Abstract
Objective
To assess some quality of life (QOL) aspects of a low protein diet, using glycomacropeptide (GMP) as a protein substitute in patients with phenylketonuria (PKU).
Methods
This was a multicentre, prospective observational cohort, study. Metabolic control, nutritional parameters, and dietary adherence were assessed in patients with PKU before (T0), and six months after (T6) starting a low protein diet using GMP. Selected items from the PKU-QOL questionnaire were used to assess patients’ acceptance of their modified diet.
Results
18 patients from three Italian Centres, completed the study. With the exception of LDL-cholesterol and vitamin 25OH-D concentrations, there were no differences between T0 and T6 in metabolic or nutritional parameters. Data suggested that patients have a good acceptance of protein substitutes containing GMP, probably because of their improved palatability.
Conclusions
According to our patients’ responses to items related to dietary regimen, GMP based protein substitutes do not appear to significantly affect QOL.
Introduction
Phenylketonuria (PKU) is an autosomal recessive inborn error of metabolism caused by defects in the phenylalanine hydroxylase (PAH) gene. 1 On average, it occurs in 1:10,000 live births in Europe, with a higher rate in some countries such as Ireland and Turkey, and a low rate in Finland. 2 Hepatic PAH catalyses the oxidation of phenylalanine (Phe) to tyrosine (Tyr), and so defects in PAH activity lead to increased plasma Phe concentrations, that can lead to detrimental, irreversible effects on brain health. 3 Since the early 1960 s, neonatal screening test for PKU has become available worldwide, and provides an extraordinary tool for the prevention and eradication of this clinical condition and its related neurodevelopmental harmful effects. Depending on the specific genetic variant (more than 1000 variants in the PAH gene have been identified so far), different degrees of PAH protein loss of function occur, and so determine the different biochemical and clinical phenotypes. 4 Currently, hyperphenylalaninemias (HPAs) are classified according to treatment needs and include non PKU HPA (Phe concentration ranging of 120–360 μmol/l on unrestricted diet) and PKU HPA (Phe concentration >360 μmol/l on unrestricted diet). 5 European guidelines recommend lifelong treatment aimed at keeping plasma Phe levels within the target range of 120–360 μmol/l until the age of 12 years and within 120–600 μmol/l for non-pregnant individuals older than 12 years. 5 A low Phe diet is the mainstay of treatment with the aim of achieving plasma Phe concentrations of 120–360 µmol/l for those patients with Phe levels above 360 μmol/l; patients with levels less than 360 μmol/l do not require treatment. 5
Some patients with PKU may respond to tetrahydrobiopterin (prescribed as sapropterin dihydrochloride), an essential cofactor for PAH, which acts by decreasing plasma Phe concentrations and may also increase dietary Phe tolerance. 6 When available, casein glycomacropeptide (GMP) or large neutral amino acids (LNAAs) are also used for the treatment of PKU. 2 GMP is a 64-amino acid peptide extracted from the whey fraction of bovine milk. 7 Although GMP based protein substitute products are low in Phe, they contain some residual Phe as a result of the extraction from other whey proteins. 8 Nevertheless, GMP has several advantages over a LNAA diet including improved palatability9–11 and fewer side effects.10–12
In addition to metabolic and nutritional aspects of the disorder, the burden of a lifelong restricted Phe diet may have significant detrimental effects on the patients’ quality of life (QOL) as well as intrafamilial relationships. 13 However, results from a study in 111 Italian adult patients with PKU found that most participants did not perceive that their dietary regimen had a negative effect on their QOL and many did not consider PKU as a disease. 14
To the best of our knowledge, there is a lack of data on the possible QOL effects of low Phe dietary treatment using a GMP based protein substitute in patients with PKU. We hypothesised that the introduction of GMP into the dietary plans of patients with PKU would be associated with several effects, among which improved palatability and a patient’s feeling of greater satiety may help ameliorate their perception of their QOL. Therefore, in this study we used selected items from the validated PKU-QOL questionnaire 15 that were related to dietary aspects and assessed patients’ acceptance of a low protein diet using a GMP based protein substitute.
Patients and Methods
This was a multicentre, prospective observational cohort, study conducted from January to August 2021. Patients were recruited from three Italian Clinical Referral Centres (Paediatric and Neonatology Unit, Guglielmo da Saliceto Hospital, Piacenza; Metabolic Unit, Giannina Gaslini Children’s Hospital, Genoa; Rare and Metabolic Diseases Unit, AORN Santobono-Pausilipon, Naples). Inclusion criteria were as follows: patients aged >9 years with PKU treated shortly after birth with a low-Phe diet; diagnosis of PKU based on unrestricted diet plasma Phe concentration of >600 µmol/l; acceptance of using GMP as a source of protein substitute. Patients who were receiving ongoing treatment with LNAAs or sapropterin were excluded from the study. The same GMP derived protein substitute (Ajinomoto-Cambrooke, Inc., MA, USA) was used for all patients.
Disease-specific questionnaires (Child PKU-QOL, 9–11 years; Adolescent PKU-QOL, 12–17 years; Adult PKU-QOL, > 18 years) were administered to the patients by a single skilled and trained psychologist before starting GMP (T0) and six months later (T6). 15 The PKU-QOL questionnaire includes items (child, adolescent, and adult contain 40, 58 and 65 items, respectively), subdivided into four modules that target the most important areas of disease management (i.e., PKU symptoms; PKU in general; administration of Phe-free protein supplements; dietary protein restriction). 15 For this study, we concentrated our analysis on five items included in the ‘administration of Phe-free protein supplements’ module.
At each outpatient visit, body weight, height and BMI (kg/m2) were recorded. In addition, patients were asked to document their food on the previous three days. From the three-day food record the following daily intakes were calculated: natural protein (g/kg/day); protein equivalent from protein substitute (g/kg/day); % GMP of total protein substitute intake; Phe intake (mg/day) from natural foods and GMP. The daily amount of Phe intake was assessed according to the average Phe tolerance.
At each clinic visit, the following parameters were assessed: fasting Phe and Tyr concentrations measured from dried blood spots using tandem mass spectrometry; fasting plasma uric acid; glucose; creatinine; urea; total cholesterol; high density lipoprotein (HDL) cholesterol, low density lipoprotein (LDL) cholesterol; triglycerides; iron; transferrin; ferritin; albumin; calcium; phosphorus; vitamin B12; vitamin 25OH-D; folic acid; white blood cells (WBC); red blood cells (RBC); haemoglobin.
The reporting of this study conforms to STROBE guidelines. 16 All participants or their parents/guardians provided written informed consent. Patient data were de-identified prior to analysis and so Ethics Committee approval was not required.
Statistical analyses
The Shapiro-Wilks test was used to determine if variables were normally distributed. Student t-test was used to compare normally distributed continuous variables and Wilcoxon signed-rank test was used for non-normally distributed variables. The Wilcoxon signed-rank test was used for the comparison of responses at T0 and T6 to PKU-QOL items. A P-value <0.05 was considered to indicate statistical significance.
Results
Of 21 selected patients with PKU, 18 patients were included in this study (Table 1). Of these, five were aged 9–11 years, four were 12–17 years and nine were > 18 years. Of the mean amount of daily protein substitutes prescribed (i.e., 42.6 g/day; range 25–60 g/day), the mean percentage of GMP derived protein substitute was 70% (range: 20–100%), and the overall mean Phe intake was 404 mg/day (range: 220–536 mg/day).
Characteristics of the study population (n = 18) at study start (T0).

Values are shown as n, or mean ± SD.
BMI, body mass index.
There were no significant differences between T0 (study entry) and T6 (six months of dietary GMP) in plasma Phe or Tyr concentrations (Table 2). With the exception of LDL-cholesterol and vitamin 25OH-D concentrations, there were no other differences between T0 and T6 in metabolic or nutritional parameters (Table 2). LDL-cholesterol levels were significantly higher (P = 0.009) and vitamin 25OH-D levels significantly lower (P = 0.004) at T0 compared with T6.
Haemato-biochemical profile of patients (n = 18) evaluated at study start (T0) and after six months on GMP-based protein substitute (T6).

Values are shown as mean ± SD.
GMP, glycomacropeptide; HDL, high density lipoprotein; LDL, low density lipoprotein; ns, not statistically significant; RBC, red blood cells; WBC, white blood cells.
Of 18 patients included in the study, 14 provided responses to the five items chosen from the ‘administration of Phe-free protein supplements’ module. The five items and their responses at T0 and T6 are shown in Figures 1 –5 and are summarised below:

Patients (n = 14) responses to the question

Patients (n = 14) responses to the question

Patients (n = 14) responses to the question
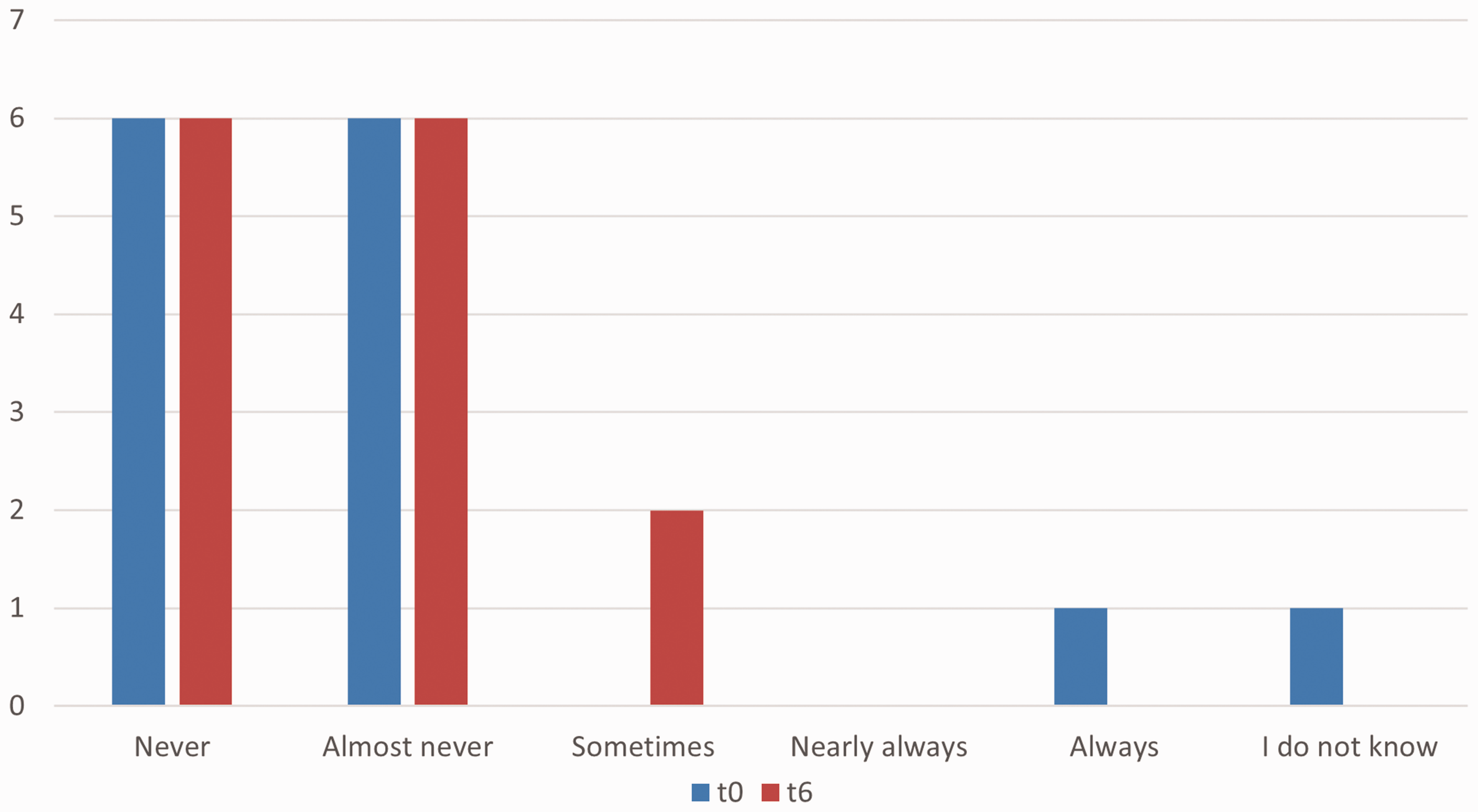
Patients (n = 14) responses to the question

Patients (n = 14) responses to the question
(1) “During the last 7 days, how hard was it to take protein substitutes?”
For, ‘not at all or a little,’ T0: 57% (8/14); T6: 79% (11/14) (Figure 1).
(2) During the last 7 days, the taste of protein substitutes has been … ”
For ‘good or excellent,’ T0: 36% (5/14); T6: 50% (7/14). For ‘bad or very bad,’ T0: 36% (5/14); T6: 7% (1/14) (Figure 2).
(3) During the last 7 days, I felt unease/embarrassed when taking protein substitutes in the presence of other people”
At T0, 64% (9/14) and at T6, 79% (11/14) recorded ‘never or almost never’ (Figure 3).
(4) During the last 7 days, was it hard to eat out, due to protein substitutes?”
For ‘never or almost never,’ T0: 86% (12/14); T6: 86% (12/14). For ‘always,’ T0: 7% (1/14). (Figure 4).
(5) I feel guilty when I do not take protein substitutes?”
For ‘not at all or a little,’ T0: 21% (3/14); T6: 7% (1/14). For ‘quite or very,’ T0: 64% (9/14); T6: 86% (12/14) (Figure 5).
As shown by the responses to the questions, the data suggest that patients have a good acceptance of protein substitutes containing GMP, probably because of their improved palatability. A trend towards a high awareness of the consequences of high plasma Phe appeared to be evident as well as a good adherence to the clinical and metabolic follow-up, especially in the adolescent and adult patients (data not shown).
Discussion
For this study, we enrolled PKU patients with insufficient metabolic control who had not previously received GMP. Six months after the introduction of GMP, we observed no significant modification of mean plasma Phe, suggesting that the Phe content of GMP derived products did not have a detrimental effect on the patients’ metabolic control. As for nutritional parameters, with the exception of vitamin 25OH-D and LDL-cholesterol, there were no significant changes after six months of GMP treatment. The significantly higher T6 concentration vitamin 25OH-D compared with T0, may have been due to a post-summer evaluation, with the obvious beneficial effect of the high exposure to sunlight over the summer months. However, it may have also been due to a high vitamin D intake from GMP derived protein substitutes compared with other amino acid mixtures. The significantly low T6 LDL-cholesterol levels may have been due to the reported beneficial effect of GMP on total and LDL-cholesterol. 17 Although not significant, a slight increase was noted in calcium and phosphorus concentrations at T6, which may reflect a potential beneficial effect of GMP on bone metabolism as well as a consequence of better mineral utilization due to the high vitamin D concentrations. Interestingly, improvements in bone health and body composition following treatment with GMP have been demonstrated in mice. 18
We hypothesised that the introduction of a GMP based protein substitute in the dietary plans of PKU patients would be associated with positive effects on the patients’ perception of their QOL. We selected five items from the ‘administration of Phe-free protein supplements’ module of the validated PKU-QOL questionnaire and assessed patients’ acceptance of their modified diet. We observed a trend towards a good acceptance and liking of the GMP based protein substitute and some psycho-emotional benefits. Interestingly, our findings are in contrast with those from a previous study in Italian adult patients with PKU that found the patients’ dietary regimen did not significantly impact their QOL. 14
The study had several limitations. For example, our results were based on a small sample of patients, using items from one module of the PKU-QOL questionnaire, recorded over a limited time interval (i.e., six months). In addition, there was no control group and so bias may have been introduced as the patients realised that they were being monitored for acceptance of the GMP based protein substitute. However, we believe a major strength of our study was that a single trained and skilled psychologist administered the questionnaire and interpreted the responses. Furthermore, the multicentre design of our study allowed us to include patients from the north and south of Italy so minimising possible biases linked to regional habits and/or lifestyle.
To the best of our knowledge, this is the first study aimed at specifically focusing on PKU patients’ acceptance of a GMP based protein diet. According to our patients’ responses to items related to dietary regimen, GMP based protein substitutes do not appear to significantly affect QOL.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221125524 - Supplemental material for Quality of life aspects of a low protein diet using GMP in patients with phenylketonuria
Supplemental material, sj-pdf-1-imr-10.1177_03000605221125524 for Quality of life aspects of a low protein diet using GMP in patients with phenylketonuria by Giulia Bensi, Maria Teresa Carbone, Maria Cristina Schiaffino, Sara Parolisi, Angela Pozzoli, Giacomo Biasucci in Journal of International Medical Research
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
Acknowledgments
The Authors thank Prof. Enrico Fabrizi, DISES & DSS, Università Cattolica del S. Cuore, Piacenza, Italy for assistance with the statistical analysis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
