Abstract
Objectives:
To compare quantiferon-TB gold “in tube” (QFT-IT) with the conventional tuberculin skin test (TST) for the diagnosis of latent tuberculosis infection (LTBI) in HIV-infected adults in a setting highly endemic for tuberculosis with BCG vaccinated population in Bangalore, Karnataka, India.
Patients and Methods:
The study population (100 BCG-vaccinated adults) was divided into 4 groups: HIV patients with TB patient contact, HIV patients with past history of TB, TB patients (positive control), and healthy volunteers (negative control).
Results:
Overall agreement between TST and QFT-IT was 52.4% (κ = 0.22). Increasing the TST cutoff value from 5 mm to 10 mm among HIV-positive groups resulted in better agreement 62.5% in Group 1 and 81.2% in Group 2.
Discussion:
In a setting with high TB prevalence, the TST gives increased false positives in HIV-positive groups if 5 mm is used as the cutoff value. This could be minimized by increasing the cutoff to 10 mm.
Conclusion:
The 2-step approach (initial testing of all cases with TST and confirmation of only those positive in TST by testing with QFT-IT) will be economical and help in treating LTBI cases in lower middle income countries like India.
Keywords
Introduction
Tuberculosis (TB) has experienced a rising trend during the last few decades mostly attributed to the effects of newly emerging diseases, namely HIV infection. 1 HIV is the strongest risk factor for developing TB disease in those with latent or new mycobacterium tuberculosis (MTB) infection. The risk of developing TB is between 20 and 37 times greater in people living with HIV than among those who do not have HIV infection. 2
Latent tuberculosis infection (LTBI) detection is crucial in HIV-infected individuals because they have a higher rate of progression to active TB than in HIV uninfected persons even with the effective use of antiretroviral therapy (ART). 3 TB is a leading killer among people living with HIV. At least 1 in 4 deaths among people living with HIV can be attributed to TB, and many of these deaths occur in resource-limited settings. 4
India is the highest TB-burden country accounting for one-fifth (21%) of the global incidence. In 2009, of the estimated global annual incidence of 9.4 million cases, 2 million cases were estimated to have occurred in India. India being the third highest HIV-burden country had an estimated 2.39 million people living with HIV/AIDS in 2009 and the state of Karnataka stands second among the worst 6 affected states. 5 With HIV pitching in, TB incidence levels in India could go up; hence, the task of controlling the dual epidemic of TB and HIV remains a major challenge for the country.
Identifying LTBI aims at identifying individuals who would benefit from the treatment, preventing future development of active TB disease. The challenge of identifying LTBI-infected individuals lies in the lack of a diagnostic gold standard for LTBI. 6
There are currently 2 diagnostic methods that support the diagnosis of LTBI, the tuberculin skin test (TST) and interferon gamma (INF-γ) release assays (IGRAs). Both the tests are immunological methods that detect an immune response to antigens and consequently do not allow a direct measure of persistent infection. The in vivo TST is based on the intracutaneous injection of MTB antigens and subsequent identification of an immune reaction at the site of injection.
7
Unfortunately, the TST has low diagnostic specificity because purified protein derivative (PPD) contains antigens that are shared by many mycobacterial species.
8,9
Thus, a positive result is not necessarily associated with MTB infection, but may also be caused by immune cross-reactions in individuals vaccinated with the BCG-attenuated strain of
The sensitivity of the TST, which is still the most widely used test for the identification of individuals with latent infection with
Quantiferon-TB gold “in tube” (QFT-IT) test is a new third-generation whole-blood IGRA, which is now used in many countries.
However, the performance of QFT-IT is not well established in detecting LTBI in HIV-infected people and a small number of studies have been conducted on this subject to date. Yet, the little available data on the utility of QFT-IT is from studies carried out in different settings and is still controversial. 18,19 According to the World health Organization (WHO) guidelines published in 2011, the use of IGRAs to identify latent TB in persons living with HIV are restricted to studies conducted in low-TB prevalence settings. 2
Hence, carrying out more investigations on the performance of QFT-IT in persons with impaired immune systems, including persons with HIV/AIDS is declared as an important research need in a scenario of high prevalence of TB.
To better understand IGRAs use in HIV infection, we compared the performance of QFT-IT with conventional TST for the diagnosis of LTBI in a BCG-vaccinated urban HIV-infected population in Bangalore, Karnataka, India.
The purpose of our study was to (1) detect the prevalence of LTBI among BCG-vaccinated urban HIV-infected population, (2) assess the performance of QFT-IT among HIV-infected persons, and (3) check the degree of agreement between QFT-IT and TST for LTBI detection among HIV-infected population.
Materials and Methods
From January 2011 to June 2011, HIV-positive adults attending the ART outpatient department clinic of Kempegowda Institute of Medical Sciences, Bangalore were enrolled for the study. Patients were consecutively recruited and were enrolled after informed consent during the course of the routine examinations. The study program was approved by the institutional ethical committee and confidentiality of all the enrolled patients was maintained.
All patients completed a detailed questionnaire regarding their age, BCG vaccination, risk factors for exposure to TB, previous TST (date and result), history of household contact with a bacteriologically confirmed adult pulmonary TB case within the preceding 3 months, history of prior active TB, chest radiography, current pregnancy status, antiretroviral (ARV) medication history, or history of usage of systemic steroids or other immunosuppressive agents within 3 months or chemotherapy within 1 year of study entry.
Individuals aged above 18 years who were not pregnant and had not received systemic steroids or other immunosuppressive agents within 3 months or chemotherapy within 1 year of the study entry were eligible to participate.
Ongoing treatment for TB, prior severe reaction to TST, previous documented positive TST, positive chest radiography, and current pregnancy were the exclusion criteria. (All the HIV-positive individuals in groups 1 and 2 were on ART.)
Interferon Gamma Assay (QFT-IT)
The QuantiFERON-TB gold “in tube” (QFT-IT) test (Cellestis Ltd, Australia) a third-generation assay was carried out at the clinical microbiology laboratory of the KIMS Hospital, according to the manufacturer’s instructions by a trained technician who was blinded to the patient’s medical history and TST results. First, 1 mL of blood was drawn by venipuncture directly into 3 vacutainer tubes coated with saline (negative control), peptides of ESAT-6, CFP-10, and TB 7.7, or phytohemagglutinin (PHA; positive/mitogen control), and the tubes were incubated for 16 to 24 hours at 37°C. Then the tubes were centrifuged at 2000 to 3000 rpm for 15 minutes. The supernatant was frozen at −20°C till the performance of the enzyme-linked immunosorbent assay (ELISA) test. Gamma interferon release in each tube was measured via ELISA, and the obtained data were analyzed in SPSS 11.5 software (measurements were read in QFT software). Indeterminate results, defined as a negative result in the positive control tube (mitogen control), were identified. Indeterminate QFT-IT results were excluded from agreement analysis.
The TST
After collection of the blood samples for the QFT-IT assay, the TST was performed on the flexor surface of the left forearm about 4 in below the elbow joint with 1 tuberculin unit (TU) of tuberculin PPD (Span Diagnostics, Kothari Distributors, Bangalore) using a sterile tuberculin syringe fitted with a short 26-gauge needle. The TST results were read 48 to 72 hours after placement, and the maximum transverse diameter of induration was recorded using ball point pen method. Positive TST results were defined as an induration of 5 mm or larger for HIV-positive groups 1 and 2 and 10 mm or larger for HIV-negative groups 3 and 4. 20
Skin test anergy was defined as a TST reaction less than or equal to 2 mm in diameter. 21
Results
Study Population
The patients were enrolled into the study over a 6-month period from January to June 2011. A total of 80 consecutive HIV-positive patients were enrolled for the study. In all, 60 of them with previous history of contact with a known case of pulmonary TB were enrolled in group 1 and 20 with a past history of TB (treated more than 3 months back) were enrolled in group 2. Groups 3 and 4 with HIV-negative participants served as the control group with 10 cases each. Group 3 included cases of pulmonary TB whose sputum was positive in direct smear for Acid-fast bacilli (positive control) and in group 4 were healthy individuals (negative control).
All the 100 participants recruited for the study had blood samples drawn and received both the TST and the QFT-IT assays. In all, 10 participants from group 1 and 4 participants from group 2 failed to return for the TST reading and were therefore excluded from the study. Two participants from group 1 had indeterminate QFT-IT and nonreactive TST (0 mm) and were also excluded from the study. Therefore, the study analysis was based on a final sample size of 84 individuals from the original participant pool (Figure 1).

Study flow diagram.
The median age was 34 years, and 52% were women. The median CD4 count was 312 cells/mm3.
The TST
A total of 86 cases had valid TST results. Of these, 54 (64.3%) had positive TST results, with a median induration of 7 mm. Of the participants in groups 1 and 2 with CD4 counts of more than 350 cells/mm3, 60.3% (20 of 33) had positive TST results, compared to 55.5% (15 of 27) with CD4 counts of 101 to 350 cells/mm3 and 33.3% (2 of 6) with CD4 cells of ≤100 cells/mm3 (Table 1).
QFT-IT and TST Results According to CD4 Counts in Groups 1 and 2 (n = 64 + 2 QFT Indetermimates).
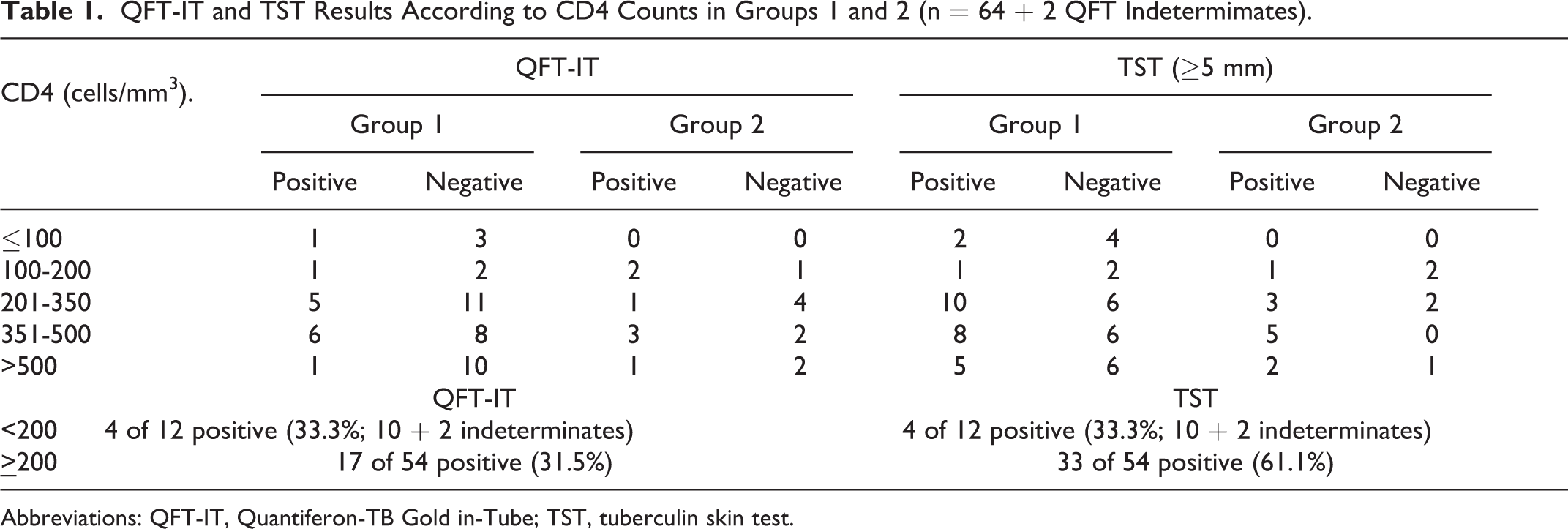
Abbreviations: QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
Agreement between QFT-IT and TST (≥5 mm).

Abbreviations: CI, confidence interval; QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
The QFT-IT
The QFT-IT testing yielded positive results in 33% (33 of 100) of the patients, negative results in 64% (64 of 100) of the patients, and indeterminate results in 3% (3 of 100) of the patients (Figure 1). All indeterminate results were due to a failure to respond adequately to the positive control. All of these 3 cases had CD4 counts less than 50 cells/mm3 (11, 18, and 35 cells/mm3).
Of the QFT-IT results in groups 1 and 2, 33.3% (11 of 33) of the patients had positive QFT-IT results when the CD4 counts were more than 350 cell/mm3, 33.3% (9 of 27) had positive QFT-IT when CD4 counts were 101 to 350 cell/mm3, and 14.3% (1 of 7) had positive QFT-IT when CD4 counts were less than 100 cells/mm3 (3 cases showed indeterminate results in this group;
Agreement between TST and QFT-IT
Excluding those with indeterminate QFT-ITs and/or TSTs not returned for interpretation, 84 had valid QFT-IT and TST results. Of these, 22 (26.2%) were in agreement with their negative results and 22 (26.2%) were in agreement with their positive results, for an overall agreement of 52.4% (κ = 0.22). (Of patients without valid TST results, 21.4% [3 of 14] had positive QFT-IT results and 7.1% [1 of 14] had indeterminate QFT-IT results [Figure 1].)
For all discordant results, patients were more likely to be TST positive and QFT-IT negative 38% (32 of 84) versus TST negative and QFT-IT positive 9.5% (8 of 84).
Overall, 64.2% of all patients had a positive TST result and 35.7% had a positive QFT-IT assay result.
The mean TST was 11.5 mm (range 0-25) for the QFT-IT-positive group and 6.2 mm (range 0-15) for the QFT-IT-negative group. The QFT-IT was positive in all patients diagnosed with active TB (group 3). The percentage of positive QFT-IT was highest in persons with TST ≥15 mm (33.3%), and this was significant when compared to the groups with TST of 10 to 14 mm (30%), 5 to 9 mm (10%), and <5 mm (23.3%;
Discussion
With regard to National immunization program and according to personal histories, all patients in our study had BCG vaccination in their childhood.
The clinical effect of NTMs on TST is normally minimal, except in parts of the world where TB prevalence is low and NTMs are more common due to moist climate. Also, very small effects of BCG on TST reactions are expected after 10 years for people vaccinated in infancy. 22 Hence, in a high TB endemic country with BCG vaccination in infancy, the TST is not likely to be affected by NTMs and BCG vaccination in an adult population. Nevertheless, lack of a diagnostic gold standard for LTBI emphasizes the need for follow-up of individuals with TST positive and QFT negative results.
Also, the lack of a gold standard for the detection of latent MTB infection complicates the direct comparison of IGRAs and the TST. 23
Overall agreement between TST and QFT-IT was 52.4% excluding indeterminate QFT-IT results. However, several studies in immunocompetent patients have revealed an agreement varying from 53% to 94%. 24 –29
Patients with a history of previously treated TB had a high rate of positive QFT-IT test results (44%) compared to those with history of household contact with patients having TB (28%).
There is a relevant concern that the performance of the QFT test, in line with the TST, could be impaired by low sensitivity in patients with advanced immunodeficiency. 30 The PHA control (positive control) serves as a surrogate marker for anergy as well as a quality control of the assay. In patients with a CD4 count of <50 cells/mm3, we found indeterminate test results due to low PHA response, and there was a significant correlation between CD4 count and PHA-induced IFN-γ release. These findings support the hypothesis that the performance of the IFN-γ-based tests is impaired in patients with advanced immune suppression.
In vivo and in vitro anergy in HIV-positive individuals is well recognized 31 –36 and the reduced antigen response correlates proportionally with CD4 count (and is reversible during ARV treatment). 32,36
Significant impediments to global TB control include limitations of current diagnostic tests. 37
Until recently, the only tool available for detection of LTBI was the TST. Although the TST is useful in clinical practice, it has several known limitations, including variable specificity, cross-reactivity with BCG vaccine and NTM infection, and problems with reliability. 38,39 So, new tests are required for the identification of MTB-infected individuals especially in tropical environments. 40
The IFN-γ assays such as QFT-G utilizing ESAT-6 and CFP-10 as recombinant antigens 24,41 or mixture of overlapping peptides 26,42 have been shown to be more specific than TST. 26 The QFT has been studied in the general population in several countries including India, and some studies have shown that it is a better indicator of the risk of MTB infection than TST in a BCG-vaccinated population. 43,44
Several studies have demonstrated the disagreement between TST and QFT results especially the type that TST was positive but QFT was negative. A study from South Africa found that among those with large TST reaction (≥15 mm), approximately one-third were negative by QFT. 45 While in a study from India, 11% of the individuals with positive TST (at least 15 mm) were negative by QFT. 46 In a study from Iran, 43.18% were positive by TST but negative by QFT. 47 In our study, 34.5% were TST positive but negative by QFT-IT (when 5 mm was used as cutoff in HIV-positive groups 1 and 2). It is probably due to prior BCG vaccination. Such discordance could also be due to false positive TST or it is plausible that QFT-IT is less sensitive than TST in detecting LTBI. Besides, in high incidence areas there are several factors that might modulate immune responses such as malnutrition, BCG vaccination, helminthic, and tropical infections that impact the T-helper (Th)1/Th2 immune balance. 48 The other possibility is that the QFT, which relies on the presence of antigen-dependant immediate effector T cells, is detecting current or more active infection, while the TST, which can detect central memory T cell responses, is detecting past, dormant, or resolved infection. 49
Disagreement of the reverse type (TST negative but QFT-IT positive) has also been documented, but it is largely unexplained. 40,46,50
In our study, increasing the TST cutoff from 5 mm to 10 mm for HIV-positive patients with history of TB contact (group 1) resulted in better agreement between the TST and QFT-IT (Table 3). While increasing the TST cutoff from 5 mm to 15 mm for HIV-positive patients with history of old treated TB (group 2) resulted in better agreement between the TST and QFT-IT (Table 3). It suggested that QFT-IT was able to identify those with TST responses in this range that were truly infected with MTB.
Agreement between QFT-IT and TST Assay Results (n = 48) in Group 1 (10 TST—Not Returned, 2—Indeterminates).

Abbreviations: CI, confidence interval; QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
Agreement between TST and QFT-IT Results in Group 2 (n = 16).

Abbreviations: CI, confidence interval; QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
Agreement between TST and QFT-IT Results in Group 3 (n = 10).

Abbreviations: CI, confidence interval; QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
Agreement between TST and QFT-IT Results in Group 4 (n = 10).
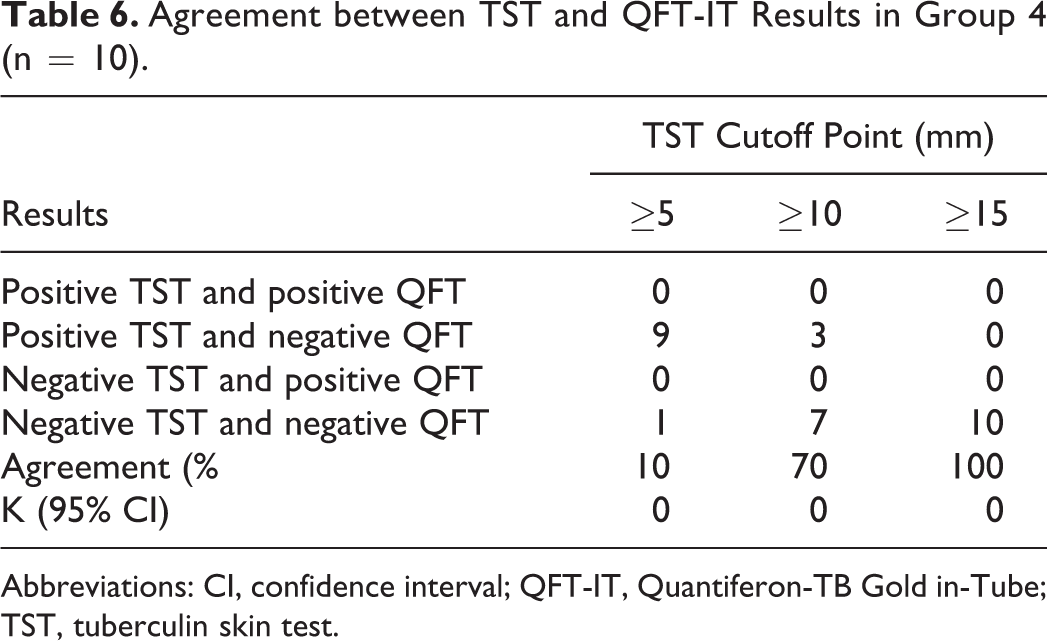
Abbreviations: CI, confidence interval; QFT-IT, Quantiferon-TB Gold in-Tube; TST, tuberculin skin test.
There is evidence suggesting that the sensitivity of IGRAs is higher than that of skin testing 51 –55 although large head-to-head studies in immunecompromised patients are lacking.
In our study, the TST sensitivity varied with the cutoff values chosen. In HIV-infected individuals, the 10 mm cutoff TST values correlated with the QFT-IT results in group 1, while 15 mm cutoff TST correlated with the QFT-IT in group 2 patients. With 5 mm cutoff which is advocated in HIV-infected population, the TST sensitivity was higher when compared to QFT-IT. The same observations were made in a large study conducted in the Netherlands among HIV-infected individuals. 56 They also reported that in HIV-infected patients the reduced sensitivity of the TST is primarily attributable to a “switch-off” of TST responsiveness (ie, an all-or-nothing phenomenon). The gradual decrease in reaction size with decreasing CD4 count was seen in limited cases. Nonetheless, a potential consequence of using 5 mm instead of 10 mm as the cutoff value for a positive TST result among HIV-infected patients is responsible for a limited reduction in false negative TST results but a much larger increase in false positive TST results. 56 This could explain the results in our study too.
The TST sensitivity and specificity is influenced by the cutoff used. A lower cutoff will result in a higher sensitivity and a lower specificity for MTB infection. The TST and IGRA results in general correlate poorly, mainly because of positive TST results in individuals vaccinated with BCG. In order to improve the specificity of test results, several national guidelines recommend initial TST screening and confirmation of a presumed infection with an IGRA. The 2-step approach (TST and confirmation of positive TST results by IGRA) will be economical and help in treating cases with LTBI . 17
Further guidelines for TB contact tracing should be based on the predicitive values of novel diagnostic tests to direct both the decision to test and the decision to treat. Most experts agree that the currently available IGRAs are superior to the TST in TB contact tracing. Novel test formats that include other antigens, additional markers, the comparative use of various patient specimens, and advanced techniques may soon replace today’s commercially available IGRAs for better diagnosis of true latent infection with MTB. Considering the limitations of the currently available assays, the main issue about testing is to restrict it to close contacts of an index case, to children and immune compromised individuals, and to those who are known to be at higher risk of developing TB and who are willing to accept treatment for latent infection with MTB. 17
The MTB-specific QFT-IT test offers methodological and logistic advantages over the TST. It requires only 1 patient visit and plasma can be frozen for later analysis. It does not induce a boosting phenomenon that is seen with the TST due to repeated injections of mycobacterial antigens. Once established in the laboratory, reproducibility is high. The number of steps resulting in direct contact with potentially contagious blood from HIV-positive individuals is minimal with the in-tube test system, thus reducing the occupational hazards of performing TB testing. By introducing the MTB-specific tests, we may be able to improve TB control by specific identification of those HIV-positive individuals with LTBI. A positive QFT-IT test is strongly suggestive of LTBI, whereas an indeterminate test result or a negative QFT-IT test result in the severely immune compromised hosts should always be interpreted with caution. 57
The performance of QFT-IT for detection of LTBI in immune compromised individuals has not been investigated in detail so far. In an ideal setting, large groups of patients with both known TST and QFT-IT results should be followed up for the development of TB. It would be preferable to do this in a setting with a low risk of reinfection during follow-up.
In analyzing severely immune compromised patients there is however a problem of cases giving negative TST and indeterminate QFT-IT results.
Conclusion
The QFT-IT assay showed good results for detecting latent MTB infection in BCG-vaccinated population (both HIV-positive and HIV-negative individuals). As QFT-IT is designed to be more specific than TST, perfect agreement is not expected. The TST and QFT-IT assay are comparable when the cutoff of TST is fixed at 10 mm for HIV-positive cases with previous history of contact and at 15 mm for HIV-positive cases who were previously treated for TB and also for the HIV-negative individuals.
The use of 1 TU dosage of PPD in our study could have contributed to the improvement in the TST specificity, as 2 TU dosage is used in many countries.
The TST and QFT-IT have different performance and operational characteristics. The QFT-IT has certain advantages such as the lack of interference by NTM or BCG vaccination, need for only a single patient contact, elimination of subjectivity both while administering the test or while reading the result, and the availability of test results within 24 hours. But QFT-IT is expensive, hence causes a constraint for its regular use in detection of LTBI in lower middle income regions. Hence, the selection of one test over the other will depend on the study population and resource availability.
We recommend in India where costly tests like QFT-IT cannot be employed for screening all HIV-positive patients, TST to be performed for all cases, followed by confirmation of presumed infection cases with QFT-IT.
There were certain limitations in our study: The sample size was small due to restricted use of the QFT-IT tests, which interferes with the interpretation of the results. Being consistent with the Indian policy, the use of 1 TU dose of PPD RT 23 limits our ability to compare our data with studies that have used 2 TU dosages. Also, the HIV patients attending the ART center were recruited, which may not exactly mimic the general population of HIV patients. Therefore, larger multicentric studies with patient recruitment from the general population are needed to compare the performance of the QFT-IT assay with TST for LTBI detection among HIV-positive individuals in TB high-endemic regions with BCG-vaccinated population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
