Abstract
Background:
Lipodystrophy is a known adverse effect of highly active antiretroviral therapy (HAART). Lipodystrophy resulting in body dysmorphism can lower the quality of life (QoL) among HAART recipients. The main aim of our study was to find the effect of lipodystrophy on QoL among people living with HIV (PLHIV) on HAART.
Methods:
This cross-sectional study was conducted in a tertiary care hospital in south India. The participants were assessed for the presence of lipodystrophy. Their QoL was assessed using HIV-AIDS-targeted QoL questionnaire (HAT-QoL). Statistical analysis was carried out using SPSS version 11.5.
Results:
Lipodystrophy was present in 21 participants (42%). The QoL among individuals with lipodystrophy was found to be significantly lower in terms of disclosure worries (P = .023) and financial worries (P = .049).
Conclusions:
Lipodystrophy adversely affects QoL among PLHIV. There is a need for studies analyzing factors that can potentially improve the QoL in such individuals.
Introduction
Highly active antiretroviral therapy (HAART) has reduced HIV-related morbidity and mortality due to benefits of early initiation of antiretroviral therapy. 1,2 The HAART has thus led to a discussion about HIV/AIDS being a chronic manageable illness. 3 With increased longevity among people living with HIV (PLHIV), their quality of life (QoL) has become an important focus for researchers. 4 World health organization defines QoL as “individuals' perceptions of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns.” 5
Drugs used to treat HIV cause a constellation of body fat composition and metabolic alterations, which have been collectively grouped under the term lipodystrophy. 6 These alterations include presence of lipoatrophy, that is, fat wasting in the face, arms, legs, and buttocks or lipohypertrophy, that is fat accumulation in the abdomen, breasts, or dorsocervical region or the presence of lipomata. 7 The increased prevalence of lipodystrophy has been linked to prolonged duration of antiretroviral therapy. 8,9 Nevertheless, survival benefits of antiretroviral therapy outweigh the adverse effect of lipodystrophy, 10 resulting in lifelong use of antiretroviral drugs. However, body changes due to lipodystrophy may stigmatize patients, causing low self-esteem, problems in social and sexual relations, anxiety, and depression. 11 This leads to a lowered QoL. The main aim of our study was to compare the QoL among PLHIV on HAART with and without lipodystrophy.
Methods
Selection and Description of Participants
The present cross-sectional study was conducted after approval by the institutional ethics committee of Kasturba Medical College, Mangalore. The study population comprised PLHIV, between the age of 18 to 60 years who were undergoing treatment at a tertiary level teaching hospital in coastal South India. The sample size was calculated based on the expected proportion of PLHIV on HAART having lipodystrophy, that is, 61% according to a study conducted in South India. 12 With absolute precision of 15% and confidence interval of 95%, sample size was calculated to be 41 PLHIV. Upon adding 20% as nonresponse, the sample size was found to be 50 PLHIV. Clinically stable PLHIV with a CD4 count of more than 200 cells/mm3 were then enrolled in the study based on the inclusion criteria of being recipients of HAART for a minimum of 1 year with over 95% adherence to HAART. Data were collected using a pretested semistructured questionnaire and the socioeconomic status of the participants was assessed using modified Kuppuswamy socioeconomic status classification, which included the educational status, occupation, and income of the respondent. 13 The treatment regimen followed by each participant was recorded along with the most recent CD4 count.
Assessment Tools
Lipodystrophy
Lipodystrophy was defined both by physician assessment (objective) and patient report (subjective) based on the recommendations by Lichtenstein et al. 14 Face, arms, and legs were assessed for thinning and the abdomen and dorsocervical region were assessed for fat accumulation. The changes were graded as “subtle (noticeable only if specifically looked for, no change in clothing fit), moderate (easily noted by patient or physician, clothing has become tight or loose), or severe (obvious to the casual observer, has required a change in clothing size).” 14 Final severity in each of the above mentioned body regions was adjudged based on physician and patient agreement. Severe change in any 1 body region and moderate changes in >2 body regions amounted to lipodystrophy.
QoL assessment
The QoL was assessed using the HIV-AIDS-targeted QoL questionnaire (HAT-QoL). 15,16 It consisted of 34 items and assesses 9 dimensions of QoL. The HAT-QoL captures information on overall function (6 items), sexual function (2 items), health worries (4 items), medication worries (5 items), disclosure worries (5 items), financial worries (3 items), HIV mastery (2 items), life satisfaction (4 items), and provider trust (3 items) over the previous 4 weeks. All items had the same 5-point Likert-type scale response options. The score for each dimension was transformed to a scale from 0 to 100, with higher scores indicating better dimension-specific QoL.
Anthropometry 17
Olecranon process of the ulna and acromion of the scapula were identified. At the midpoint of an imaginary line joining these 2 bony landmarks, mid-arm circumference (MAC) was measured using a nonstretchable measuring tape and triceps skinfold thickness (TSFT) was measured by a skin pinch using a Lange skinfold calipers (Power Systems, Tennessee).
Data Analysis
Data collected was entered into SPSS Version 11.5 and statistically analyzed in 2 groups based on the presence of lipodystrophy. Chi-square test was done to compare the qualitative variables and student t test was used to compare the quantitative variables. A P value <.05 was taken to be statistically significant.
Results
The study participants included 50 PLHIV who met the inclusion criteria and signed a written informed consent. Out of this, 37 (74%) were males and 13 (26%)were females. Seventy percent belonged to the class IV (upper lower) of the modified Kuppuswamy socioeconomic scale. Fifty-eight percent of our study participants were married (Table 1). Majority of the participants (94%) were on a combination of 3 drugs that consisted of 2 NRTI and 1 NNRTI (Table 2).
Sociodemographic Characteristics of the Study Population (N = 50).

Comparison of Treatment Regimen.
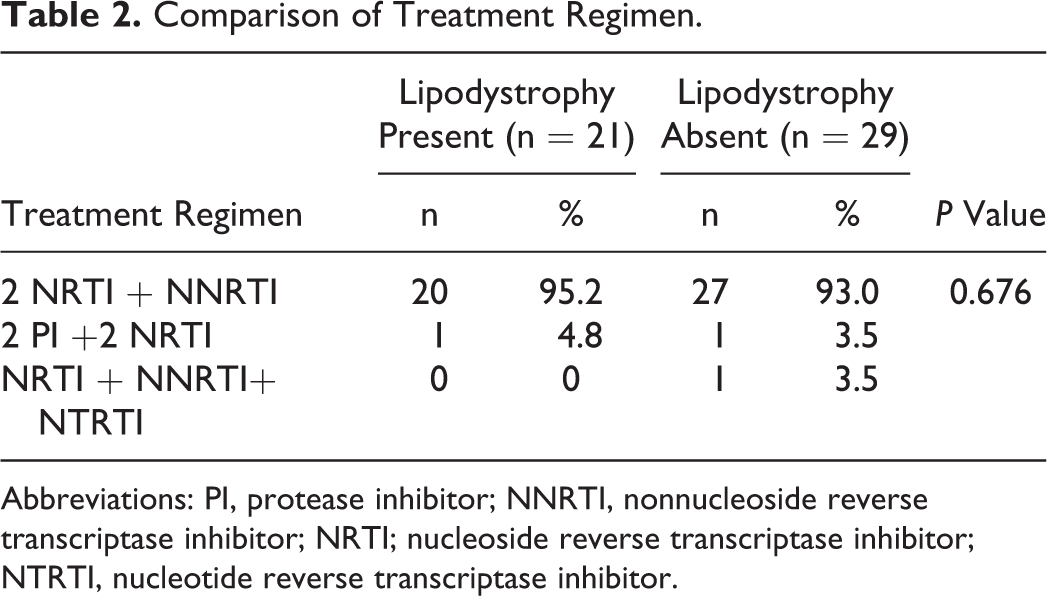
Abbreviations: PI, protease inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; NRTI; nucleoside reverse transcriptase inhibitor; NTRTI, nucleotide reverse transcriptase inhibitor.
Lipodystrophy was recorded to be present in 21 participants (42%) and absent in 29 participants (58%). All the 21 participants with lipodystrophy had facial lipoatrophy. The mean (standard deviation [SD]) HAART duration (years) among PLHIV with and without lipodystrophy was 3.71 (2.542) and 3.58 (2.161), which was not found to be statistically significant (P = .840). The mean (SD) CD4 count (cells/mm3) among participants with and without lipodystrophy was 552.95 (155.89) and 474.59 (209.75) which was again not statistically significant (P = .155). Mean (SD) TSFT (mm) was 7.10 (2.791) in participants with lipodystrophy and 9.86 (3.399) in participants without lipodystrophy, which was found to be statistically significant (Table 3). People living with HIV without lipodystrophy had higher QoL scores than those with lipodystrophy on most components of HAT-QoL. However, statistically significant lowering of QoL was established in disclosure and financial worries. There was an excellent provider trust seen in both groups (Table 4).
Anthropometry Assessment.

Comparison of QoL among PLHIV With and Without Lipodystrophy.
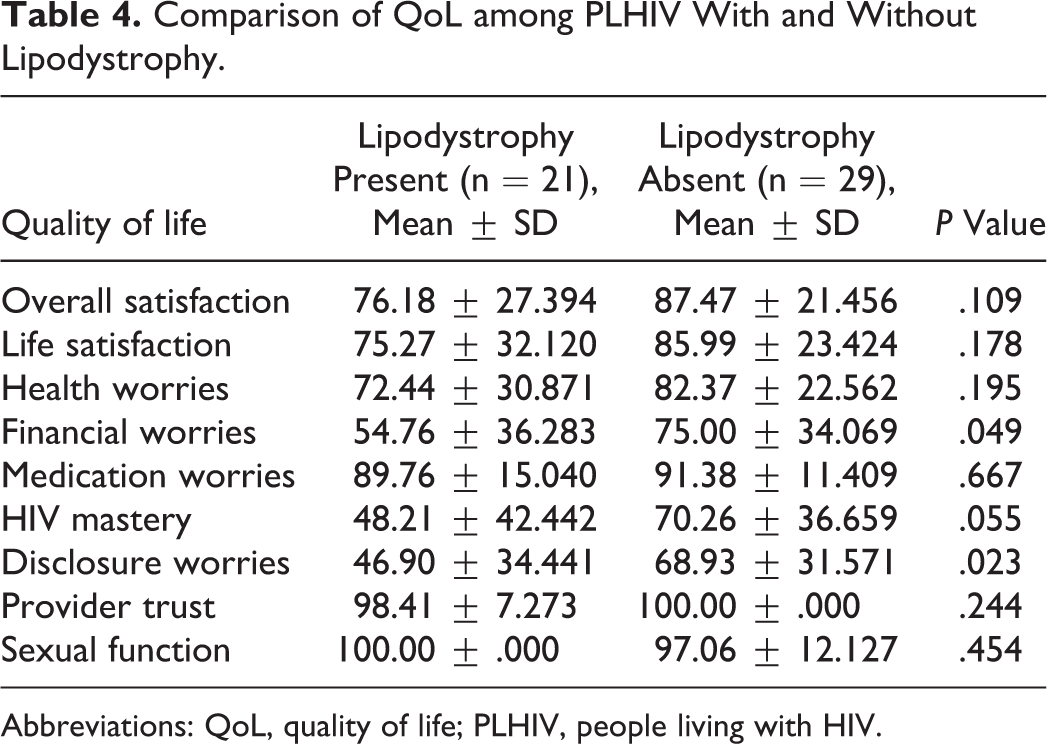
Abbreviations: QoL, quality of life; PLHIV, people living with HIV.
Discussion
Our study included 50 PLHIV out of which 21 had clinically assessed lipodystrophy. The TSFT measurement revealed significantly lower values among PLHIV with lipodystrophy. This is in concurrence with an earlier report published by Gerrior et al, 18 substantiating the use of TSFT for objective recognition of lipodystrophy.
The QoL among PLHIV with lipodystrophy was found to be significantly lower in terms of disclosure worries and financial worries that were statistically significant. Despite being visible on the arms, legs, and buttocks, lipodystrophy is more apparent on the face. 19 The appearance of lipoatrophic features on the face can be a persistent reminder of the illness. 20 Therefore, it leads to worries that the illness that is kept a secret by the patient will become public. 21 The lipodystrophic features can also lead to a fear of persistent questioning by the society and that further increases the disclosure worries. 20 Patients with facial lipoatrophy have no way of keeping their condition under control. Patients may also become socially isolated, 22 thus leading to loss of livelihood resulting in financial worries. A study conducted in Puducherry, India, has reported a decrease in income following the diagnosis of HIV infection among PLHIV, which has been attributed to disease manifestations, decreased working capacity, and loss of work days. 23 Lipodystrophy resulting in disclosure of disease status and stigma can add to this existing financial problem.
In our study, though all the other components of QoL had lower values among PLHIV with lipodystrophy, the difference was not found to be statistically significant. The reason may be the excellent provider trust seen in our study participants.
A study conducted by Rajagopalan et al 24 demonstrated that lipodystrophy further decreases the already lowered QoL among PLHIV. On similar lines, Nicholas et al 25 have also shown that lipodystrophy lowers QoL. People living with HIV with lipodsytrophy have reported its negative impact on health-related QoL with respect to self-esteem, social contacts, sexuality, and daily activities. 26
A worrisome consequence of lipodystrophy is the stoppage of treatment by patients. 27 Cabrero et al 28 have reported a high prevalence of lipodystrophy, with its negative impact on PLHIV suggesting the need to tackle lipodystrophy to ensure patient well-being.
Studies from India have shown that QoL among PLHIV is low. 29,30 However, there is no enough literature about the effects of lipodystrophy on the QoL among PLHIV on HAART from India. In other parts of the world, as studies recommend techniques to deal with lipodystrophy, 31 our study aims to throw some light on the gravity of the problem in India. As treatment regimens minimizing lipodystrophy are planned, reports indicate that patient-perceived morphological changes may take several years for reversal/improvement. 32 Therefore, it is imperative to study factors improving QoL in PLHIV with lipodystrophy alongside implementation of alterations in therapy.
Our study had some limitations. Our sample size was small. Additional studies by using a larger number of patients should be performed. Second, lipodystrophy was defined clinically, and tests like dual X-ray absorptiometry (DEXA) were not used to assess lipodystrophy in our study. Finally, results of our study may not be generalized to other HIV-infected populations as our population was predominantly urban.
Conclusion
People living with HIV in general have a low QoL and lipodystrophy further affects it. With patients living longer on HAART, prevalence of lipodystrophy has increased. Negative impact of lipodystrophy on QoL may lead to reduced adherence, and hence HAART program in our country may fail. Therefore, there is a need to study factors that can improve QoL even in the presence of lipodystrophy alongside the study of treatment regimens that can reduce the incidence of lipodystrophy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Indian Council of Medical Research (ICMR)–Short Term Studentship Sanction no.21/125/08—BMS.
