Abstract
Introduction:
This study describes the epidemiologic features and clinical course of children with blood transfusion-associated HIV infection (TAHI) in Ibadan, Nigeria.
Methodology:
All children diagnosed to have TAHI at the University College Hospital, Ibadan, were studied and compared with children who acquired HIV vertically using the pediatric HIV database in the hospital.
Results:
Transfusion-associated HIV infection accounted for 14 (2.3%) of the 597 children diagnosed to have HIV infection between January 2004 and December 2011. The mean age at diagnosis of TAHI was 10.2 years and that of vertically acquired HIV infection was 3.9 years (P < .001). In 9 cases, blood transfusion took place in private hospitals and in 5 cases in public hospitals. Median interval between infection and diagnosis of AIDS was 84 months in cases with TAHI and 48 months in vertically acquired cases (P = .542).
Conclusion:
Optimal blood safety practices are advocated for prevention of TAHI in Nigeria.
Introduction
Most cases of pediatric HIV infection result from mother-to-child transfusion. 1 However, transfusion of contaminated blood remains an important, although less common, cause in developing countries. 2, 3 Unfortunately, there is a dearth of publications that focus on this potentially preventable source of HIV infection. Although a number of workers have studied the risk of HIV in sub-Saharan countries based on HIV prevalence in blood donations, 4 there is little literature on the epidemiology and clinical course of pediatric HIV infections resulting from or related to blood transfusions.
Nigeria accounts for 10% of global pediatric HIV and AIDS. 5 Few available studies in the country suggest markedly varied contributions of blood transfusion to pediatric HIV. An early study of the HIV epidemic in Nigeria (somewhere in southeastern Nigeria) reported that 68% of cases were associated with blood transfusion. 6 More recent data indicate that it contributes to 4.6% to 16.4% of HIV in children. 3, 7 On the contrary, a 5-year study involving 840 infected children in Sokoto, northwestern Nigeria, found no cases attributable to blood transfusion. 8 This wide variation calls for more studies across the country to ascertain the true picture of transfusion-associated HIV infection (TAHI). In addition, data are still lacking on the epidemiology and clinical course of the condition in the country. The objectives of the study were to determine the frequency of TAHI in children and to describe the demographic characteristics, epidemiologic features, and clinical course of affected children in Ibadan, Nigeria.
Methodology
Nigeria is divided into 6 geopolitical zones and the Federal Capital Territory. 9 There are 3 zones in the northern part of the country, namely north-west, north-central, north-eastern, and 3 in the southern part of the country, namely south-west, south-south, and south-east. This study took place at the University College Hospital in Ibadan, capital of Oyo state located in the south-western geopolitical zone. The hospital is a tertiary 850-bed hospital that serves as a referral center for Oyo state generally and other parts of the country for certain conditions.
This study involved all children diagnosed with HIV infection, at the University College Hospital, Ibadan, between January 2004 and December 2011 but focused on those thought to have acquired the infection from transfusion with infected blood. Inclusion criteria were HIV infection and a history of blood transfusion in children whose mothers were HIV negative and who had no history of sexual activity or other risk factors for HIV infection. Data were extracted from the database of HIV-infected children and from their case notes at the hospital. Initial laboratory diagnosis of HIV infection among children aged 18 months or more was done using a fourth-generation enzyme-linked immunosorbent assay (ELISA; Genescreen, Biorad, Paris, France). All ELISA reactive samples were then confirmed by Western immunoblot. However, infection among children <18 months was established using a commercially available HIV DNA polymerase chain reaction kit (Roche amplicor, Roche Molecular Systems, Inc, Branchburg, New Jersey). All assays were performed according to the manufacturer’s recommendation.
Data extracted included demographic variables such as age at diagnosis of HIV infection and sex. Other data extracted included date and health facility in which blood transfusion took place and the estimated time intervals between blood transfusion and onset of HIV-related clinical events. HIV-related clinical events and clinical stage at diagnosis were assessed based on World Health Organization (WHO) guidelines. 10 Intervals between HIV infection and diagnosis of advanced HIV disease and AIDS were also computed. For cases of TAHI, the date of infection is presumed to be the date of transfusion while for vertically acquired cases, the date of infection was presumed to be the date of birth. 11 AIDS was defined as clinical diagnosis (presumptive or definitive) of any stage 4 condition with confirmed HIV infection, or immunological diagnosis with confirmed HIV infection and in children >5 years of age, first-ever documented CD4 count less than 200 cells/mm3 or %CD4 <15, among children aged 12 to 35 months with confirmed HIV infection, first ever documented %CD4 < 20, or among children of less than 12 months of age with confirmed HIV infection, first ever documented %CD4 < 25. 10 CD4 counts or %CD4 at diagnosis was also done for all patients and results categorized into severe or not severe immunosuppression based on WHO age-appropriate absolute CD4 count thresholds for severe immunosuppression. 10
The study was carried out in compliance with Helsinki declaration, and ethical approval was obtained from the ethics committee of the University of Ibadan/University College Hospital, Ibadan.
Statistical analysis was done using Statistical Package for the Social Sciences version 16 software package. Means (and standard deviations [SDs]) and medians were computed for continuous variables and frequencies for categorical variables. Means were compared using t test, and association between categorical variables tested using chi-square (χ2) test and when appropriate Fisher exact test. Kaplan-Meier survival analysis method was used to determine the median times between infection and diagnosis of advanced disease and AIDS and comparisons between cases of TAHI and vertically acquired HIV infection made using log rank test. Statistical significance was set at P < .05.
Results
Prevalence and Demographic Variables
During the study period, a total of 597 HIV-positive children, comprising 320 (53.2%) boys and 277 (46.4%) girls, were recruited in the study. Of the 597 cases, TAHI accounted for 14 (2.3%) cases and vertical transmission for 579 (97%) cases. The route of HIV acquisition was unknown in 4 (0.7%) children: these were children from orphanages, whose mothers died under uncertain circumstances and were not tested for HIV before their death. Of the 14 cases of TAHI, there were 5 boys and 9 girls, giving a male–female ratio of 1:1.8; whereas among the cases of vertically acquired HIV infection, there were 313 boys and 266 girls, yielding a male–female ratio of 1.2:1 (Fisher exact test P = .188).
Clinical Presentation
The most common clinical features at presentations for transfusion-associated and vertically acquired HIV infections were prolonged fever, weight loss, and chronic cough (Table 1). Next in frequency were generalized lymphadenopathy, chronic diarrhea, and papular pruritic dermatitis. Parotid swelling and nephropathy occurred only in vertically acquired HIV infection but not in transfusion-associated cases. However, except for seborrheic dermatitis that was more frequent in cases of TAHI, there was no statistically significant difference in the clinical features between TAHI and vertically acquired HIV infection.
Clinical Features of Cases with HIV Infection at Presentation.

a Fisher exact test.
Pulmonary tuberculosis was present in 4 (28.6%) of 14 cases with TAHI compared with 256 (44.2%) of 579 cases with vertically acquired HIV infection (Fisher exact test P = .286). Non-Hodgkin lymphoma occurred in 1 (7.7%) case with TAHI and Kaposi sarcoma in 1 case with vertically acquired HIV infection.
In cases with TAHI, the age at receipt of blood transfusion ranged from 1.3 to 12.4 years, with a mean of 5.9 (SD 2.9) years and a median of 5.4 years. The age at diagnosis of HIV infection ranged from 2.3 to 15.0 years, with a mean of 10.2 (SD 3.4) years among children with TAHI, whereas it ranged from 0.08 to 14 years, with a mean of 3.9 (SD 3.2) years (P < .001) among children with vertically acquired HIV infection. The median age at diagnosis of HIV infection was 10.4 years in cases with TAHI and 3 years in cases with vertically acquired infection.
The illnesses that led to blood transfusion were anemia associated with febrile illnesses (including malaria and meningitis) in 11 children, sickle cell anemia in 1 child, epistaxis in 1 child, and post-traumatic (gunshot) splenectomy and hemorrhage in 1 child. Hemoglobin phenotype was known in only 6 children with TAHI, of whom only 1 was hemoglobin SS and the remaining 5 were hemoglobin AA.
Location and Age at Which Blood Transfusion Took Place
With regard to health facilities where the blood transfusions took place, 9 (64.3%) were in private hospitals, 4 (28.6%) in government-owned secondary care hospitals, and 1 (7.1%) in a government-owned tertiary care hospital. The units of blood used for transfusions in the private hospitals and in 1 state hospital were from private laboratories. All others were supplied by the hospitals.
Figure 1 shows the years in which the children received blood transfusions that were believed to have resulted in HIV infection, indicating that between 1996 and 2010 about 1 to 2 cases occurred per year but none recorded in 1997 to 1999 and 2009. Table 2 suggests a relatively high incidence of TAHI in relation to the overall HIV diagnosis between 2004 and 2005 when the total number of new cases was few compared to subsequent years when the total number of new cases increased annually.

Number of blood transfusions per year.
Incidence of TAHI in Relation to Overall HIV Diagnoses Over the Study Period.
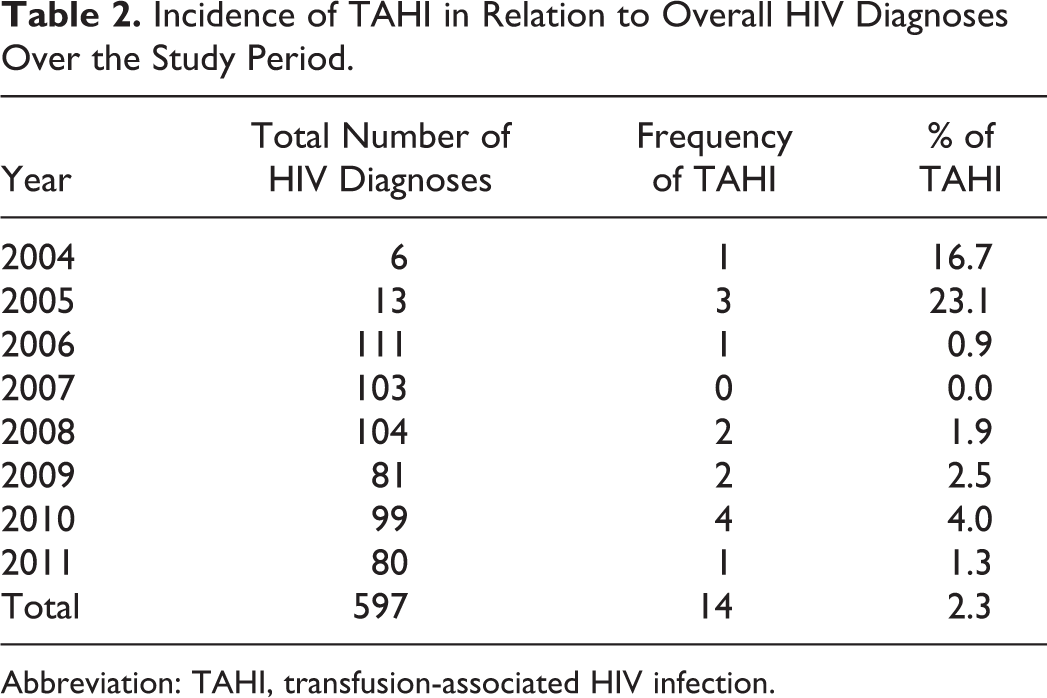
Abbreviation: TAHI, transfusion-associated HIV infection.
Clinical Stage and Immunological Status at Diagnosis
At the time of diagnosis, 10 (71.5%) children with TAHI had advanced and severe HIV symptoms compared with 393 (67.9%) children with vertically acquired HIV infection (Table 3). At diagnosis, 3 (21.4%) children with TAHI had severe immunosuppression compared to 316 (54.6%) children with vertically acquired HIV infection (Table 3). Children with vertically acquired HIV infection were more likely to have severe immunosupression at diagnosis than those with TAHI (odds ratio 4.4, 95% confidence interval [CI] 1.2-16.0). Advanced disease was present in 10 (71.4%) of the 14 cases with TAHI and in 479 (82.7%) of the 579 cases with vertically acquired HIV infection (Fisher exact test P = .284). At diagnosis, AIDS was present in 7 (50%) of the 14 cases with TAHI compared with 377 (65.1%) of 579 cases with vertically acquired HIV infection (Fisher exact test P = .265).
World Health Organization Clinical Stages and Immunological Classification of Children With Vertically Acquired and Transfusion-Associated HIV Infections at Presentation.

Time Intervals between Year of Blood Transfusion and HIV Disease
Interval between blood transfusion and onset of HIV-related symptoms ranged from 2 to 114 months, with a median of 18 months. Median interval between infection and diagnosis of advanced disease in cases of TAHI was 60 months (95% CI 46.1-73.9), whereas the median interval from birth to diagnosis of advanced disease in vertically acquired HIV cases was 40.9 (95% CI 36.8-45.0). There was no significant difference between time to diagnosis of advanced disease between the cases of TAHI and vertically acquired cases (log-rank (Mantel-Cox) χ2 = 0.131, P = .717). Median interval between infection and diagnosis of AIDS in cases with TAHI was 84 months (95% CI 39.7-128.3) and in those with vertically acquired infection was 48 months (95% CI 41.2-54.8). There was no significant difference in the time interval from infection to diagnosis of AIDS between the cases with TAHI and those with vertically acquired infection (log-rank [Mantel-Cox] χ2 = 0.372, P = .542).
Discussion
Nigeria established the National Blood Transfusion Service (NBTS) in 2005 and the National Blood Policy in 2006 in order to improve blood safety and reduce transfusion-transmissible infections; the NBTS established regulations for reducing transfusion-related HIV. 12,13 However, the impact of these regulations on the contribution of blood transfusion to HIV infection has yet to be assessed in the country. The 2.3% prevalence of TAHI among HIV-infected children revealed in our study is lower than the 5.7% reported from a previous study in India and 4.6% to 68% in other parts of Nigeria. 2, 3, 6, 7 However, the expected impact of the NBTS which is a downward trend in the occurrence of TAHI after the implementation is not evident in our center. It may be argued that given the incubation period of the infection and therefore time to presentation, a few years more should be allowed to assess the impact of the NBTS and National Blood Policy. However, although our data are small, the annual numbers of transfusions that resulted in HIV infection prior to and after 2006 in our study are not appreciably different. This does not come as a surprise as the guidelines outlined by NTBS have not been widely implemented and most donors are still being paid in the country. 14
The median age at which blood transfusion was received was 5.4 years in the present study which contrasts significantly with TAHI reported in children in the United States with a median age at transfusion of less than 2 months. 11 Although 71% of transfusions occurred in the first year of life in the United States, all cases in our study took place after 1 year of age. Another difference in the epidemiology of TAHI in the United States and Nigeria is in the sex distribution. In the United States, boys were overrepresented in pediatric TAHI, but sex was found to be evenly distributed in perinatally acquired HIV. 11 On the contrary, in our study, girls were slightly more frequently affected in TAHI, although the difference did not reach statistical significance. The differences in the ages at receipt of blood transfusion and sex distribution of cases of TAHI in Nigeria compared to the United States may reflect the different underlying conditions that warrant neonatal transfusion and infant transfusions in the United States in contrast to conditions that warrant blood transfusion in older ages in our study. In our study, most transfusions resulted from febrile illnesses probably due to infections such as malaria, which is known to be a major contributor to severe anemia requiring blood transfusions in Africa. 15 In addition to malaria, other causes of anemia that led to blood transfusion in the present study, such as sickle cell anemia, epistaxis, and gunshot injuries, are uncommon in early infancy and not known to have any sex predilection.
A previous study in Nigeria had revealed that almost all cases with TAHI resulted from transfusions that took place in private health facilities. 16 Our study confirms that more cases occur in private health facilities and also reveals that over a third of cases took place in public health facilities. Therefore, an audit of blood transfusion and safety practices is necessary in both public and private health facilities in the country, with special attention to private facilities.
In our study, the age at diagnosis of TAHI is higher than that of perinatally acquired HIV infection in keeping with findings in the United States. 11 In addition, the median age of 10.4 years at diagnosis of TAHI in Ibadan is higher than the median age of 4 years at diagnosis of transfusion-associated AIDS in the United States, presumably due to the older age at transfusion in our patients.
In this study, the median time to onset of HIV-related symptoms in cases with TAHI was 18 months, which is similar to 17.8 months reported in cases resulting from neonatal transfusion in the United States, 17 but longer than 9 months reported in a previous study in Nigeria. 16 A study by Jones and colleagues reported a shorter incubation period for the development of AIDS in perinatally acquired AIDS compared to transfusion acquired AIDS in children. 11 In the present study, although the median interval between infection and diagnosis of AIDS in cases of TAHI appeared longer than that of vertically acquired HIV infection, the difference was not statistically significant in our study. However, the median time of 84 months (7 years) from transfusion to diagnosis of AIDS in cases with TAHI in the present study is much longer than 3.5 years in pediatric transfusion-associated AIDS in the United States. 11 Given the fact that most of the transfusions in the United States occurred in infancy unlike in our study in which transfusions occurred at an older age, the shorter interval to diagnosis of AIDS in the former may be due to immunological immaturity of infants. 18 Similarly, the greater likelihood of children with vertically acquired HIV infection to have severe immunosuppression at diagnosis compared to cases with TAHI in our study may be explained by the younger age at infection and relative immunological immaturity.
The illnesses reported in pediatric transfusion-associated AIDS cases in the United States are similar to those reported in perinatal AIDS cases except that in cases with pediatric transfusion-associated AIDS, lymphoid interstitial pneumonitis is less likely and opportunistic infections and HIV encephalopathy are more likely to be reported. 11 Although the present study was not restricted to cases with AIDS and so may not be completely comparable to that of the United States, we also observed similar presenting features between cases with TAHI and those with vertically acquired HIV infection but with increased tendency for seborrheic dermatitis to occur in cases with TAHI. This has not been previously documented, and the reason for the difference is not clear but may reflect a difference in the behavior of HIV infection acquired through the 2 different modes. Studies with larger numbers of patients are required to verify our findings. The high percentage of children with advanced disease and severe immunosuppression at diagnosis in our study is a reflection of late diagnosis; a common occurrence in Nigeria. 7
The continued occurrence of new cases of TAHI in Nigeria is probably due to the sole reliance on ELISA testing for HIV in the country, which is inadequate for screening blood from donors since new infections contracted 90 days prior to testing cannot be detected. 19 Such new cases may be diagnosed by screening with antigen and DNA tests in addition to ELISA testing. 19 This approach in blood banking might help in the reduction of HIV transmission through blood transfusion.
Limitation
The safety of blood and types of screening tests carried out, if any could not be ascertained by the parents. In 1 case, the blood donor was the father who was subsequently diagnosed HIV positive; in this case, it is not known whether the blood was screened before transfusion. Although the number of cases with TAHI studied was small, the findings from the study would add to what is known about TAHI in Nigeria.
Conclusion
Blood transfusion continues to be a mode of transmission of pediatric HIV infection in Ibadan and its environs. Blood transfusions in most cases took place in private hospitals but state hospitals are also involved. Implementation of optimal blood safety practices at all levels of health care is advocated to prevent the occurrence of TAHI. Targeted screening of children with a history of blood transfusion would also aid in early diagnosis of HIV and prompt management.
Footnotes
Acknowledgments
The laboratory investigations for the diagnosis of HIV and monitoring of treatment as well as antiretroviral drugs administered were sponsored by the AIDS Prevention Initiative in Nigeria Plus Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no financial support for authorship and publication of this article but the research was partly funded by the AIDS Prevention Initiative in Nigeria Plus.
