Abstract
Cardiac metastases, particularly those originating from non-Hodgkin lymphoma, are often underdiagnosed and represent a rare but critical manifestation of advanced cancer. This case report discusses a 57-year-old female with a history of hypertension who initially presented with symptoms of cardiac tamponade, leading to the discovery of a large cardiac mass, ultimately diagnosed as diffuse large B-cell lymphoma following a cytological examination of the pericardial fluid and lymph node biopsy. Further imaging, including echocardiography and computed tomography scans, showed extensive metastasis to the pericardium, mediastinum, and other areas, such as the thyroid and peritoneum. Despite the prompt initiation of management, the patient suffered a massive pulmonary embolism, highlighting the poor prognosis associated with metastatic cardiac lymphoma. This case underscores the importance of bedside echocardiography as a rapid initial method for identifying cardiac tamponade, as well as maintaining a high index of suspicion for hematologic malignancies in patients presenting with unexplained pericardial effusions. It emphasizes the need for comprehensive diagnostic approaches and multidisciplinary management to improve patient outcomes in this rare and challenging clinical scenario.
Keywords
Introduction
Cardiac metastases remain an understudied aspect of oncology, with reported incidence rates ranging from 2.3% to 18.3% in postmortem studies, highlighting inconsistencies in detection and reporting practices. 1 Lymphoma, particularly non-Hodgkin lymphoma (NHL), is known to frequently metastasize to the heart, though such manifestations are often subclinical or go undetected until advanced stages.2,3 In this case report, we present a female patient who initially showed symptoms of cardiac tamponade, which led to the discovery of a large cardiac mass that was ultimately diagnosed as diffuse large B-cell lymphoma (DLBCL).
Case Presentation
Our patient is a 57-year-old Asian female with a past medical history of hypertension who presented to the emergency department with a constellation of symptoms, including chest pain, shortness of breath, progressive exertional dyspnea, and lower extremity edema. She denied any history of alcohol or drug abuse and had no known allergies or adverse reactions. The patient underwent a comprehensive series of tests, including a chest X-ray and an electrocardiogram, which revealed slight left and right pleural effusions. She was promptly admitted for further evaluation and management.
In the emergency department, the patient’s initial vital signs included a blood pressure (BP) of 80/60 mmHg, heart rate of 112 beats/minute, respiratory rate of 18 breaths/minute, and oxygen saturation (SpO2) of 98% on room air. Labs were significant for B-type natriuretic peptide 774 pg/mL (reference range <100 pg/mL), troponin I 61 ng/L (reference <14 ng/L), blood urea nitrogen 43 mg/dL (reference 7-20 mg/dL), creatinine 1.44 mg/dL (reference 0.6-1.1 mg/dL), and elevated liver transaminases. The physical examination indicated 3+ bilateral lower extremity edema, mild respiratory distress, and left cervical lymph node enlargement. Bedside echocardiography was performed, revealing a large pericardial effusion with cardiac tamponade physiology and a large mass invading and compressing the right heart, confirming cardiac tamponade (Figure 1).

(A, B) Pre-drainage 4-chamber bedside echocardiography showing sizeable pericardial effusion with physiological tamponade (indicated by the arrows) and tumor invading right ventricular and right atrium collapsing during diastole. (C, D) Pre-drainage short-axis bedside echocardiography showing sizeable pericardial effusion with physiological tamponade and tumor invading and compressing the right ventricular.
Cardiothoracic surgery (CTS) was urgently consulted, and an emergent pericardial window was performed, removing ~700 cc of pericardial fluid. Samples were sent for culture and cytological examination, which revealed positive results for malignant cells consistent with involvement by CD5-positive large B-cell lymphoma, primarily composed of large B cells. After the procedure, the patient was transferred to the Cardiac Care Unit, and subsequently, the oncology team was consulted.
Follow-up transthoracic echocardiography (TTE) revealed a normal left ventricular ejection fraction of 55% to 60% with concentric hypertrophy and mildly increased wall thickness. The right ventricular free wall appeared infiltrated by a mass. The cavity appears to contain a mass extending from the moderately dilated right atrium. It also identified a large pericardial mass and a mass in the right atrium, with a mild pericardial effusion after the procedure (Figure 2).
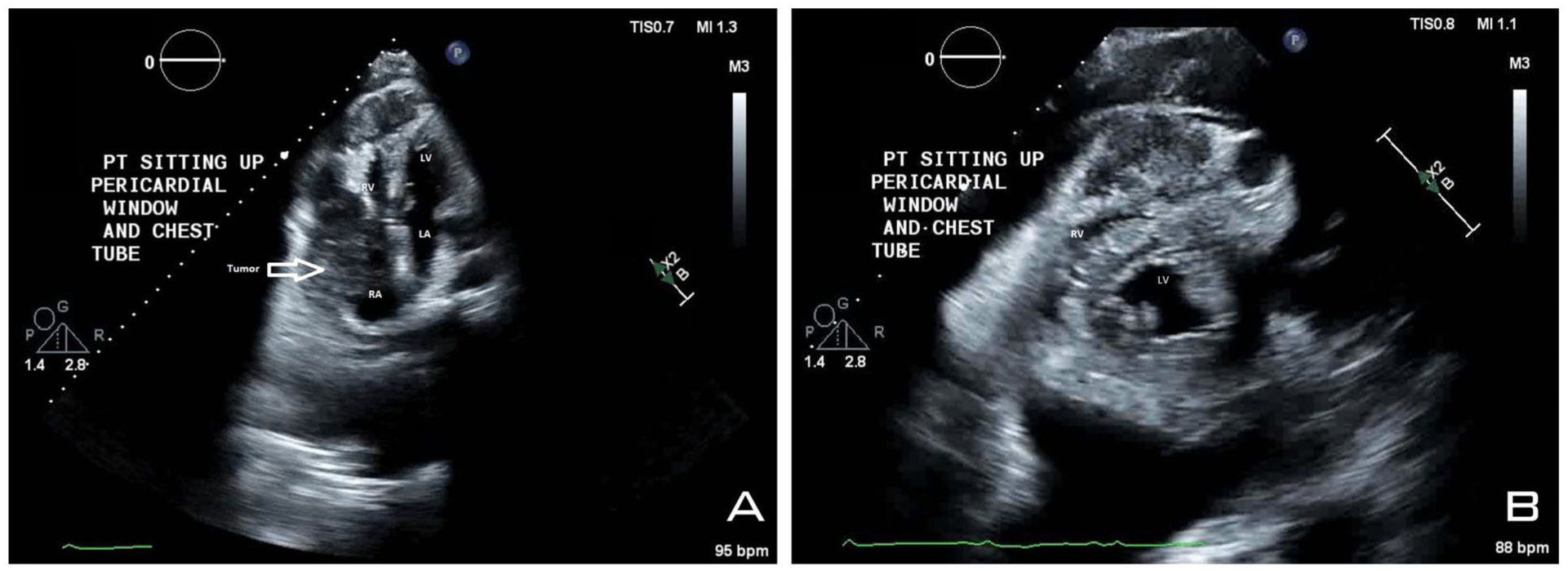
(A) Four-chamber echocardiography showing mild effusion post-pericardial window procedure with tumor invading the right ventricle and the right atrium. (B) Short-axis echocardiography showing mild effusion post-pericardial window procedure with tumor invading and compressing the right ventricle.
A computed tomography (CT) scan of the chest, abdomen, and pelvis with contrast was performed to locate the source of cancer. It revealed extensive mediastinal, pericardial, and retroperitoneal adenopathy, with evidence of peritoneal carcinomatosis. The scan also showed bilateral pleural effusions and ascites. In addition, left inguinal adenopathy and bilateral thyroid nodules were noted (Figure 3).
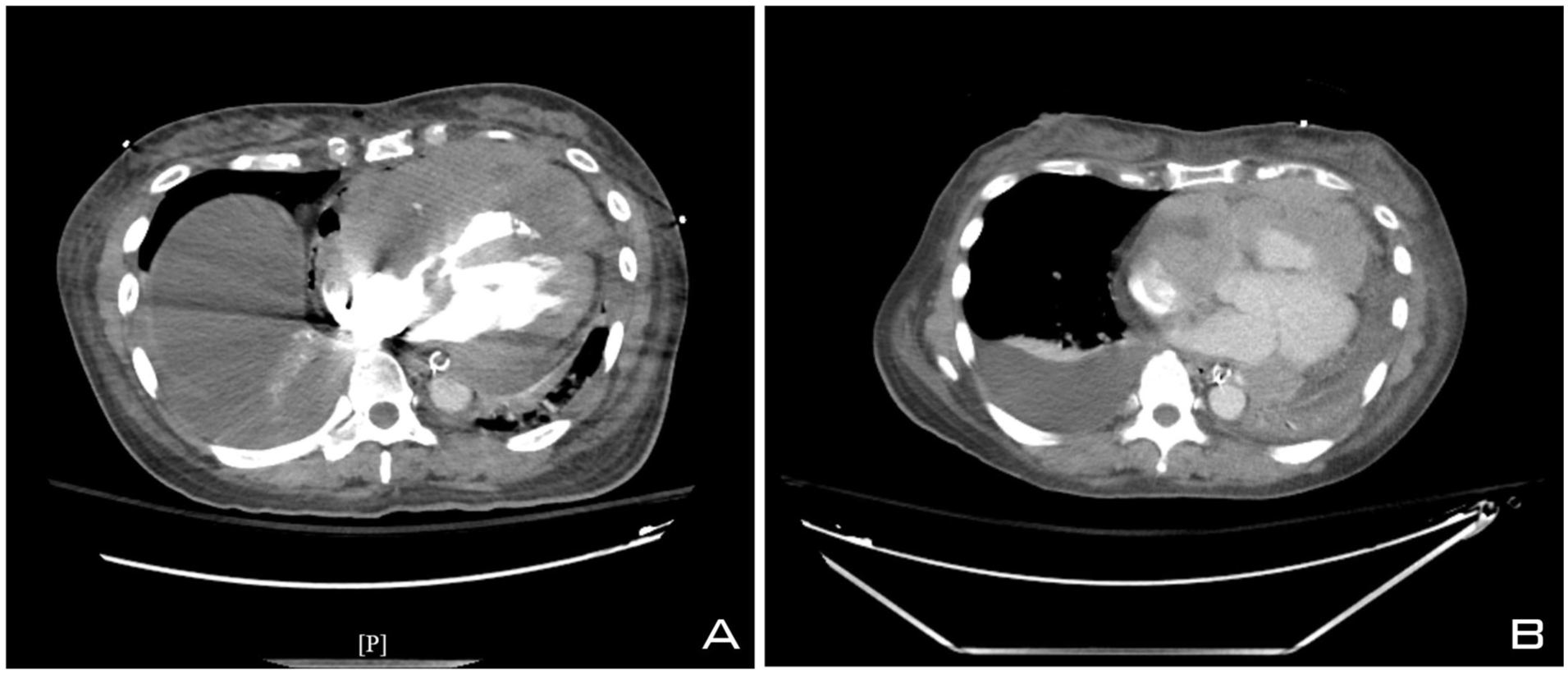
(A, B) CT scan showing moderate right and moderate to large pleural effusion. Cardiomegaly with residual pericardial effusion, with pericardial and mediastinal adenopathy, and a large mass invading the heart. CT, computed tomography.
A CT-guided left thoracentesis was performed, draining 800 mL of fluid, which was then sent for laboratory analysis.
At the same time, the surgical team was consulted for a left cervical lymph node biopsy, which was performed, confirming the diagnosis of DLBCL with a high Ki-67 proliferation index (80%-90%). A chemo-port was placed during the same procedure, and the patient was subsequently transferred to the telemetry unit with plans to initiate chemotherapy using the R-CHOP regimen [CHOP is a combination regimen consisting of Cyclophosphamide, Hydroxydaunorubicin (doxorubicin), Oncovin (vincristine), and Prednisone, given in-hospital, with Rituximab as an outpatient].
A day later, the patient experienced hypoxemia, with SpO2 dropping to 83%. She was also tachypneic, tachycardic, and hypotensive, with a BP of 73/48 mmHg. In addition, she reported new-onset chest and abdominal pain. Blood gas analysis revealed a pH 7.31 (reference 7.35-7.45), pCO2 32 mmHg (reference 35-45 mmHg), pO2 188 mmHg (reference 80-100 mmHg), and a lactate level of 5.6 mmol/L (reference <2 mmol/L).
Given the patient’s hemodynamic instability, a CT pulmonary angiogram was obtained, revealing acute pulmonary embolism (PE) in the right pulmonary artery with associated right heart strain due to the mediastinal mass. A full septic workup was initiated. Surgical oncology indicated that the PE was likely secondary to the tumor burden and its invasion into cardiac structures. Central venous and arterial lines were inserted, and the patient was intubated due to worsening respiratory failure and hypoxemia, and was transferred to the medical intensive care unit.
Despite aggressive supportive measures, including ventilatory and hemodynamic support, the patient’s condition continued to deteriorate, and she ultimately succumbed to her illness.
Discussion
In the United States, the annual incidence of NHL is ~7 cases/100 000 people, with DLBCL being a common subtype that accounts for about 40% of all lymphomas.4,5 While primary cardiac lymphoma accounts for only 1% to 2% of all cardiac tumors, secondary cardiac involvement in DLBCL typically occurs as a late manifestation, often going undetected until advanced stages or diagnosed at autopsy.2,3,6,7 In most cases, cardiac involvement is clinically silent, and patients do not manifest overt cardiovascular symptoms, with findings often discovered incidentally during imaging or at autopsy. The prognosis for patients with cardiac metastasis is poor, with a median survival of <1 year in most cases.8-10 Cardiovascular symptoms may include pericarditis, pericardial effusion, angina, arrhythmias, and congestive heart failure.6,7 Our case represents one of these rare occurrences involving a middle-aged Asian female patient with NHL who presented with cardiac tamponade secondary to cardiac metastasis as an initial presentation of DLBCL, a rare and severe manifestation of cardiac involvement, highlighting that although unusual, significant cardiac decompensation can occur. The diagnosis was confirmed through cytology of the malignant pericardial effusion, followed by histopathological examination of a cervical lymph node biopsy, which confirmed DLBCL. The patient had no prior history of lymphoma-related symptoms, such as B-symptoms (fever, night sweats, weight loss), indicating that the cardiac event was indeed the first clinical manifestation, suggesting a late diagnosis.
Multimodal imaging is essential for assessing cardiac masses and diagnosing DLBCL. Echocardiography serves as the initial imaging modality, with transesophageal echocardiography showing greater sensitivity in evaluating cardiac chambers and the pericardium involvement compared to the transthoracic approach, which offers a more limited acoustic window. However, advanced imaging modalities such as CT and magnetic resonance imaging (MRI) provide superior tissue characterization and anatomical delineation. Nuclear imaging is also critical in monitoring treatment response and assessing disease progression, thereby optimizing patient management. A CT scan and CT angiography of our patient revealed a large anterior mediastinal mass that had invaded the right ventricle. In addition, there was pulmonary thromboembolism in the right lung, pleural effusion, and significantly enlarged lymph nodes in the cervical, mediastinal, peritoneal, abdominal, and pelvic regions, along with involvement of the thyroid and peritoneum.
Right atrial and pericardial masses, cardiac or metastatic in origin, represent a diagnostic challenge. The most common non-cardiac tumor is located in the right atrium. 11 While TTE can detect such lesions, its limited acoustic window necessitates the use of complementary imaging modalities. Cardiac MRI remains the gold standard for diagnosis, given its superior ability to characterize tissue composition, vascularity, and morphology.12,13
It is essential to emphasize that multimodality imaging should always be complemented by histopathological confirmation to establish a definitive diagnosis. Pericardial or pleural fluid cytology is especially useful when accessible lymph nodes or masses for biopsy are absent. In this case, the patient’s cervical lymph node biopsy confirmed DLBCL.
While the R-CHOP chemotherapy regimen, which includes rituximab, doxorubicin, cyclophosphamide, vincristine, and prednisone, significantly improves the prognosis of extranodal disease, data on the prognosis of patients with cardiac involvement in disseminated lymphoma are limited to case reports.14,15 In our patient, the absence of preceding lymphoma symptoms and late presentation significantly restricted treatment options, underscoring the challenges of managing advanced disease with cardiac involvement.
According to the 2015 European Society of Cardiology Guidelines for malignant effusions, pericardial drainage remains the first-line treatment for malignant effusions, with pericardial window creation serving as a viable alternative to repeated pericardiocentesis. Pericardiectomy is reserved for recurrent or constrictive cases. 16 Radiotherapy may provide symptomatic relief in secondary lymphomatous pericardial effusions; however, it carries risks such as myocarditis and chronic pericarditis.14,15
It is also critical to recognize the role of cancer-associated thrombosis (CAT) in the morbidity and mortality of patients with advanced malignancy. Malignant cells can induce a prothrombotic state through multiple mechanisms, including increased expression of tissue factor, release of procoagulant microparticles, and dysregulation of cytokines such as interleukin-6 and tumor necrosis factor-alpha.17,18 Moreover, chemotherapy and endothelial injury further amplify this hypercoagulable milieu, predisposing patients to venous thromboembolism and PE.17,18 In our patient, the extensive tumor burden and vascular invasion likely contributed to a paraneoplastic thrombotic cascade, ultimately resulting in fatal PE.
Ultimately, our patient’s demise due to PE underscores the devastating consequences of CAT in advanced lymphoma. This case highlights the importance of early recognition of cardiac involvement in hematologic malignancies and maintaining a high index of suspicion for thrombotic complications. It also reinforces the need for multidisciplinary collaboration among cardiology, oncology, and CTS teams to facilitate timely diagnosis, comprehensive management, and improved outcomes.
Conclusion
In conclusion, this case underscores a rare presentation of DLBCL manifesting initially as a large pericardial effusion—an uncommon but critical diagnostic consideration often mistaken for infectious or autoimmune causes. The diagnosis, confirmed by pericardial fluid cytology and lymph node biopsy, underscores the importance of considering hematologic malignancies in the differential diagnosis of undifferentiated pericardial effusions. Early recognition through multimodal imaging and tissue confirmation is essential to improve outcomes, as delayed diagnosis can lead to life-threatening complications such as cardiac tamponade and thromboembolism.
Footnotes
Author Note
Prior Presentation Statement: The case was presented as an oral presentation at the CHEST 2025 conference in Chicago, Illinois, on October 19-22, 2025.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
The patient gave written informed consent to publish this case report and any accompanying images. The patient was informed that identifying information would be removed to ensure anonymity and consented to using the patient’s medical history for educational and research purposes.
Consent for Publication
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
