severity categories (P < .005; Figure 5), with the worst score reported by patients with severe pain.
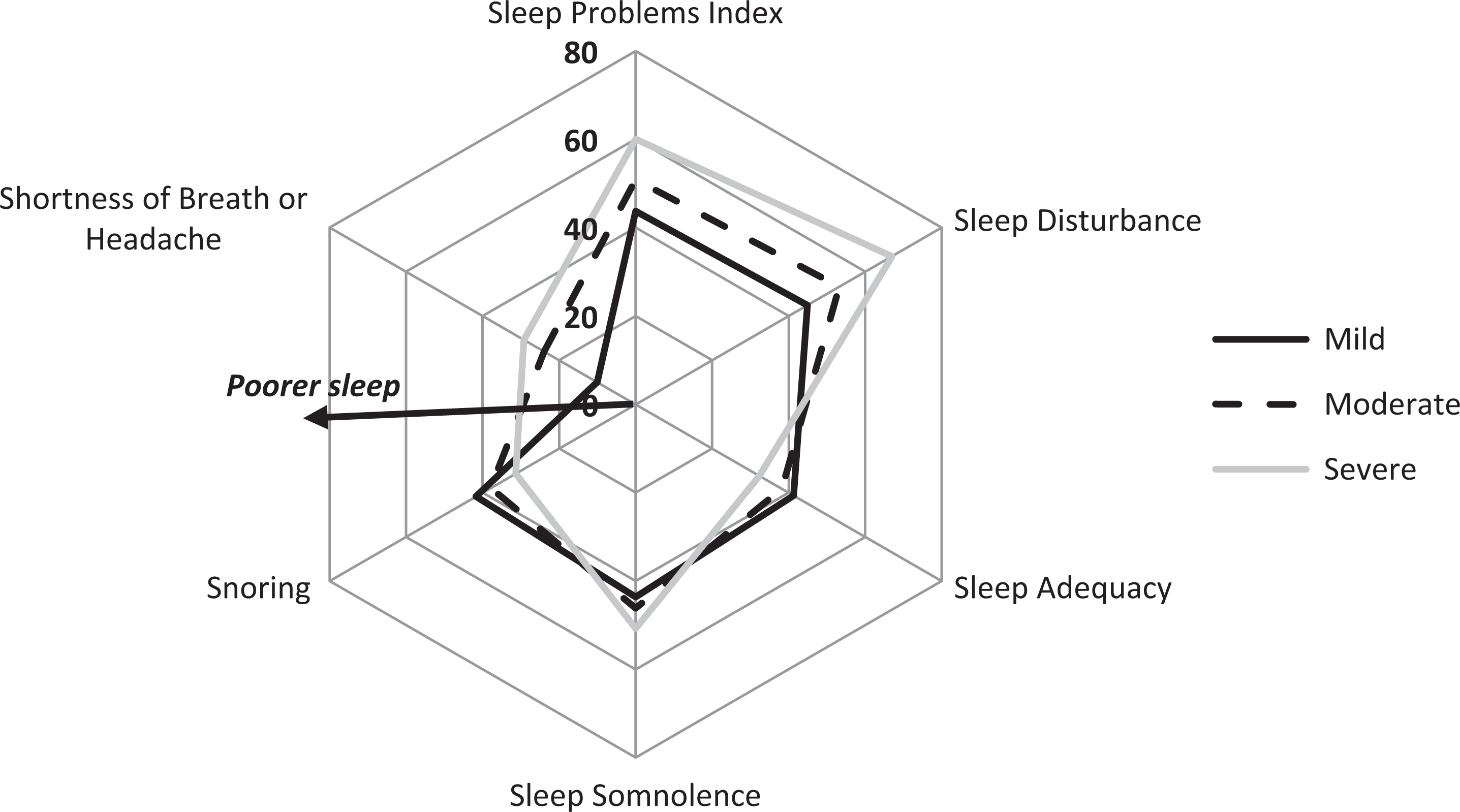
Sleep outcomes among patients with HIV-NeP by pain severity. The MOS-SS sleep problems index and the individual domains are scored on a 0 to 100 scale (higher score indicates greater sleep problems, except for sleep adequacy, where higher scores indicate better sleep outcomes). A significant difference was observed across pain severity levels for the sleep problems index, sleep disturbance, and shortness of breath or headache: all Ps < .005. Note: Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain; MOS-SS, Medical Outcomes Study Sleep Scale.
Health Resource Utilization
Most (87.4%) of the patients were prescribed 1 or more medications for the treatment of HIV-NeP in the past 6 months. No statistically significant differences were observed in health care resource use across pain severity categories (Table 3).
Resource Utilization for HIV-NeP over the Past 6 Months by Average Pain Severity.a

Abbreviations: BPI-SF, Brief Pain Inventory–Short Form; ED, emergency department; HIV-NeP, HIV-related neuropathic pain; NeP, neuropathic pain; SD, standard deviation; TENS, transcutaneous electrical nerve stimulation.
aMild, moderate, and severe classification was based on the BPI-SF average pain severity score. One patient did not respond to all required items needed to calculate a BPI-SF average pain severity score and thus was not included in any analysis by pain severity category. Distribution of pain severity in the HIV-NeP sample has appeared in previous publications. 14,15
bP values are from the Kruskal-Wallis test for continuous variables and Fisher exact test for categorical variables, mild versus moderate versus severe.
The top 2 prescription medication classes in the overall sample were antiepileptics (55.3%) and opioids (42.7%). Significant differences across pain severity categories were seen for the opioids (all) and strong short-acting opioids medication classes (Figure 6).
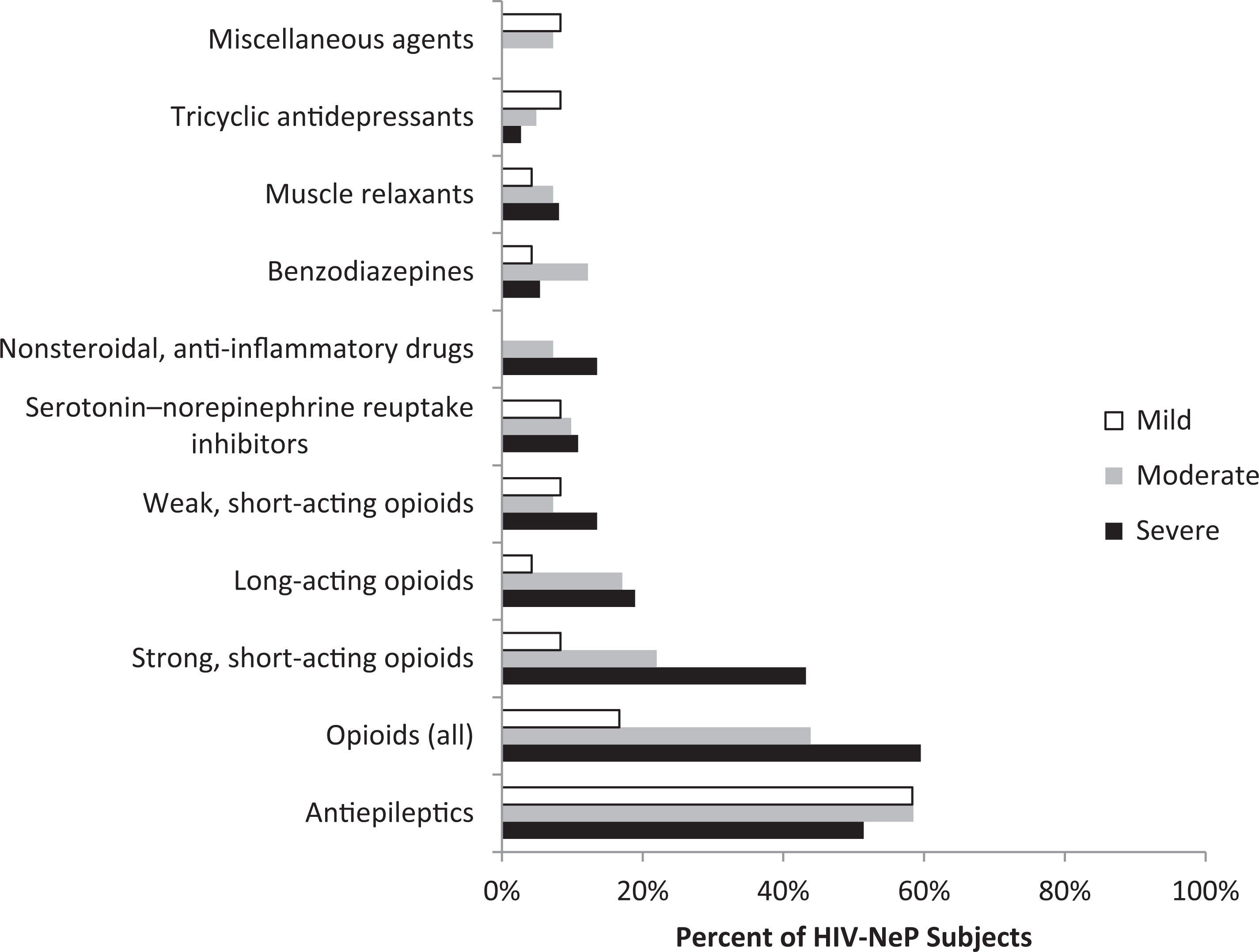
Medications prescribed for the management of HIV-NeP in the past 6 months, by average pain severity.a Figure includes all reported classes with ≥2% of patients in the sample prescribed 1 or more medications in the class. Opioids (all) include strong short-acting opioids, long-acting opioids, and weak short-acting opioids. aSignificant differences were observed across pain severity levels for opioids (all) and strong short-acting opioids (P < .007). Patients classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain.
Patients were prescribed 1.6 medications on average over the past 6 months and also reported taking an average of 0.7 nonprescription medications for their HIV-NeP over the previous 4-week period. Table 4 presents details on the number and type of patient-reported nonprescription treatments used by pain severity category.
Patient-Reported Nonprescription Treatments Used to Treat HIV-NeP by Average Pain Severity over the Past 4 Weeks.a

Abbreviations: BPI-SF, Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain; SD, standard deviation.
aMild, moderate, and severe classification was based on the BPI-SF average pain severity score. One patient did not respond to all required items needed to calculate a BPI-SF average pain severity score and thus was not included in any analysis by pain severity category. Distribution of pain severity in the HIV-NeP sample has appeared in previous publications. 14,15
bP values are from the Kruskal-Wallis test for continuous variables and the Fisher exact test for categorical variables, mild versus moderate versus severe.
cAs more than 1 response may be selected, the sum of percentages across response options may exceed 100.
The mean number of nonprescription treatments did not differ significantly across pain severity groups. Vitamins and ibuprofen were commonly taken.
Similar to medication utilization, the use of office visits, tests, and procedures, and other health resources did not differ significantly by pain severity (Table 3). There was 1 hospitalization, 2 hospital outpatient visits by 1 patient, and 4 ED visits by 4 patients for HIV-NeP reported in the 6 months before consent.
Lost Productivity
Work productivity was assessed to understand the indirect costs of HIV-NeP, with higher values indicating greater impairment. Overall, the proportion of patients employed for pay declined with increasing pain severity (P < .05; Table 5).
Work Productivity and Activity Impairment: HIV-NeP by Average Pain Severity.a
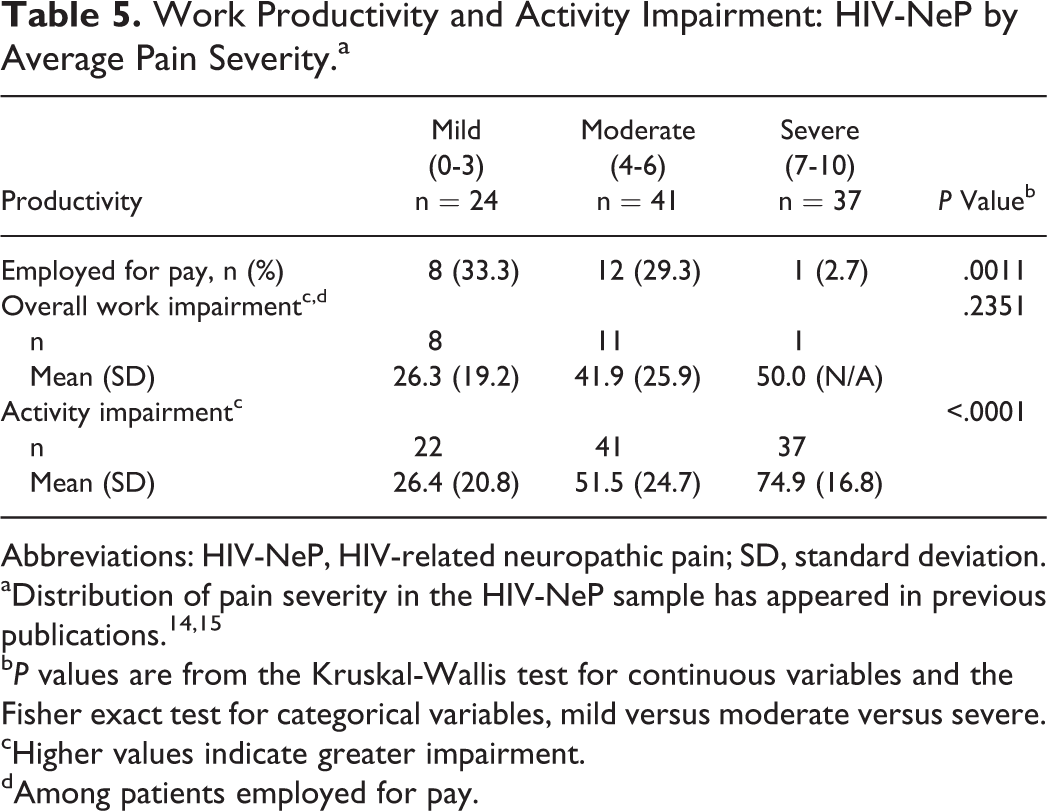
Abbreviations: HIV-NeP, HIV-related neuropathic pain; SD, standard deviation.
bP values are from the Kruskal-Wallis test for continuous variables and the Fisher exact test for categorical variables, mild versus moderate versus severe.
cHigher values indicate greater impairment.
dAmong patients employed for pay.
Among those employed for pay, patients reported an average of 36.1% overall work impairment due to HIV-NeP. Mean activity impairment increased with increasing pain (P < .0001).
The majority of patients with HIV-NeP did not alter their employment status (mild: 79.2%, moderate: 63.4%, and severe: 59.5%); however, 28 patients reported being disabled and 4 reduced their work hours (2 with mild pain and 2 with moderate pain) due to HIV-NeP. Of the patients disabled due to HIV-NeP, 40.5% were categorized with severe pain compared with 8.3% with mild pain and 26.8% with moderate pain. Six of 28 patients disabled due to HIV-NeP reported having less comprehensive health insurance coverage on medical disability as a result of the change in their employment status. Of the 4 patients who had reduced employment hours due to HIV-NeP, 2 (both with mild pain) reported having less comprehensive health insurance coverage as a result of the change in their employment status.
Costs
Total (direct and indirect) unadjusted annualized costs due to HIV-NeP were US$9 990, US$22 250, and US$25 822 per mild, moderate, and severe patient, respectively (P = .0476), with approximately 75% due to indirect costs in the moderate and severe cohorts. Although total unadjusted costs ranged widely between the mild and severe pain cohorts, no significant differences were observed across pain severity levels for total direct costs (P = .2037). Total indirect costs increased as pain severity increased (P = .0507). Significant differences were observed across the 3 groups for total costs (P = .0476). Changes in employment status due to HIV-NeP-related disability were the primary driver of indirect costs across pain severity cohorts.
After adjustment, total annualized direct costs for patients with HIV-NeP in the overall sample averaged US$5288 per patient with mild pain, US$3944 per patient with moderate pain, and US$6861 per patient with severe pain. Total annualized adjusted indirect costs averaged US$4213 per patient with mild pain, US$16 188 per patient with moderate pain, and US$23 014 per patient with severe pain. The models for annualized adjusted direct, indirect, and total costs were statistically significant (P < .0001), and pairwise significant differences were observed for direct costs (moderate versus severe patients), indirect costs (mild versus moderate and mild versus severe patients), and total costs (mild versus severe patients). Total adjusted annualized costs are presented by pain severity in Figure 7.

Annual adjusted cost per patient with HIV-NeP by pain severity. Annual adjusted cost values are least squares mean estimates from multiple linear regression adjusted for confounding demographic and clinical variables; specifically, pain severity, ethnicity, employment status, time since diagnosis, and comorbidities (chronic low back pain and restless leg syndrome). The model was statistically significant (P < .0001), and significant difference was observed for mild versus severe pain severity levels for total costs. Patients were classified by BPI-SF average pain severity score (mild = 0-3, moderate = 4-6, and severe = 7-10). One patient did not respond to all required items needed to calculate an average pain severity score and thus was not included in any analysis by pain severity category. BPI-SF indicates Brief Pain Inventory–Short Form; HIV-NeP, HIV-related neuropathic pain.
Discussion
In this cross-sectional analysis of HIV-NeP, we have, for the first time in the United States, comprehensively evaluated the burden of illness (ie, quality of life, health care resource utilization, lost productivity, and costs) among adults with HIV-NeP. We found association between pain severity and patient-reported health and well-being. Health status decreased as pain severity increased, as shown by both the SF-12v2 PCS and EQ-5D-3L. Sleep, as measured by the MOS-SS sleep scale, also worsened with increasing pain severity. Patients with HIV-NeP experienced substantially lower health status than the general US population, as reflected by MCS, PCS, and general health status scores (EQ-5D-3L utility), compared to the respective US norms of 49.5, 49.7, and 0.87. 19,29 Activity impairment was also substantial and increased with increasing pain severity, as shown by the BPI-SF pain interference index and WPAI:HIV-NeP activity impairment scores.
In addition to patient-reported health and well-being, this study also provided a comprehensive evaluation of the direct and indirect costs of HIV-NeP, by severity level, in the United States. Patients had notable levels of work impairment due to HIV-NeP, and a third or less were employed for pay across pain severity levels. Total annualized costs due to HIV-NeP were high, particularly for those with moderate or severe pain. Indirect costs were driven by change in employment status due to HIV-NeP–related disability. These results point to potential economic benefits to patients and employers of improved HIV-NeP treatment.
The patient-reported outcomes were assessed using multiple measures to ensure patient burden was adequately assessed. This is important in light of a recent analysis of a self-assessment of treatment questionnaire in patients with NeP, including HIV-NeP, showing that the activity level and quality of life sections were better assessed when they were divided into 3 concepts each rather than assessed as 1 overarching concept. 30 Because patients in the aforementioned analysis felt “activity level” was “too broad and unspecific,” it was divided into self-care, daily activities, and physical activities. Similarly, changes in quality of life were interpreted as unspecific by patients, and the question was divided into sleep, emotions, and social function. The scales used within this study assess these domains separately.
The current study sample is generally consistent with the samples in published studies of pain in HIV, HIV-NeP, and other types of NeP in the United States and Europe, with regard to age, gender, proportion of patients employed, and high proportion of patients with moderate or severe pain. 31 –34 In a US longitudinal study of HIV-infected adults, 82.0% of patients with pain reported experiencing severe or very severe pain using the Memorial Symptom Assessment Scale 12 compared with 75.7% of patients who reported experiencing moderate to severe pain in this study. Patients living with HIV experiencing pain were more likely than patients living with HIV and not experiencing pain to experience sleep disturbances, higher levels of anxiety, more severe fatigue, and higher depression scores. 12 Our results were similar in that, except for anxiety and depression, those same factors worsened significantly with increasing pain severity. The impact of neuropathy is similar to that noted in diabetic patients with painful diabetic peripheral neuropathy (pDPN), where pDPN has been associated with sleep disturbances, decreased enjoyment of life, higher prevalence of depression, and impairment of physical and mental functioning and work productivity. 34 –36
Given the improved prognosis of patients living with HIV and treated with HAART, quality of life of patients and the adverse effects of HAART have become important considerations. In a study of the impact of adverse effects of HIV medications, neuropathy was present in over half of patients. 13 The adverse effect of neuropathy from HIV/AIDS medications was associated with changes in health status, absenteeism, presenteeism, overall work impairment, and activity impairment similar to the profound effect of neuropathy on productivity found in this analysis. 13 It is important to note that quality of life appears to be related to mental state as well as pain severity. In patients with HIV-associated distal NeP, Keltner et al 37 found that severity of depression correlated more closely with HRQoL than with pain intensity. Based on the evidence presented here and in similar studies, 8,12,37 it appears that patients with HIV-NeP are worse off than patients living with HIV alone.
Limitations
Several limitations warrant discussion. Selection bias may be inherent in this type of analysis because patients were enrolled in the study only during a routine medical appointment, which captures those actively seeking medical care for HIV-NeP. Patients had to have been managed at the physicians’ practice and diagnosed with HIV-NeP for 6 or more months prior to enrollment. Therefore, findings may not apply to patients with HIV-NeP who do not regularly visit their physician to seek treatment.
Additionally, this study required a retrospective review of medical records, which could lead to underreporting of health care resource utilization. The patients’ medical records may not have included all visits to other physicians, health care providers, or facilities, as well as HIV-NeP–related tests and procedures conducted, and medications prescribed outside the study site. To address this limitation, physicians and site coordinators were asked to review the health care resource utilization with patients during their appointment.
Actual costs to the payer may be higher or lower than reported here, given that costs were assigned using standard algorithms. Similarly, the indirect costs based on average wage data may overestimate or underestimate actual costs to employers and society. Finally, patient-reported out-of-pocket costs may be subject to recall bias; however, the 4-week recall period was selected in order to address this potential bias.
Conclusion
Unrelieved pain is a significant problem in patients living with HIV; however, published data detailing the burden associated with HIV-NeP are limited. Despite patients being treated by their physicians for HIV-NeP, this study found that patients experience moderate and severe pain, poor to moderate overall health and sleep, and moderate anxiety and depression. The direct and indirect costs of HIV-NeP are substantial, particularly for those with moderate or severe pain. These data suggest the need for a more thorough communication of symptoms and outcomes of currently used treatment between the patient and physician.
Footnotes
Acknowledgments
The authors thank Gergana Zlateva, PhD, an employee of Pfizer Inc, and Felicia Bergstrom, MSPH, and Shoshana Daniel, PhD, employees of Covance Market Access Services Inc, for their contributions to the study design and analysis. We thank Karen Spach, PhD, an employee of Covance Market Access Services Inc, who was a paid consultant to Pfizer for the development of the manuscript, for her editorial contribution to this manuscript.
Authors’ Note
AS, CS, RM, BP, BS, MT, and EN made substantial contributions to study conception and design. SN was an investigator in the study, making substantial contributions to the acquisition of data. CS and RM contributed to study implementation and the acquisition of data. AS, CS, RM, and RB made substantial contributions to data analysis. AS, CS, RM, BP, SN, BS, MT, and EN made substantial contributions to the interpretation of data and results. CS, RM, and RB drafted and revised the manuscript. AS, BP, SN, BS, MT, and EN critically reviewed and revised the manuscript. All authors read and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Rachael Mann, Caroline Schaefer, and Rebecca Baik are employees of Covance Market Access Services Inc, who were paid consultants to Pfizer Inc, in the development and execution of this study, analysis, and manuscript. Dr Srivinas Nalamachu was a paid investigator for the study but was not financially compensated for his collaborative efforts to publication-related activities. Drs Brett Stacey, Edward Nieshoff, and Michael Tuchman were not financially compensated for their collaborative efforts on this project, including publication-related activities.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Pfizer Inc. Alesia Sadosky and Bruce Parsons are paid employees of Pfizer Inc.
