Abstract
Background:
Screening for latent tuberculosis infection using Interferon-Gamma Release Assays is a routine procedure prior to the initiation of anti-tumor necrosis factor (TNF) biotherapy or immunosuppressive therapy. However, indeterminate results are relatively frequent and are an obstacle to treatment initiation.
Aim:
The aim of this cross-sectional study was to estimate the frequency of indeterminate QuantiFERON-TB Gold Plus® test results in Tunisian patients, and to analyze the potential clinico-biological risk factors associated with these indeterminate results.
Methods:
Whole blood samples from 712 patients being monitored for autoimmune diseases and candidates for anti-TNF biotherapy or switch of immunosuppressive therapy were used to screen for latent tuberculosis infection using the QuantiFERON-TB Gold Plus® test. Based on literature background, the following variables were tested for the association with indeterminate results: gender, age, diabetes, immunosuppressive drugs, lymphocyte count, Neutrophil-to-lymphocyte ratio, serum albumin, and estimated glomerular filtration rate.
Results:
The QuantiFERON-TB Gold Plus® test was negative in 572 (80.3%) patients, positive in 106 (14.9%), and indeterminate in 34 (4.8%) cases. Positive results were significantly associated with a family history of confirmed and treated tuberculosis, OR (95% CI) = 52 (20.2–134.3). The use of immunosuppressive drugs and duration of treatment were significantly associated with the occurrence of indeterminate results: OR (95% CI) = 24.5 (5.8–103) and OR (95% CI) = 1.004 (1.002–1.007), respectively. Biologically, lymphopenia, hypoalbuminemia, and decreased estimated glomerular filtration rate were significant risk factors for indeterminate results: p = 5 E-6, p = 4.3 E-4, and p = 0.002, respectively. Thus, a multiple logistic regression model based on these three biological parameters enabled us to develop a predictive score for indeterminate results with a sensitivity of 91.2% and a specificity of 99.9%, AUC = 0.9964 (0.9917–1), p = 2.8 E-52.
Conclusion:
Immunosuppressive therapy, lymphopenia, hypoalbuminemia, and kidney failure appeared to be risk factors for indeterminate QuantiFERON-TB Gold Plus® results.
Introduction
According to the WHO, the worldwide annual incidence of tuberculosis (TB) is approximately ≈10 million. 1 This disease remains one of the leading causes of mortality, estimated at 1.6 million per year. 1 Despite major efforts in prevention and treatment, both worldwide by the WHO and locally by national programs, TB is still endemic in developing countries. 1 The difficulty lies in the presence of a large reservoir of latent tuberculosis infections (LTBI) affecting approximately a quarter (1/4) of the world’s population. 2 In adults, the risk of progression from LTBI to TB disease is 10%. 3 In children, on the other hand, the risk of progression is much greater, particularly during the first 2 years of life. 3
Screening for LTBI is therefore an essential prerequisite for a successful TB control program. One method of screening for LTBI is the tuberculin skin test (TST). However, TST lacks specificity, particularly in populations where BCG vaccination is routine. 4 To overcome the TST weaknesses, Interferon-Gamma Release Assays (IGRAs) such as QuantiFERON-TB and T-SPOT.TB have been developed. These tests are based on the ability of memory CD4 and CD8 T cells to produce IFN-γ following stimulation by mycobacterium tuberculosis (MTB)-specific antigens.
Nevertheless, these IGRA assays are subject to indeterminate results with a frequency that varies from 3% for T-SPOT.TB to 5% for QuantiFERON-TB.4,5 These indeterminate results are mainly linked to a defect in IFN-γ synthesis after mitogen stimulation.4,5 A recent meta-analysis revealed that, while the risk of progression to TB disease was half in the case of an indeterminate result compared with a positive result, it was three times greater in the case of an indeterminate result compared with a negative result. 6 Thus, these indeterminate results could be an impediment to the initiation of anti-TNF biotherapy or immunosuppressive therapy in patients with autoimmune diseases (AID). Besides, some factors including immunodeficiency and extreme age have been reported to be associated with the occurrence of indeterminate results of IGRAs. 4
Therefore, we sought in the present study to investigate to estimate the frequency of indeterminate QuantiFERON-TB Gold Plus® (QFT) test results in Tunisian patients and to identify potential clinico-biological risk factors associated with indeterminate IGRA test results to build practical predictive model. In addition, we aimed to build a predictive model for indeterminate results based on biological parameters.
Material and methods
This study was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies. 7
Patients
Training cohort
The required sample size was calculated using an online calculator (https://statulator.com/SampleSize/ss1P.html) to achieve 80% power with a significance level set at 0.05: Given that the frequency of indeterminate results in previous studies varied from 3% to 5%,4,5 the required sample size was 73 patients.
This cross-sectional study, conducted between January 2018 and March 2024, included 712 Tunisian patients followed and treated at Charles Nicolle Hospital (Tunis) for AID, including chronic inflammatory bowel disease (IBD: Crohn’s disease and ulcerative colitis), rheumatoid arthritis (RA), axial spondyloarthritis (AS), multiple sclerosis (MS), and psoriasis. All patients were candidates for anti-TNF biotherapy or immunosuppressive drug switching, and in whom screening for LTBI was required. Clinical and biological features of patients are depicted in Tables 1 and 2.
Clinical features of included patients.
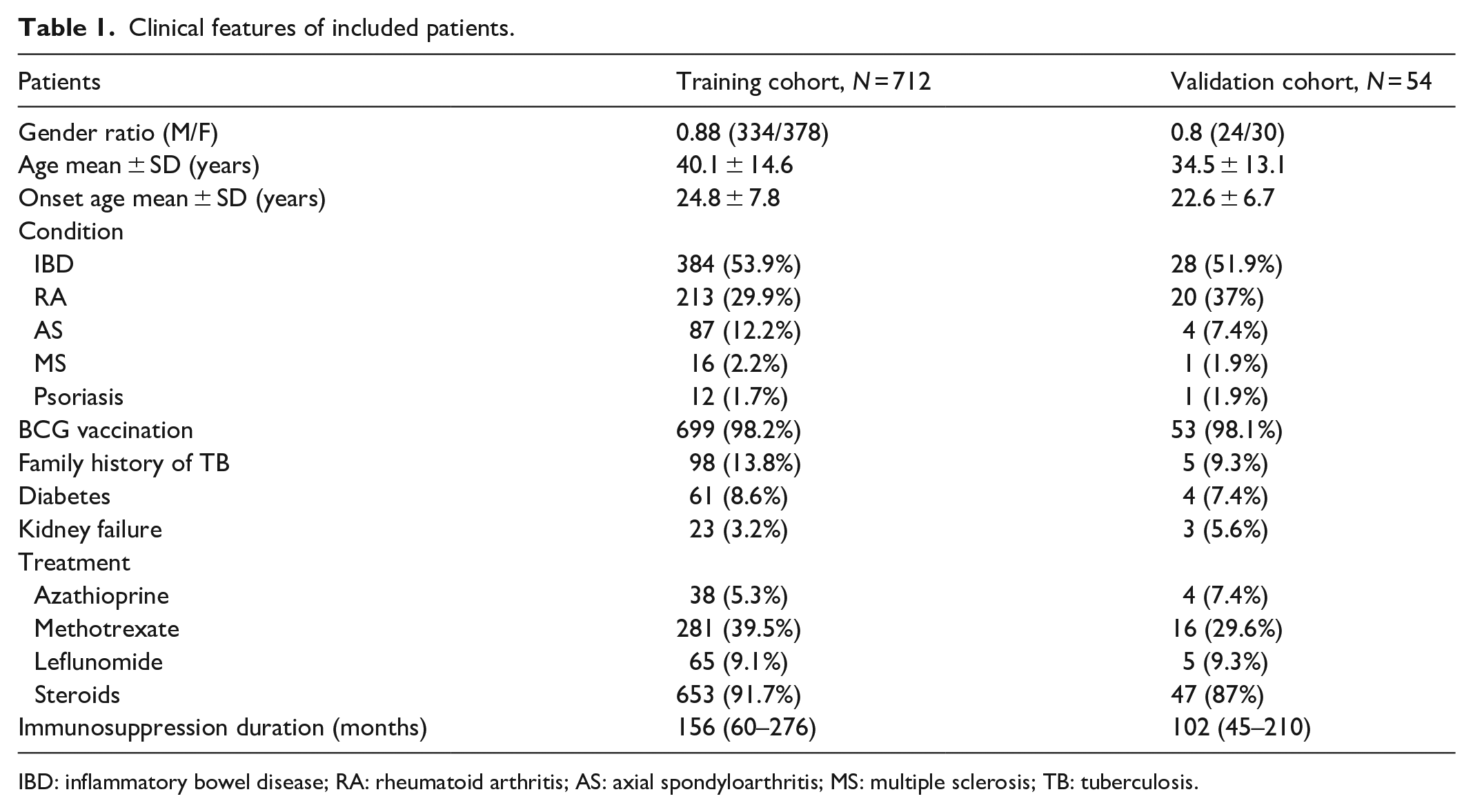
IBD: inflammatory bowel disease; RA: rheumatoid arthritis; AS: axial spondyloarthritis; MS: multiple sclerosis; TB: tuberculosis.
Biological features of included patients.
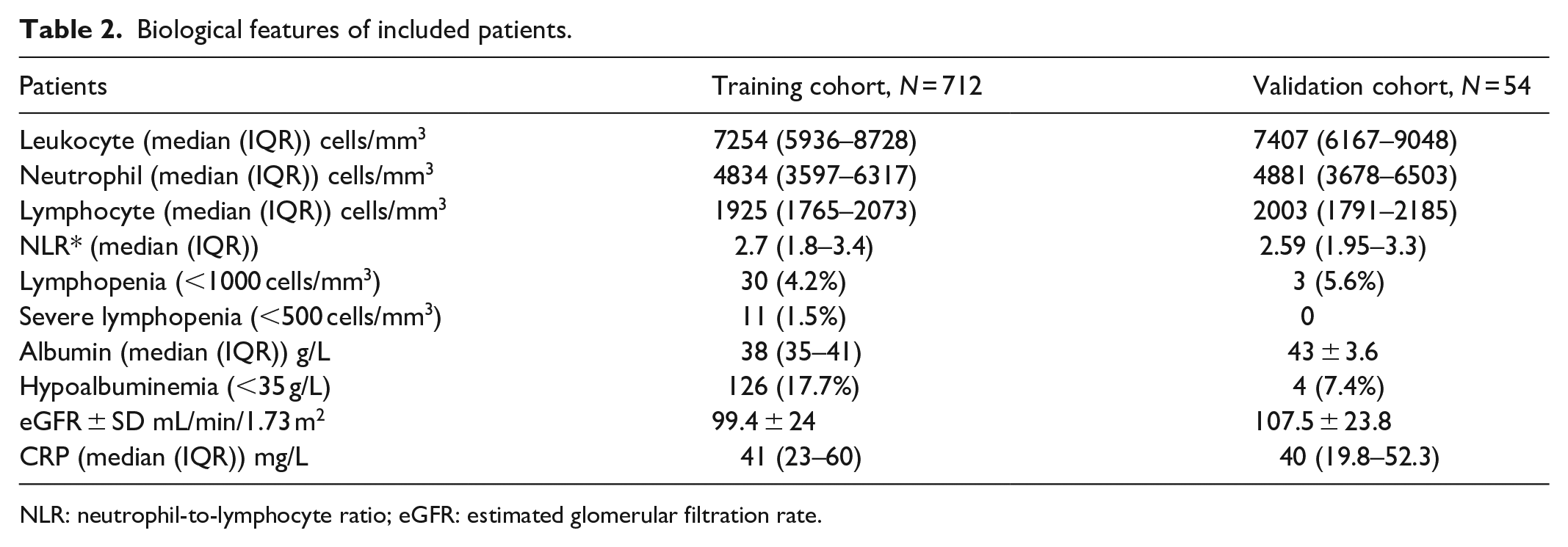
NLR: neutrophil-to-lymphocyte ratio; eGFR: estimated glomerular filtration rate.
Inclusion criteria:
• Patient candidate for anti-TNF biotherapy or immunosuppressive therapy.
Exclusion criteria:
• Suspicion of pulmonary or extra-pulmonary TB (intestinal, peritoneal, lymph node, tuberculous uveitis, etc.)
• Non-consenting patient.
• Mycobacterium TB present in sputum.
• X-ray images suggestive of TB.
Validation cohort
Additional 54 consecutive patients, followed by IBD and RA, were enrolled from the same hospital between 4 March and 12 July 2024, and were included as the validation cohort. These patients were selected based on the same criteria as the initial training cohort, with the aim of validating the predictive model of indeterminate results.
Quantification of released IFN-γ by the QFT assay
Patient blood samples were collected in the immunology department of Charles Nicolle Hospital in two tubes with heparin.
On receipt, 1 ml of whole blood was poured into each of the four tubes of the QFT kit:
• The first (Nil), containing only heparin, serves as a negative control and measures baseline IFN-γ release.
• The second (TB1), contains antigenic peptides from the BK proteins ESAT-6 and CFP-10, targeting CD4+ T lymphocytes (LT).
• The third (TB2) contains peptides from the ESAT-6 and CFP-10 proteins, targeting both CD4+ and CD8+ T lymphocytes.
• The last (Mit) contains a mitogen (phytohemagglutinin) which serves as a positive control, both to verify the functional integrity of the patient’s LT and to ensure that the blood samples have been handled and incubated correctly.
After an incubation time of 16–24 h, the tubes are centrifuged for 15 min at 2000–3000 rpm. The gel in the four tubes separates the cells from the supernatant plasma, which is collected for immediate use or stored at +4°C for up to 8 weeks, or below −20°C (down to −80°C) for longer period. Released IFN-γ was then quantified by indirect ELISA. Four standards, S1 (4 lU/ml), S2 (1 IU/ml), S3 (0.25 IU/ml), and S4 (0 UI/ml), which contain only the diluent, were used for IFN-γ quantification. For samples with high IFN-γ concentrations, a second analysis using eight standards (8, 4, 2, 1, 0.5, 0.25, 0.125, and 0) was performed to obtain accurate measurements of IFN-γ levels. The OD values measured are entered into the QFT_TBGoldPlus_v2.71.2 Build06 software (Qiagen, Hilden, Germany), which enables quality control of the reaction and subsequent conversion of these OD values into INF-γ concentrations expressed in (IU/ml) using the OD values of the four standards. When using eight standards for samples with high concentrations of IFN-γ, the QuantiFERON_RD_v5.03.0_Build08 software (Qiagen, Hilden, Germany) automatically generated calibration curves and inferred the concentrations of IFN-γ. According to the manufacturer’s recommendations, interpretation of the results requires consideration of the IFN-γ concentrations in the “Nil” and “Mit” tubes. The INF-γ concentration value in the “Nil” tube is subtracted from those in the three tubes TB1, TB2, and Mit.
Definition of indeterminate result
A QFT result was considered as indeterminate when:
• The concentration in the “Nil” tube is greater than 8 IU/ml.
• The concentration in the “Mit” tube is less than 0.5 IU/ml.
Study endpoints
The primary endpoint of this study was to determine the frequency of QFT indeterminate results in Tunisian patients. The secondary endpoint was to identify risk factors associated with the aforementioned indeterminate results. In addition, we sought to build a simple predictive model of indeterminate results based on biological parameters.
Statistical analysis
Statistical analysis was carried out using Statistical Package for the Social Sciences (SPSS) version 11 (IBM®, Armonk, USA) and GraphPad Prism 5 (Dotmatics®). p-values < 0.05 were considered significant.
Chi-square or Fisher exact tests were used to test the association between categorical variables. Odds ratio (OR) together with 95% confidence intervals (95% CI) were calculated to estimate the strength of the association. ANOVA, Mann–Whitney U, and Kruskal–Wallis tests were used to analyze quantitative variables as appropriate. Non-parametric tests (Mann–Whitney U and Kruskal–Wallis) were applied when dependent quantitative variables were not normally distributed (absolute values of skewness and/or kurtosis > 1, significant Kolmogorov–Smirnov and Shapiro–Wilk tests). To measure the strength of the relationship between quantitative variables, Pearson and Spearman (non-parametric) correlations were performed according to the type of distribution of the quantitative variables and the nature of the relationship (linear or non-linear). The effect size for the Pearson correlation was estimated using the r2.
In case of significant results in the univariate analyses, multivariable analyses using multiple linear regression and multiple logistic regression were performed:
• Multiple linear regression: this test was used to verify that the linear relationship between two quantitative variables is independent of the potential presence of other confounding cofactors. The absence of multicollinearity of the independent variables, the absence of multivariable extreme values (verified with Mahalanobis and Cook distances), and homoscedasticity (homogeneity of residual variances), via White’s test and the modified Breusch–Pagan test were checked beforehand. Effect size was estimated using r2.
• Multiple logistic regression: this test was used to check the impact of one or more independent variables on a qualitative dependent variable, and to detect possible confounding factors or interactions (effect-modifying factors). Variables with a p-univariate < 0.05, age, gender, variables not significantly associated but with a p-univariate > 0.2, and any interaction terms were included in the adjustment models. Effect size was measured using Nagelkerke’s pseudo-r2.
Besides, a multiple logistic regression model based on biological parameters was built to predict indeterminate results. To test the stability of the model, sensitivity analysis was performed through sequential weighting of logistic regression model by age, gender, disease type, immunosuppressive therapy duration, immunosuppressive drug use, and diabetes. The “generalized linear models” menu was used to weigh logistic regression predictive model.
A Supplemental file 1 with additional tables and figures is provided with this article. Patient QuantiFERON request and consent form is available as a Supplemental file 2. The STROBE checklist is depicted in Supplemental file 3.
Results
Patient characteristics
Since January 2018, 1473 patients, for whom QuantiFERON-TB test was requested, have been identified. Five hundred and eleven patients were excluded for incorrect sampling (n = 72), or for requests from private practitioners without clinical data (n = 439). Nine hundred and sixty-two patients were assessed for eligibility, of whom 11 were excluded for confirmed TB and 96 for non-consent. Finally, 766 patients were included after the exclusion of 89 cases for incomplete clinical and biological data (Figure 1). Included patients were then divided into a training cohort (n = 712) and a validation cohort (n = 54).

Flow diagram describing participants’ selection process.
A total of 712 patients followed and treated for AID were included in the training cohort. Mean age of patients was 40.1 ± 14.6 years with gender ratio (male/female) of 0.88 (334/378; Table 1). Most (98.2%) patients received BCG vaccination, and family history of TB was present in 98 (13.8%) cases (Table 1). Median duration of immunosuppressive therapy was 156 (60–276) months. Thirty (4.2%) patients had lymphopenia (<1000 cells/mm3) and severe lymphopenia (<500 cells/mm3) was observed in 11 (1.5%) cases (Table 2). Hypoalbuminemia (<35 g/L) was observed in 126 patients and mean eGFR was 99.4 ± 24 mL/min/1.73 m2 (Table 2).
Besides, lymphocyte count was significantly lower in the case of immunosuppressive drug use (Table S1 and Figure S1). Likewise, lymphocyte count was negatively correlated with age, and immunosuppressive therapy duration level, and positively correlated with serum albumin level and eGFR (Table S1 and Figure S2). Moreover, lymphopenia was significantly associated with age, diabetes, kidney failure, immunosuppression and its duration, albumin, and eGFR (Table S2). Furthermore, neutrophil-to-lymphocyte ratio (NLR) was significantly higher in patients with kidney failure and under immunosuppressive therapy (Table S3 and Figure S3). Similarly, NLR was significantly correlated with patient’s age, treatment duration, albumin, and eGFR (Table S3).
QFT results
The QFT test was negative in 572 (80.3%) patients, positive in 106 (14.9%), and indeterminate in 34 (4.8%) cases (Table 3). All indeterminate results were due to IFN-γ levels <0.5 IU/ml after stimulation with the mitogen. Besides, IFN-γ levels with TB1 and TB2 tubes were significantly and positively correlated, r = 0.974, p < 1 E-200 (Figure S4). Of note, positive results were significantly associated with patients’ age and family history of TB (Table S4).
QFT test results.
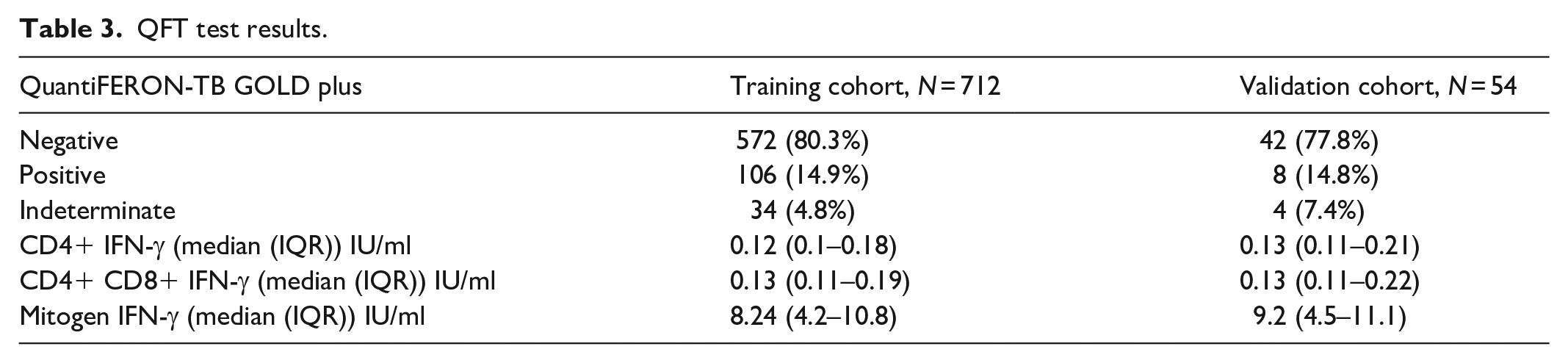
Factors associated with mitogen-induced IFN-γ level
Patients’ age was negatively correlated with mitogen-induced IFN-γ levels (p = 0.013), while the results showed no association with gender. IFN-γ levels after mitogen stimulation were significantly lower in hypertensive patients and those with renal failure: p = 0.0007 and p = 1 E-8, respectively (Table 4). Similarly, immunosuppressive treatments and corticosteroid therapy were significantly associated with lower mitogen-induced IFN-γ levels (Table 4 and Figure 2). Furthermore, immunosuppression duration was negatively correlated with IFN-γ levels, r = −0.139 (−0.210 to −0.066), p = 0.0002. Biologically, lymphopenia, albumin, and eGFR significantly influenced mitogen-induced IFN-γ levels (Table 4 and Figure 3). Multivariable analysis using multiple linear regression confirmed the association of kidney failure, immunosuppressive treatments, corticosteroid therapy, lymphopenia, albumin, and eGFR with variation in the level of IFN-γ released after mitogen (Table 4).
Factors associated with mitogen-induced IFN-γ level.

Significant associations/correlations are shown in bold.

Significant lower mitogen-induced IFN-γ levels in case of immunosuppressive drugs or steroid intake: (a) Azathioprine, (b) Methotrexate, (c) Leflunomide and (d) Steroids.

Pearson r correlation matrix including mitogen-induced IFN-γ level, age, and relevant biological parameters.
Factors associated with QFT indeterminate results
The frequency of indeterminate results was significantly higher in patients with renal failure, p = 1 E-14, OR (95% CI) = 52 (20.2–134.3) (Table 5). Similarly, indeterminate results were significantly more frequent in patients receiving immunosuppressive drugs and/or corticosteroids (Table 5). Interestingly, longer duration of immunosuppressive therapy was significantly associated with indeterminate QFT test results, p = 2.5 E-4 (Table 5). Regarding the biological markers, lymphopenia, NLR, hypoalbuminemia, and eGFR were significantly associated with indeterminate QFT test results (Table 5 and Figure 4). Thus, lymphocyte count, albumin level, and eGFR were significantly lower in case of indeterminate results (Table 5 and Figure 4). Besides, a ROC analysis was performed to estimate the performance of lymphocyte count in predicting indeterminate QFT results and revealed an area under the receiver operating characteristics (ROC) curve (AUC) of 0.983 (0.965–0.999), p = 2 E-21 (Figure 5). Sensitivities and specificities according to the different thresholds are summarized in Table 6.
Factors associated with QFT indeterminate results.

eGFR: estimated glomerular filtration rate. Significant associations are shown in bold.

Biological parameter associations with indeterminate QFT test results. (a) Significant lower lymphocyte count in case of indeterminate results. (b) Significant higher NLR in case of indeterminate results. (c) Significant lower albumin levels in case of indeterminate results. (d) Significant lower eGFR in case of indeterminate results.

ROC curve showing lymphocyte count determining performances in predicting indeterminate QFT results.
Lymphocyte count thresholds and corresponding performance in predicting indeterminate QFT test results.

significant associations/correlations are shown in bold.
Next, we built two multiple logistic regression models based on the use of immunosuppressive drugs and biological markers to avoid multicollinearity between lymphopenia and lymphocyte count, between hypoalbuminemia and albumin level, and between kidney failure and eGFR (Table 7). Multivariable analysis confirmed that kidney failure, immunosuppressive drug use, longer duration of immunosuppression, lymphopenia, hypoalbuminemia, and low eGFR were risk factors for indeterminate QFT results (Table 7).
Multivariate analysis for risk factors associated with indeterminate QFT results.

Regression predictive model for QFT test indeterminate results
The search for the most parsimonious fitting model based on biological parameters only for practical purposes showed that lymphocyte count, albumin, and eGFR together predicted indeterminate results with an accuracy of 99.6% (sensitivity = 91.2% and specificity = 100%), AUC = 0.9964 (0.9917 – 1), Nagelkerke R2 = 0.906, p = 2.8 E-52: Logit (indeterminate result) = − 0.009 * Lymphocyte count − 0.960 * Albumin − 0.022*eGFR + 46.343 (Figure 6). Subsequently, risk of indeterminate result can be obtained by exponentiating the Logit result (Table S5). Sensitivity analysis through weighting the logistic regression model showed identical trends with very similar β-coefficients regardless of the weighting variable (Table S6). Hence, sensitivity analysis for the predictive model of indeterminate results revealed stable and reliable results.

Multiple logistic regression model for indeterminate result prediction. (a) ROC curve assessing the performances of the fitting model. (b) Predicted versus observed plot: the model predicts an indeterminate result when the probability is ⩾50%.
Validation cohort for the predictive model of indeterminate results
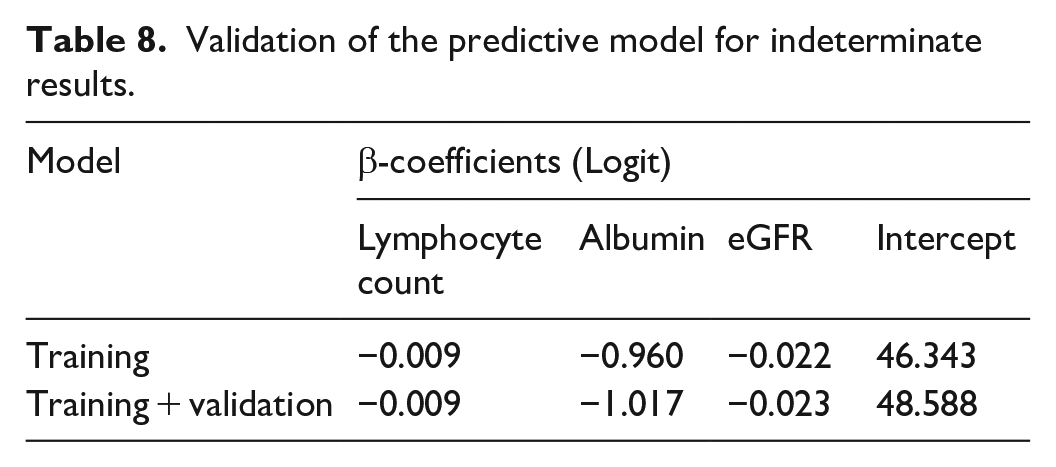
The validation cohort consisted of 54 consecutive patients followed for AID. Clinical and biological features of these 54 patients are shown in Tables 1 and 2. Forty-two (77.8%) patients had a negative QFT test, eight (14.8%) were positive, and four (7.4%) patients had an indeterminate result (Table 3). The formula obtained from the training cohort was tested using data from the validation cohort. Risk for each patient was obtained by exponentiating the Logit formula result. Probability of indeterminate result was obtained through the following formula: computed risk/1 + computed risk. All patients with a determinate result had probabilities below 0.5. For the four patients with indeterminate results, probabilities ranged from 0.93 to 1. Hence, when tested on the validation cohort, the predictive model had a sensitivity and a specificity of 100%. Then we merged the two cohorts (training + validation) to verify the stability of the model coefficients. As shown in Table 8, β-coefficients were very analogous, which suggested that the predictive model is consistent and yields reliable results.
Validation of the predictive model for indeterminate results.
Discussion
Screening patients for LTBI remains mandatory prior to starting a biotherapy with anti-TNF drugs. In fact, accurate LTBI diagnosis is critical to prevent TB reactivation through chemoprophylaxis. 8 In this regard, IGRA tests, namely T-SPOT.TB and QuantiFERON-TB, have been developed to overcome the low sensitivity and specificity of the TST. These IGRA are now being widely used in routine for LTBI screening. One of the main drawbacks of IGRA tests is indeterminate results that can occur in 3% of cases for T-SPOST.TB and 5% of cases for QuantiFERON-TB.4,5 In addition, data on clinical and biological risk factors for indeterminate QFT tests are still inconclusive and need to be extensively investigated.
In the present study, 712 patients diagnosed with AID and screened for LTBI by QFT assay were enrolled in the training cohort. The QFT test was negative in 572 (80.3%) patients, positive in 106 (14.9%), and indeterminate in 34 (4.8%). These relative frequencies corroborate those of the majority of previous studies and a subsequent meta-analysis. 4 Of note, another recent meta-analysis published in 2023 and which included 403 studies, reported that while the pooled frequency of indeterminate results for all types of IGRAs of all generations was 3.9%, the risk of indeterminate results with the new generation of QFT (Gold Plus) was four times lower than with TSPOT.TB and the lowest across all generations of QFT. 5 Hence, there might be a potential advantage to using the newest generation of QFT (Gold Plus), owing to its lower indeterminate result rate. 5
In this study, patient age was not associated with indeterminate QFT result risk. This result corroborates those of the majority of previous studies.9–13 Conversely, extreme ages (<18 years and >65 years) were associated with indeterminate results in 3263 English patients. 14 Likewise, in another study in 77 Korean patients with suspected TB, age over 65 years was associated with an 11.29-fold increase in the risk of indeterminate results. 15 However, two recent meta-analyses4,5 showed no association between advanced patient age and indeterminate IGRA test results. Hence, it is possible that the risk of indeterminate results conferred by age is low, which explains the absence of association in the majority of published studies. In addition, small numbers of patients with extreme ages may have reduced statistical power.
In this study, immunosuppressive therapy use was significantly associated with indeterminate outcomes in both univariable and multivariable analyses. This result is consistent with those of previous studies.10,15–25 Hence, these data indicate that immunosuppression significantly increases the risk of indeterminate IGRA results. Consequently, discontinuation of immunosuppressive therapy for a few weeks may reduce the risk of indeterminate results.
Besides, given that the majority (91.7%) of our patients were under steroid treatment, the analysis showed no association between steroid use and indeterminate QFT test results. Unfortunately, data on steroid doses were not available for our patients, which prevented us from further stratification. The impact of steroid therapy on IGRA performance has been disparately estimated. In a retrospective study of 2037 immunocompetent Korean children, the use of corticosteroids was significantly associated with indeterminate results. 26 Similarly, corticosteroid therapy and anti-TNF biotherapy were predictive of indeterminate outcomes in 400 American IBD patients. 11 In another American study, while high-dose corticosteroid therapy (>20 mg/kg/day) was associated with the risk of indeterminate results, low-dose corticosteroids were not. 12 Conversely, in a Korean study of 401 patients with chronic inflammatory rheumatism corticosteroids were not associated with indeterminate QFT test results through a multiple logistic regression model. 9 These divergent results can be explained by: (1) large variations in corticosteroid doses, (2) association of steroid use with immunosuppressive drugs, which could act as a confounding factor, and (3) the inner architecture of multivariable adjustment models as the incorporation of two collinear variables, such as corticosteroid intake and lymphopenia, is likely to hide the association of one or the other with indeterminate results.
In our patients, lymphopenia (<1000 cells/mm3) was associated with approximately an 85-fold increase in the risk of indeterminate QFT test results. Similarly, lymphocyte count and NLR were significantly associated with indeterminate result outcome. This has also been reported in previous studies.9–12,14–19,26–30 Thus, these results indicate that lymphocyte count is one of the main predictors of indeterminate QFT test results. Subsequent ROC analysis in our patients revealed that for a threshold of 1169 cells/mm3, sensitivity and specificity of lymphocyte count in predicting indeterminate results were 88.2% (73.4–95.3) and 100% (99.4–100), respectively. Therefore, determining lymphocyte count seems to be a necessary prerequisite prior to performing the QFT test.
In the present study, low albumin level (<35 g/L) was associated with a 7.7-fold (3.8–15.7) increase in the risk of indeterminate QFT test results. Thus, an increase of 1 g albumin/L reduced the risk of an indeterminate result by 47.7%. These results are in line with previous reports.9,11,16–19,29,30 Low albumin and total protein levels, which might reflect a hepatic dysfunction, might reduce immune cell activation and IFN-γ synthesis. 18 Nevertheless, pathophysiological evaluation is needed in the future to better understand the link between low albumin levels and indeterminate QFT results.
In our patients, kidney failure was associated with a 52-fold (20.2–134.3) increase in the risk of indeterminate results. In addition, a 1 mL/min/1.73 m2 decrease in eGFR increased the risk of the occurrence of an indeterminate result by 3.1%. These associations were confirmed by multivariable analysis. Only one study 9 investigated the impact of kidney function on indeterminate outcomes. However, this study did not show any significant difference. Thus, our peculiar finding requires replication on large independent cohorts in the future.
In the present study, the most parsimonious adjustment model was obtained by multiple logistic regression, including only biological parameters retained lymphocyte count, albumin, and eGFR. The calculation of the risk of an indeterminate result is based on the following formula: Logit (indeterminate result) = − 0.009 * Lymphocytes − 0.960 * Albumin − 0.022 * eGFR + 46.343. Subsequently, risk of indeterminate result can be obtained by exponentiation of the Logit result. This predictive model could be used to predict the risk of an indeterminate result prior to IGRA testing. Sensitivity analysis through sequential weighting of the predictive model confirmed the stability of the model under diverse conditions. Subsequent validation of the predictive model in a cohort of 54 patients showed specificity and sensitivity of 100%. Moreover, after merging the training and validation cohorts, the β-coefficients obtained were very similar to those of the original predictive model, indicating that it is consistent and reliable. However, in the future, a larger validation cohort might better assess its performance and refine β-coefficient estimation as the number of patients increases.
Besides, in our patients, the levels of IFN-γ released after mitogen stimulation were significantly lower in the case of immunosuppressive drugs and steroids use, and in patients with lymphopenia. These findings corroborate the data found in the scientific literature.13,14,16,27,28 Furthermore, our results revealed significant correlations of lymphocyte count, albumin, and eGFR levels with IFN-γ mitogen levels. These unprecedented findings require replication in large independent cohorts.
The present study has some limitations that need to be addressed. First, our study is monocentric; a multicentric study could have provided more representative results for the Tunisian population. Second, given the absence of a “GOLD STANDARD” for the diagnosis of LTBI, a prolonged follow-up of patients and a 3-month check-up would have enabled us to estimate the sensitivity of the QFT test. Third, the number of indeterminate results was relatively low (n = 34), which may have reduced statistical power. Thus, increasing the number of patients could help refine the predictive model for indeterminate QFT results. Fourth, we had no data on alcohol use which could have influenced the risk of indeterminate results. Fifth, the validation cohort was relatively small (n = 54), and only four patients had indeterminate QFT results. Therefore, a larger validation cohort is needed in the future to better assess the predictive model performance and refine β-coefficient estimation.
In summary, (1) immunosuppressive drug intake seems to be associated with an increased risk of indeterminate QFT results, (2) regarding the biological parameters, lymphopenia, hypoalbuminemia, and low eGFR were significant risk factors for indeterminate QFT results, and (3) the level of IFN-γ released following mitogen stimulation was influenced by immunosuppressive and/or corticosteroid therapy, lymphopenia, hypoalbuminemia, and low eGFR.
Conclusions
Immunosuppressive therapy, lymphopenia, hypoalbuminemia, and kidney failure appear to be risk factors for indeterminate QFT results. Discontinuation of immunosuppressive therapy for a few weeks may reduce the risk of indeterminate results.
Supplemental Material
sj-doc-3-smo-10.1177_20503121241279116 – Supplemental material for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients
Supplemental material, sj-doc-3-smo-10.1177_20503121241279116 for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients by Rahma Bellagha, Tarak Dhaouadi, Awatef Riahi, Wahiba Ben Rehouma, Hajer Jedidi, Leila Mouelhi, Leila Abdelmoula, Taïeb Ben Abdallah, Yousr Gorgi and Imen Sfar in SAGE Open Medicine
Supplemental Material
sj-docx-1-smo-10.1177_20503121241279116 – Supplemental material for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients
Supplemental material, sj-docx-1-smo-10.1177_20503121241279116 for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients by Rahma Bellagha, Tarak Dhaouadi, Awatef Riahi, Wahiba Ben Rehouma, Hajer Jedidi, Leila Mouelhi, Leila Abdelmoula, Taïeb Ben Abdallah, Yousr Gorgi and Imen Sfar in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241279116 – Supplemental material for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients
Supplemental material, sj-docx-2-smo-10.1177_20503121241279116 for A regression predictive model for QuantiFERON-TB Gold Plus® indeterminate results in immunosuppressed patients by Rahma Bellagha, Tarak Dhaouadi, Awatef Riahi, Wahiba Ben Rehouma, Hajer Jedidi, Leila Mouelhi, Leila Abdelmoula, Taïeb Ben Abdallah, Yousr Gorgi and Imen Sfar in SAGE Open Medicine
Footnotes
Author contributions
Conceptualization: Yousr Gorgi, Taïeb Ben Abdallah, Imen Sfar, Leila Mouelhi, and Leila Abdelmoula. Data curation: Rahma Bellagha, Tarak Dhaouadi, and Awatef Riahi. Formal analysis: Tarak Dhaouadi. Funding acquisition: Yousr Gorgi, Taïeb Ben Abdallah, and Imen Sfar. Investigation: Rahma Bellagha, Tarak Dhaouadi, Awatef Riahi, and Imen Sfar. Methodology: Tarak Dhaouadi, Imen Sfar, and Yousr Gorgi. Project administration: Yousr Gorgi, Taïeb Ben Abdallah, and Imen Sfar. Resources: Yousr Gorgi, Taïeb Ben Abdallah, and Imen Sfar. Supervision: Yousr Gorgi, Taïeb Ben Abdallah, Imen Sfar, Leila Mouelhi, and Leila Abdelmoula. Writing – original draft: Tarak Dhaouadi. Writing – review & editing: Tarak Dhaouadi.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article (and/or) its supplementary materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was granted and supported by the Research Laboratory in Immunology of Renal Transplantation and Immunopathology (LR03SP01), Charles Nicolle Hospital, Tunis El Manar University, Tunisia.
Ethics approval and consent to participate
All patients and controls gave written informed consent to participate in the study, and the local ethics committee of Charles Nicolle Hospital approved this study (Approval Number: HCN_2024_18; April 23, 2024. FWA00032748, IORG0011243). No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
