Abstract
Eligibility criteria were (1) having previously failed first-line nonnucleoside reverse transcriptase inhibitor–based regimens and (2) having achieved virologic suppression >6 months while receiving a protease inhibitor (PI)-based regimen as second-line treatment. Eligible participants were randomized to receive either (1) ritonavir-boosted lopinavir (LPV/r) monotherapy (n = 29) or (2) LPV/r with optimized background regimens (OBRs; n = 31). Median duration of viral suppression before randomization was 45 months. At week 48, viral suppression during LPV/r monotherapy was 86.2% and did not differ from the suppression achieved with LPV/r with OBRs (87.1%, P = 1.000). However, persistent viremia during LPV/r monotherapy tended to be higher than during LPV/r with OBRs (10.3% versus 3.2%, P = .346). History of viral blip during virologic suppression with second-line PI-based regimen is a predictor of achieving viral suppression at all visits (adjusted relative risk 0.255 [95% confidence interval 0.080-0.821], P = .022). Use of LPV/r monotherapy as maintenance regimen in this study produced persistent viremia that tended to be higher than LPV/r monotherapy with OBRs.
Introduction
Nonnucleoside analog reverse transcriptase inhibitor (NNRTI)-based regimens, a current recommended first-line regimen, 1 –3 produce treatment failure rates of approximately 20% to 40%. 4 –6 Guidelines recommend that HIV-infected patients who fail a first-line NNRTI-based regimen use a new antiretroviral (ARV) regimen that includes a ritonavir-boosted protease inhibitor (PI/r) and active nucleoside analog reverse transcriptase inhibitors (NRTIs). 3,7 However, as choices of active NRTIs may be limited by resistance or toxicity, a new ARV drug from other classes (such as etravirine [ETR], integrase strand transfer inhibitor [INSTI], or CCR5 antagonist) is an alternative option.
There are several problems associated with the use of PI/r with optimized background regimens (OBRs) in this setting: (1) the new ARV drugs (such as ETR, INSTI, and CCR5 antagonists) are generally not affordable in a resource-limited setting and (2) toxicities from NRTIs (eg, mitochondrial toxicity, bone marrow suppression, and pancreatitis) appear in approximately 8% of patients. 8
Previous studies evaluated the role of ritonavir-boosted lopinavir (LPV/r) monotherapy in HIV-infected patients who failed first-line NNRTI-based regimens and subsequently received LPV/r monotherapy as either (1) a second-line regimen after failing NNRTI-based regimens 9,10 or (2) an induction-maintenance strategy (such as, giving a standard LPV/r regimen 11 or LPV/r with raltegravir [RAL] 12 for a period of time and switching to LPV/r monotherapy later). The proportion of undetectable plasma HIV-1 RNA levels in these studies was significantly lower in patients receiving LPV/r monotherapy than in patients receiving LPV/r with 2 NRTIs. 9,11,12
Using LPV/r monotherapy as a maintenance strategy (ie, giving LPV/r monotherapy to patients who already achieved undetectable plasma HIV-1 RNA levels for a period of time) is different from the other strategies as previously mentioned (such as the HIVSTAR 9 and EARNEST 12 study).
Previous studies of HIV-infected patients who received first-line PI-based regimens revealed that the proportion of virologic suppression at the end of the study was not significantly different between those patients who receive maintenance PI/r monotherapy and those receiving standard PI/r regimen. 13 –15 Protease inhibitor resistance-associated mutations were relatively uncommon in the case of virologic failure in response to maintenance PI/r monotherapy. 14 –17 The reintroduction of NRTIs after low-level viremia or virologic failure in response to maintenance PI/r monotherapy was generally successful in achieving HIV-1 RNA suppression. 14,15,17 –19
To reduce NRTI-related toxicities and to lower the cost of drugs, a regimen-simplification strategy using maintenance PI/r monotherapy, after virologic suppression with second-line PI/r plus 2 NRTIs may be appropriate, especially in a resource-limited setting.
The objective of this study was to compare the virologic failure rate achieved with LPV/r monotherapy and LPV/r with OBRs in HIV-1-infected patients who failed a first-line NNRTI-based regimen and exhibited successful viral suppression during a second-line PI-based regimen.
Methods
Study Design and Participants
This 48-week-long, randomized, controlled, and open-label pilot clinical trial was conducted at the Bamrasnaradura Infectious Diseases Institute, Nonthaburi, Thailand. All patients were enrolled between January 2011 and December 2011. The eligibility criteria were as follows: (1) an age of 18 to 60 years; (2) documented HIV infection; (3) previously failed first-line NNRTI-based regimens (defined as having either an HIV-RNA level >400 copies/mL at 2 consecutive tests or any genotypic resistance to NNRTIs or NRTIs); (4) no history of failing PI-based regimens defined as HIV-1 RNA levels greater than 400 copies/mL at 2 consecutive tests or having a PI resistance-associated mutation; (5) receiving PI/r plus OBRs; and (6) having an undetectable HIV-1 RNA level for at least 6 months prior to enrollment. The exclusion criteria were as follows: (1) pregnancy or lactation; (2) hepatitis B coinfection; (3) moribund status; (4) serious systemic diseases (such as liver cirrhosis, Child-Pugh B/C, renal failure, and malignancy); (5) receiving medications that have a significant drug interaction with LPV/r; and (6) hemoglobin lower than 8 g/dL, platelets lower than 50 000/mm3, aspartate aminotransferase or alanine aminotransferase greater than 3 upper limit of normal, and estimated creatinine clearance lower than 50 mL/min.
Participants who experienced viral blips (defined as isolated viral rebound that was followed by viral suppression 1 ) during second-line PI-based regimens and who already had undetectable HIV-1 RNA levels were also eligible.
The study was approved by the institutional review board of the Department of Disease Control, Ministry of Public Health, Thailand. All participants gave written informed consent. The study was registered at “ClinicalTrials.gov” under the Identifier NCT01189695.
Randomization and Interventions
Block randomization was performed with an allocation ratio of 1:1. The allocation concealment was conducted by (1) separating the person with the assignment sequence from the person who assigned participants to groups and (2) sealing each assignment in sequentially numbered, opaque, and sealed envelopes.
Eligible participants were randomized to receive LPV/r either (1) as a monotherapy or (2) with OBRs. LPV/r could be (1) Aluvia (100/25 mg tablet; Abbott, Chicago) or (2) Lopinavir/Ritonavir (200/50 mg tablet; Matrix Laboratories Limited, India). The OBRs consisted of the same ARV medications as the regimens received before enrollment. The OBRs were modified only if toxicity or resistance occurred.
Follow-Up and Assessments
Participants attended study visits at weeks −4 to −1 (screening), 0 (start of the intervention), 2, 6, 12, 36, 48, and 52 (follow-up). Plasma CD4 counts and HIV-1 RNA levels were assessed at weeks 0, 12, 24, 36, and 48. If the HIV-1 RNA levels were higher than 400 copies/mL, the participant’s HIV-1 RNA level was reevaluated 4 weeks later. The plasma HIV-1 RNA assay and CD4 count were performed at the Immunology and Virology Laboratory, Medical Technology Section, Bamrasnaradura Infectious Diseases Institute. Before July 2011, plasma HIV-1 RNA was quantified by real-time polymerase chain reaction using the COBAS AmpliPrep/TaqMan HIV-1 Test (version 1.0) that can measure HIV-1 RNA levels between 40 and 10 000 000 copies/mL. After July 2011, plasma HIV-1 RNA was quantified with RT TaqMan using the COBAS AmpliPrep/TaqMan HIV-1 Test (version 2.0) that can measure HIV-1 RNA levels between 20 and 10 000 000 copies/mL. Adverse events were evaluated according to the Division of AIDS Table for Grading the Severity of Adult and Pediatric Adverse Events (DAIDS AE grading table).
Adherence to ARV treatment was assessed with a questionnaire. Nonadherence was defined as either (1) missing any dose of ARV treatment since the last visit or (2) missing any dose of ARV treatment in the previous 7 days.
Statistical Analysis
The primary outcome was virologic failure. Virologic failure was defined as 2 consecutive test results showing more than 400 HIV-RNA copies/mL. The secondary outcomes were as follows: (1) the proportion of participants with undetectable HIV-1 RNA levels at week 48 and at all visits, (2) the proportion of participants with persistent viremia (defined as 2 consecutive results showing detectable HIV-1 RNA levels), (3) the plasma CD4 count and the change in the CD4 count from baseline to week 48, and (4) all grades of adverse events according to the DAIDS AE grading table.
Persistent viremia was defined as 2 consecutive tests showing detectable HIV-RNA levels between 20 (or 40, as appropriate) and 400 copies/mL.
Viral blips before the start of the study were defined as those that occurred after achieving viral suppression by second-line PI-based regimens.
Categorical data were analyzed using a chi-square or Fisher exact test, as appropriate. Continuous data were analyzed using a Mann-Whitney U test. Logistic regression was performed to adjust for the effects of unbalancing characteristics. All P values were 2 sided, and P < .05 was considered to be statistically significant.
The baseline characteristic features and outcomes were reported as “per protocol analysis,” which is consistent with the CONSORT guidelines. 20 The analysis included participants who received the allocated treatment (n = 60; Figure 1). Three patients who were randomized withdrew consent before starting the study (week 0) and did not receive the allocated interventions.
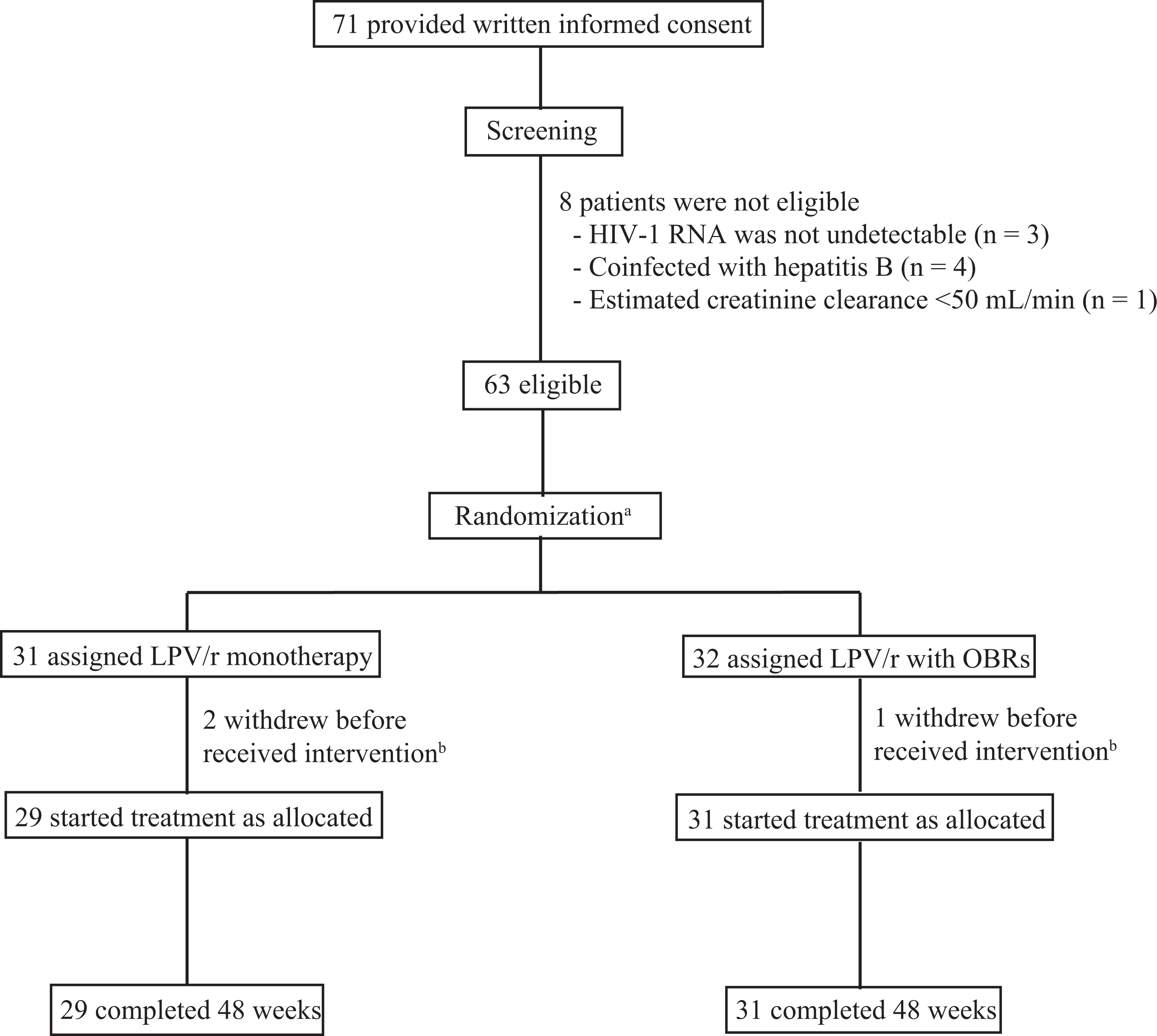
Schematic summary of study patients. LPV/r, ritonavir-boosted lopinavir; OBRs, optimized background regimens. aBlock randomization was performed and the allocation ratio was 1:1. bTwo patients in LPV/r monotherapy and 1 patient in LPV/r with OBRs withdrew consent before they received intervention; these 3 patients were therefore excluded from the analysis.
Results
Study Populations and Baseline Characteristics
Figure 1 shows the number of participants at each stage of the trial. In total, 63 participants were randomly assigned. In all, 2 participants in the PI/r monotherapy group and 1 participant in the PI/r plus OBRs group withdrew their consent after randomization and did not receive their allocated interventions. All participants who received interventions (29 in the PI/r monotherapy group and 31 in the PI/r plus OBRs group) completed the protocol. However, 4 participants (2 in each group) missed 1 clinical visit during the study period because of the great flood in Thailand in 2011.
Table 1 presents the baseline characteristics. Demographic features at the start of study were not different between the 2 groups. At baseline, most participants in both groups had high CD4 counts (median CD4 count of 527 cell/mm3 [interquartile range (IQR) 400-642]), a long duration of PI-based regimen, a long duration of undetectable HIV-1 RNA levels (median duration of 45 months [IQR 27-67]), and were receiving LPV/r.
Baseline Characteristics of Study Participants (n = 60).

Abbreviations: ZDV, zidovudine; DRV/r, ritonavir-boosted darunavir; ETR, etravirine; IQR, interquartile range; LPV/r, ritonavir-boosted lopinavir; NNRTI, nonnucleoside reverse transcriptase inhibitor; OBR, optimized background regimen; PI, protease inhibitor; TDF, tenofovir; 3TC, lamivudine; ALT, alanine aminotransferase.
a P value for difference between PI/r monotherapy and PI/r plus OBRs.
b Duration of undetectable HIV-1 RNA levels defined as the period from the first date that participants had 2 consecutive tests showing undetectable HIV-1 RNA levels until the start of treatment (week 0).
c Includes only viral blips that occurred while the participants received second-line PI-based regimens and when they already had undetectable HIV-1 RNA levels.
d According to IAS-USA Drug Resistance Mutations Group (December 2010 version). 21
Main Outcomes
Table 2 and Figure 2 detail the main outcomes of the study. Nearly all participants adhered to the protocol and their ARV medication regimens.

Virologic outcomes at the end of the study (week 48). LPV/r, ritonavir-boosted lopinavir; OBRs, optimized background regimens. aVirologic suppression was defined as having plasma HIV-1 RNA levels of either (1) less than 20 copies/mL (based on RT TaqMan using COBAS AmpliPrep/TaqMan HIV-1 Test, version 2.0; Roche Molecular Systems Inc, Branchburg, New Jersey) or (2) less than 40 copies/mL (based on real-time polymerase chain reaction [RT-PCR] using COBAS AmpliPrep/COBAS TaqMan HIV-1 Test; Roche Molecular Systems Inc, Branchburg, New Jersey). bViral blip was defined as isolated detectable HIV-1 RNA levels followed by undetectable HIV-1 RNA levels. c“Persistent viremia” was defined as 2 consecutive tests showing detectable HIV-RNA levels between 20 (or 40, as appropriate) and 400 copies/mL.
Comparison of Outcomes Achieved with LPV/r Monotherapy and LPV/r Plus OBRs (n = 60).

Abbreviations: IQR, interquartile range; LPV/r, ritonavir-boosted lopinavir; OBR, optimized background regimen; RT-PCR, real-time polymerase chain reaction.
a One participant reported at week 2 that she had missed a dose of antiretroviral drugs since her last visit; another participant reported at week 2 that she had missed a dose of antiretroviral drugs in the previous 7 days.
b This participant changed from Aluvia (100/25 mg tablet; Abbott, North Chicago) to Lopinavir/Ritonavir (200/50 mg tablet; Matrix Laboratories Limited, India) for financial reasons.
c These 2 participants changed from ZDV to TDF because of ZDV-related toxicities.
d Virologic failure was defined as 2 consecutive tests showing more than 400 HIV-RNA copies/mL.
e Undetectable plasma HIV-1 RNA levels were defined as having plasma HIV-1 RNA levels of either (1) less than 20 copies/mL (based on RT TaqMan using COBAS AmpliPrep/TaqMan HIV-1 Test, version 2.0; Roche Molecular Systems Inc, Branchburg, New Jersey) or (2) less than 40 copies/mL (based on RT-PCR using COBAS AmpliPrep/COBAS TaqMan HIV-1 Test; Roche Molecular Systems Inc, Branchburg, New Jersey).
f Two consecutive tests showing detectable HIV-RNA levels between 20 (or 40, as appropriate) and 400 copies/mL.
g Statistically significant.
h Adverse events included all severities of adverse events according to DAIDS AE grading table.
No virologic failure (primary outcome) occurred until the end of the study. The proportion of participants with undetectable HIV-1 RNA levels at week 48 was not significantly different between the 2 groups. However, the proportion of participants with persistent viremia tended to be higher among patients receiving PI/r monotherapy than in patients receiving PI/r with OBRs (10.3% versus 3.2%; P = .346; Table 2; Figure 2). At the end of the study, all 3 participants receiving PI/r monotherapy who had persistent viremia had undetectable HIV-1 RNA levels after receiving their baseline OBRs anew.
Patients receiving PI/r monotherapy had a median (IQR) CD4 count at week 48 that was significantly higher than that of patients receiving PI/r with OBRs (627 [520-808] versus 523 [426-635], P = .008). Moreover, the median (IQR) increase in the CD4 count from baseline to week 48 tended to be higher in patients receiving PI/r monotherapy than in patients receiving PI/r with OBRs (Table 2). Subgroup analysis of PI/r with OBRs indicated that the median (IQR) CD4 count at week 48 of those who received ZDV tended to be lower than those who did not (517 [386-635] versus 607 [497-761], P = .071).
Participants who had undetectable HIV-1 RNA levels at all study visits had a significantly lower incidence of viral blips before the start of the study than participants who did not have undetectable levels (22% versus 53%, P = .035). The adjusted relative risk of having undetectable HIV-1 RNA levels at all study visits for participants who had a history of a viral blip compared to those who did not have a history of viral blip before the start of the study was 0.221 (95% confidence interval 0.059-0.823, P = .024; Table 3).
Factors Associated with Undetectable HIV-1 RNA Levels at all Visits (n = 60).

Abbreviations: CI, confidence interval; IQR, interquartile range; LPV/r, ritonavir-boosted lopinavir; OBR, optimized background regimen; PI, protease inhibitor; RR, relative risk.
a Included participants who experienced viral blip during the study period (n = 15) and who had persistent low viremia (n = 4).
b The duration of having undetectable HIV-1 RNA levels was defined as the period from the first date that participants had 2 consecutive tests showing undetectable HIV-1 RNA levels until the start of treatment (week 0).
c Includes only viral blips that occurred while participants achieved viral suppression by second-line PI-based regimen and before the start of the study.
d Multivariate analysis was constructed with binary logistic regression model.
e Adjusted for receiving LPV/r monotherapy, nadir plasma CD4 count.
f Statistically significant.
g According to IAS-USA Drug Resistance Mutations Group (December 2010 version). 21
Adverse Events
The severity of all adverse events according to the DAIDS AE grading table is described in Table 2. Two participants (6.5%) in the PI/r with OBRs group experienced zidovudine (ZDV)-related toxicities and had to switch from ZDV to other NRTIs.
Discussion
The use of LPV/r monotherapy as a maintenance regimen in HIV-infected patients who failed a first-line NNRTI-based regimen and experienced viral suppression during a second-line PI-based regimen resulted in persistent viremia; the viremia of these patients tended to be higher than that of patients receiving the control treatment (LPV/r with OBRs; Figure 2 and Table 2). However, the participants who had persistent low viremia could resuppress HIV-1 RNA after intensified regimens with baseline OBRs. These data are consistent with the results of a previous study on PI/r monotherapy as a maintenance regimen after viral suppression. 13 –15,22 Ritonavir-boosted lopinavir as a maintenance monotherapy produces better immunological outcomes and less toxicity from NRTIs than the control therapy (Tables 2 and 3).
The EARNEST 12 study randomized HIV-infected patients who failed first-line NNRTI-based regimens to receive (1) LPV/r with NRTIs, (2) LPV/r with RAL, and (3) LPV/r with RAL during the 12-week induction followed by LPV/r monotherapy. The proportion of viral suppression in LPV/r monotherapy was significantly lower than LPV/r with NRTIs (74% versus 44%, P < .0001). The HIVSTAR 9 study reported that participants who received LPV/r as a second-line therapy after failing NNRTI-based regimens had undetectable plasma HIV-1 RNA levels at week 48, which are significantly less frequently than those receiving LPV/r with 2 NRTIs (61% versus 83%; P < .01). The SARA study, another clinical trial in HIV-infected patients who failed first-line NNRTI-based regimens, used LPV/r monotherapy after 24 weeks of LPV/r with 2 NRTIs. 11 This study also showed that fewer participants receiving LPV/r monotherapy had undetectable plasma HIV-1 RNA levels at week 24 than those receiving LPV/r with 2 NRTIs (60% versus 77%; P = .009).
Our study differs from the EARNEST, 12 HIVSTAR, 9 and SARA 11 studies in the following respects: (1) a different strategy consisting of a maintenance strategy that started LPV/r monotherapy after achieving viral suppression was used in this study and (2) the virologic outcome was better in the current study. The proportion of participants receiving LPV/r monotherapy with undetectable plasma HIV-1 RNA levels at week 48 was not different from the proportion of patients receiving LPV/r with OBRs (86.2% versus 87.1%; P = 1.000 [perprotocol analysis]; Table 2).
Although the virologic outcome of PI/r monotherapy is inferior to the outcome of a standard PI/r regimen, 19 the efficacy of PI/r monotherapy can be improved if PI/r monotherapy is started after viral suppression is achieved for a period of time. 16 A long duration of plasma viral suppression before receiving PI/r monotherapy was associated with a good virologic outcome. 23 HIV-infected patients who had undetectable plasma HIV-1 RNA levels for more than 2 years had lower residual viral replication than patients who had undetectable HIV-1 RNA levels for less than 2 years. 24 Additionally, in this study, participants who had experienced a viral blip before randomization had a virologic outcome inferior to the outcome of individuals without a viral blip (Table 3). Therefore, PI/r monotherapy as a maintenance regimen should be used in HIV-infected patients who have very low residual viral replication indicated by (1) a lack of a viral blip before starting PI/r monotherapy and (2) suppressed HIV-1 RNA levels for a long period of time.
Current guidelines do not recommend using PI/r monotherapy as a maintenance regimen outside of clinical studies. 1,7 However, Moltó et al conducted a retrospective study of 51 HIV-infected patients using LPV/r monotherapy as a maintenance regimen 25 during routine clinical practice. The median duration of viral suppression before LPV/r monotherapy was 30 (15-56) months, and only 2 patients had persistent viremia. These 2 patients did not have any PI mutation and achieved viral suppression after baseline NRTIs were reintroduced. Thus, regimen simplification with maintenance PI/r monotherapy might be an option in selected clinical settings, especially in a resource-limited setting in which a patient has experienced several NRTI toxicities and cannot afford new ARV drug classes.
To improve the virologic outcome of maintenance PI/r monotherapy, several clinical characteristics should be considered before using PI/r monotherapy: (1) a long duration of viral suppression; (2) a viral blip during the period of viral suppression; (3) hepatitis C virus (HCV) coinfection (as HCV was a predictor of persistent viremia in a clinical trial examining ritonavir-boosted darunavir [DRV/r] monotherapy as a maintenance regimen 14 ); and (4) a nadir CD4 count of less than 100 cells/μL, which is a predictor of the loss of virologic suppression (adjusted hazard ratio of 4.1 [1.3-13.5], P = .02). 26 These clinical characteristics should also be considered carefully when designing future studies on PI/r monotherapy.
Detectable HIV-1 RNA in anatomical sanctuaries (such as the cerebrospinal fluid [CSF] and semen) is a concern when using PI/r monotherapy. 17,27 In all, 15% and 13% of HIV-infected patients with suppressed plasma HIV-1 RNA levels had detectable HIV-1 RNA in the CSF and seminal plasma, respectively. 27 The long-term clinical consequence of suboptimal viral suppression in the central nervous system during PI/r monotherapy should be considered. Acute or subacute neurological symptoms and HIV encephalopathy were reported in a patient who had suppressed plasma HIV-1 RNA and persistent detectable HIV-1 RNA in the CSF. 28,29 Studies of the long-term clinical outcome of PI/r monotherapy are therefore warranted.
The lower CD4 count observed at week 48 in participants who received LPV/r with OBRs may be due to ZDV. Previous studies showed that individuals who received ZDV as an NRTI backbone in ARV regimens had a lower CD4 count than individuals who did not receive ZDV. 30 Here, 17 (55%) participants receiving LPV/r with OBRs also received ZDV. At week 48, the median CD4 count of participants who received ZDV tended to be lower than that of those who did not.
The small sample size of each allocated group and the short duration of the follow-up period are the 2 limitations of this study that should be considered when interpreting the data.
In conclusion, LPV/r monotherapy used as a maintenance regimen in participants who failed a first-line NNRTI-based regimen more frequently resulted in a persistent viremia than LPV/r with OBRs. Participants who had persistent viremia during LPV/r monotherapy could achieve viral suppression after regimen intensification with their baseline OBRs. A history of a viral blip during the period of viral suppression before enrollment was associated with an increased likelihood of achieving full viral suppression at all visits. Ritonavir-boosted lopinavir with OBRs yielded NRTI-related toxicities and a lower CD4 count at week 48 than LPV/r monotherapy.
Footnotes
Authors’ Note
All authors contributed to writing the study protocol and editing the final article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from the Department of Disease Control, Ministry of Public Health, Thailand.
