Abstract
Systemic sclerosis with negative serology, particularly that complicated by scleroderma renal crisis (SRC), is rarely encountered. We describe a patient with seronegative systemic sclerosis who developed acute kidney injury, proteinuria, and hypertensive emergency following motor vehicle-related trauma and in the setting of nonsteroidal anti-inflammatory drug use. Findings on physical examination, imaging, and skin biopsy led to a clinical diagnosis of scleroderma, despite the lack of supportive laboratory data. IgG lambda paraproteinemia was detected on workup. Bone marrow biopsy showed plasmacytosis and trace lambda-restricted plasma cells consistent with monoclonal gammopathy of undetermined significance. Chemotherapy was initially started given concern for myeloma with cast nephropathy but was later stopped after a kidney biopsy revealed thrombotic microangiopathy (TMA). The SRC associated with TMA was ultimately diagnosed, though atypical hemolytic uremic syndrome (aHUS) induced perhaps by monoclonal gammopathy or hypertension was also possible. Captopril and eculizumab were initiated for SRC and aHUS, respectively. Despite therapy, renal function did not recover, and the patient required hemodialysis indefinitely. This case highlights clinical features common to both SRC and aHUS as well as points out a few ways to differentiate between them.
Keywords
Introduction
Systemic sclerosis (SSc), also known as scleroderma, is an immune-mediated disease defined by extensive tissue fibrosis of the skin and internal organs. 1 Symptoms vary based on organ-specific involvement. One of the most severe complications of SSc is scleroderma renal crisis (SRC), which involves intimal proliferation in small arcuate and interlobular arteries leading to obliterative vasculopathy. 2 Small vessel thrombi are seen, reflecting thrombotic microangiopathy (TMA). Typical manifestations include hypertension, microangiopathic hemolytic anemia (MAHA), thrombocytopenia, and rapidly progressive renal failure. 3 Risk factors for SRC include diffuse skin involvement, presence of RNA polymerase III antibodies, and corticosteroid use. Certain nephrotoxic medications such as cyclosporine or nonsteroidal anti-inflammatory drugs (NSAIDs), hypovolemia, and sepsis may precipitate SRC. 3 Angiotensin-converting enzyme inhibitors are the cornerstone of therapy, improving survival and renal recovery. 2
Monoclonal gammopathy of undetermined significance (MGUS) is a premalignant plasma cell dyscrasia. Diagnosis of MGUS requires the presence of a monoclonal protein in the serum at a concentration of less than 3 g/dL and less than 10% monoclonal plasma cells on bone marrow biopsy. Myeloma-associated complications including hypercalcemia, renal impairment, anemia, and bony lytic lesions must be absent. 4 Management of MGUS primarily involves monitoring for progression to smoldering myeloma or multiple myeloma. Monoclonal gammopathy of undetermined significance is associated with disorders such as amyloidosis, cryoglobulinemia, and connective tissue diseases, including scleroderma and scleromyxedema.4,5 Monoclonal gammopathies (MGUS or otherwise) have also been observed in association with TMA, including atypical hemolytic uremic syndrome (aHUS).6 -8
Scleroderma renal crisis developing in a patient with seronegative SSc is rare. As such, it may raise concern for possible scleroderma mimickers instead, such as scleromyxedema. Scleromyxedema is often associated with an underlying paraproteinemia and may involve the kidneys, although uncommonly, in the form of an SRC-like acute kidney injury (AKI) event. 9 We describe a patient who presented with seronegative scleroderma complicated by renal crisis and TMA, possibly induced by monoclonal gammopathy or hypertension.
Case Presentation
A 47-year-old man presented with encephalopathy after a low-speed motor vehicle accident. He was intubated for airway protection and admitted to the surgical intensive care unit. On hospital day 2, he was extubated but remained encephalopathic. Systolic blood pressures (SBP) consistently ranged from 150 to 170s mm Hg. Laboratory investigation was notable for AKI with creatinine of 1.45 mg/dL and mild thrombocytopenia with a platelet count of 118 × 103/µL. Urinalysis showed 3+ protein and 3+ hemoglobin. Magnetic resonance imaging of the brain was consistent with posterior reversible encephalopathy syndrome. Hypertensive emergency was initially managed with clevidipine infusion. Encephalopathy gradually resolved. The patient was discharged on amlodipine 10 mg daily and labetalol 300 mg twice daily.
Two weeks later, the patient was evaluated in nephrology clinic for uncontrolled hypertension, proteinuria, hematuria, and elevated creatinine. At this point, his main symptoms were fatigue and lower extremity edema. He also reported a 5-month history of paresthesia in his feet. The patient had seen a neurologist but was lost to follow-up. In addition, he endorsed taking ibuprofen 600 mg twice daily for the previous 2 months. Serum creatinine was 1.61 mg/dL, and urinalysis again showed 3+ protein and hemoglobin. Urine protein to creatinine ratio was 1.43 g/g.
Several weeks later, he was seen in follow-up and subsequently admitted with malaise, volume overload, and decreased urine output. At this time, he also endorsed dysphagia and reflux symptoms. He denied weight changes, fevers, chest pain, shortness of breath, abdominal pain, diarrhea, urinary symptoms, and Raynaud’s symptoms. The SBP ranged from 140 to 160s mm Hg. Physical examination findings of sclerodactyly, salt and pepper pigmentation of the skin, and telangiectasias on the face, upper chest, extremities, and hands were identified. Bilateral 2+ pitting edema was present up to the ankles. Dilated nailfold capillaries were observed on capillaroscopy. Musculoskeletal examination showed no joint deformities, synovitis, or tendon friction rubs.
Laboratory investigations at this point included a complete blood count which showed a mild leukocytosis and mild anemia. Platelets were low during most of the hospital course, ranging from 161 to 170 × 103/µL (Table 1). Blood urea nitrogen and creatinine levels had markedly increased from prior (Table 2).
Complete Blood Count and Metabolic Profile.
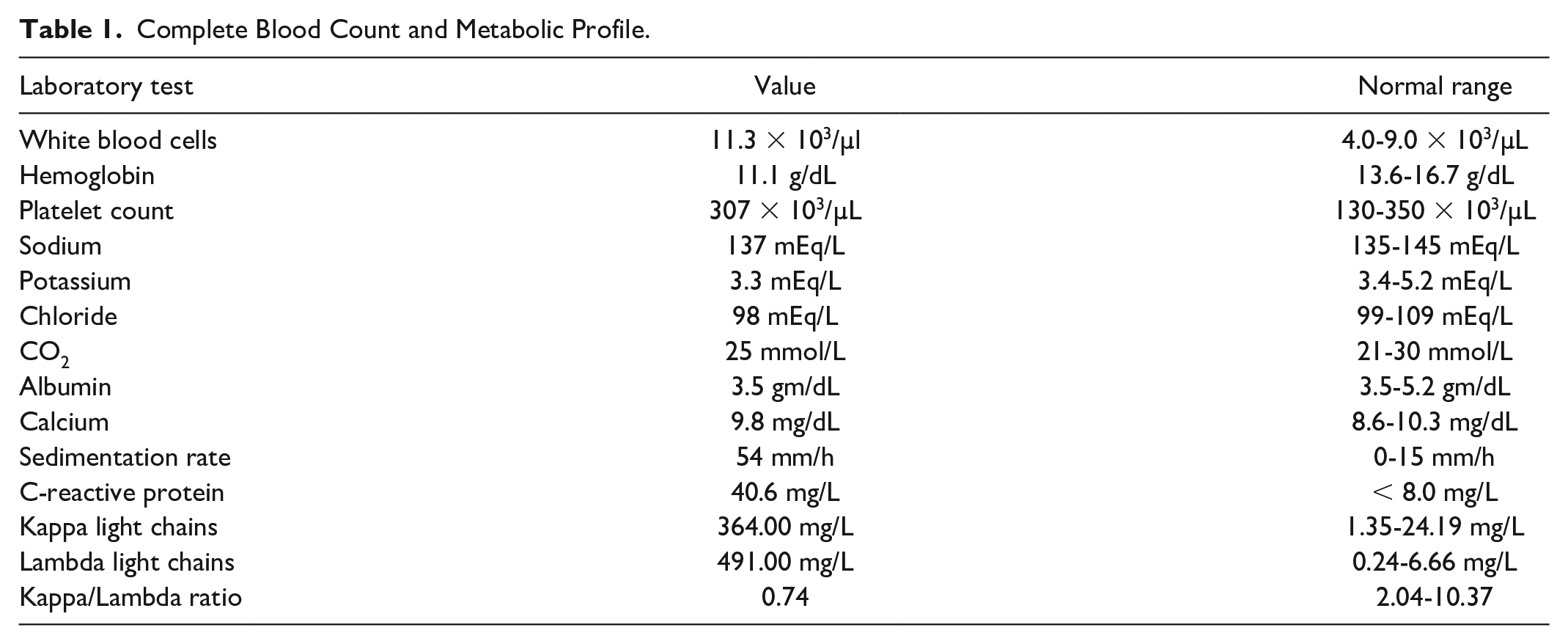
Measurements of Serum Creatinine, Blood Urea Nitrogen and Estimated Glomerular Filtration Rate (eGFR) Over Time.

Estimated using MDRD (modification of diet in renal disease) equation.
Liver enzymes, total bilirubin, haptoglobin, and lactate dehydrogenase were normal. Erythrocyte sedimentation rate and C-reactive protein were moderately elevated. Urinalysis showed 3+ protein, 2+ hemoglobin, 3 to 5 RBCs/HPF, and course granular casts. Urine protein to creatinine ratio was 1.95 g/g.
Serum protein electrophoresis revealed a paraprotein in the gamma globulin region. Paraproteins were not detected on urine protein electrophoresis. Serum and urine immunoelectrophoresis revealed an IgG lambda paraprotein with a serum concentration of 0.22 g/dL. Urine-free light-chain investigation showed markedly elevated kappa and lambda free light chains with a decreased kappa lambda ratio (Table 1). β-2 microglobulin levels were elevated at 24.9 mg/L (0.6-2.4 mg/L).
A peripheral blood smear revealed schistocytes, prompting ADAMTS13 activity testing which was normal at 62%. Serology for antinuclear antibody (ANA) using IFA Hep-2 substrate (INOVA) was <1:40. Antibodies directed against topoisomerase I, RNA polymerase III, centromere, exosome (anti-PM/Scl), double-stranded DNA, U1-ribonuclear protein, U3-ribonuclear protein, Th/To, Ro, La, glomerular basement membrane, and antineutrophil cytoplasmic antibodies (ANCA) were negative. C3 and C4 complement levels were within normal limits. Serum cryoglobulins were not detected.
Imaging studies included a computed tomography of the chest, abdomen, and pelvis which showed circumferential thickening of the distal esophageal wall and distal rectum with no fractures or hemorrhage. An esophagram revealed esophageal dysmotility in the middle and distal portions of the esophagus without focal narrowing. A renal ultrasound showed no hydronephrosis, nephrolithiasis, or masses. Electromyography demonstrated severe symmetric sensory and motor axonal peripheral polyneuropathy of the legs and right hand.
A kidney biopsy revealed findings of both TMA and pathologic changes indistinguishable from malignant hypertension—overall a picture compatible with SRC. Thrombotic microangiopathy was predominantly affecting small intrarenal arteries and arterioles. Small interlobular arteries showed severe luminal narrowing by intimal fibroblastic cellular proliferation (Figure 1A), intimal fibrinoid degeneration, and endothelial swelling, focally imparting an “onion skin” appearance (Figure 1C). Arterioles demonstrated medial thickening with areas of fibrin thrombi (Figure 1B). Small arteries and arterioles demonstrated intimal foci of entrapped schistocytes and focal karyorrhexis. There were no necrotizing vasculitic changes or crescent formations. Glomeruli demonstrated “ischemic collapse” with wrinkled bloodless capillary loops (Figure 1A-C). Immunofluorescence staining and ultrastructural analysis showed no evidence of immune-mediated glomerulonephritis, podocyte effacement, or monoclonal immunoglobulin deposition. Congo red staining was performed and showed no evidence of amyloid deposits. At this point, the 2 main diagnostic considerations were aHUS and SRC.

Scleroderma diagnosis supported by a kidney biopsy with thrombotic microangiopathy and a skin biopsy with dermal sclerosis. (A) Interlobular artery with severe luminal narrowing by a cellular intimal proliferation with admixed mucoid intimal edema (arrow) while the adjacent glomerulus has global ischemic collapse (Periodic acid-Schiff stain, 200×). (B) Glomerulus with fibrin thrombus (arrow) expanding an arteriole at vascular pole (Jones methenamine silver stain, 600×). (C) Interlobular artery wall with concentric “onion skin” like intimal lamination with associated mucoid intimal edema and severe narrowing of its lumen (Jones methenamine silver stain, 600×). (D) Forearm skin biopsy demonstrating papillary and mid-dermal sclerosis with thickened bands of collagen surrounding adnexal structures (H&E stain, 40×).
A bone marrow core biopsy demonstrated mildly hypercellular bone marrow with 20% plasma cells (Figure 2). Despite the plasmacytosis on bone marrow biopsy, flow cytometry showed only a trace population of lambda-restricted plasma cells.
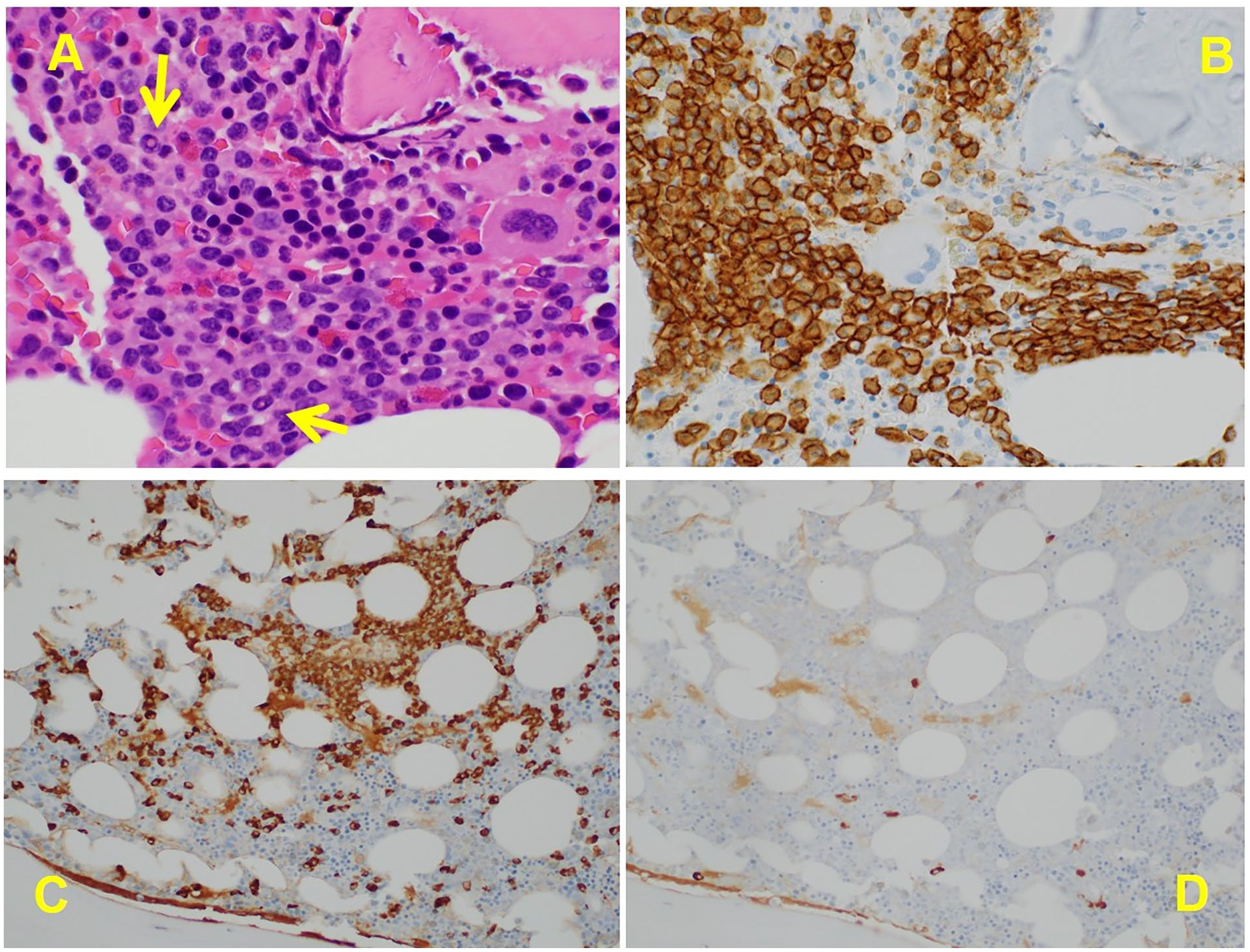
Bone marrow biopsy revealing monoclonal gammopathy. (A) Increased numbers of plasma cells, with arrows demonstrating intranuclear inclusions/Dutcher bodies (H&E stain, 600×). (B) Immunohistochemical stain CD138 confirms plasma cell lineage and highlights their increased number and sheet-like arrangements. (C) The plasma cells are lambda restricted by immunohistochemistry for lambda (C, 400×) and kappa (D, 400×).
Treatment with bortezomib, lenalidomide, and dexamethasone was started initially due to concern for myeloma kidney. Six months later, a repeat marrow biopsy showed 24% plasma cells with only 2% lambda-restricted plasma cells. This finding was more consistent with MGUS rather than myeloma, and treatment was discontinued by hematology.
Skin biopsies from the left and right forearm revealed subepithelial papillary to mid-dermal sclerosis (Figure 1D). Congo red staining of skin tissue was negative for amyloid material. Mucin deposition was not detected utilizing Alcian Blue/PAS staining. Immunofluorescence demonstrated perivascular fibrinogen staining and 2+ granular basement membrane staining for C5b-9. Combined with the findings of skin thickening, telangiectasias, and mucosal thickening of the distal esophagus and rectum, scleroderma was diagnosed.
Taking all elements of the presentation, laboratory, imaging, and biopsy findings, the final diagnosis was seronegative scleroderma complicated by SRC along with a TMA picture that potentially was triggered by hypertension or monoclonal gammopathy.
Intermittent hemodialysis was required for severe AKI. For SRC, captopril was initiated at 25 mg 3 times daily, and increased to 50 mg 3 times daily upon discharge. Due to the strong suspicion of aHUS and its life-threatening complications if left untreated, eculizumab was started with induction of 900 mg weekly infusions for 4 doses and continued for suspected aHUS. Renal function did not recover, and the patient required indefinite hemodialysis. As the patient was felt to have MGUS rather than myeloma, chemotherapy was discontinued. He was monitored regularly without progression of MGUS. For management of scleroderma, the patient was followed by rheumatology without progression of skin findings. Unfortunately, the patient died nearly 2 years after initial presentation.
Discussion
We outlined a case of seronegative scleroderma presenting with SRC and concurrent IgG lambda monoclonal gammopathy. Scleromyxedema was part of the differential diagnosis given its association with monoclonal gammopathy; however, histologically, scleromyxedema is characterized by mucin deposition in the mid to upper dermis with increased collagen and fibroblasts. 5 The skin biopsy findings of papillary and mid-dermal sclerosis without mucin deposition were more consistent with scleroderma than scleromyxedema. Immunoglobulin light-chain (AL) amyloidosis with scleroderma-like features was also considered in the differential especially given the neuropathy and gammopathy, though histology again did not support this diagnosis.
Regarding the differential of AKI, SRC and TMA were suspected given the nephritic presentation, marked hypertension, and MAHA (mild anemia, thrombocytopenia, schistocytes). The NSAID use could have contributed to SRC development or caused nephropathy independently. Otherwise, light-chain cast nephropathy was considered in the differential but appeared less likely in the presence of microhematuria. The ANCA-associated vasculitis, systemic lupus erythematosus, and cryoglobulinemic vasculitis were also considered; however, serologic and histologic results ultimately ruled out these alternative possibilities.
Several diagnoses were considered for the etiology of TMA. Thrombotic thrombocytopenic purpura (TTP) was ruled out by normal ADAMTS13 testing. In addition, TTP generally exhibits less prominent renal involvement than other causes of TMA. This left the main diagnostic considerations of aHUS and SRC. Differentiation between these disorders required clinical correlation with laboratory and histologic findings. Decreased serum levels of C3 with normal C4 are frequently seen in aHUS, although complement levels are not considered a reliable marker of disease.10,11 Complement levels were both normal in this case. Histologically, the disorders are quite similar but do have subtle differences. Acute and chronic vascular injury in SRC is generally located in small renal arteries and arterioles, whereas the vascular injury in aHUS tends to be located within glomerular capillaries and include double contour formation when the injury is chronic.2,12 The biopsy of our patient showed TMA lesions in small arteries and arterioles rather than glomerular capillaries. Scleroderma renal crisis is also characterized by the presence of “onion skin” lesions in arteries and arterioles, 2 which were present in the biopsy. These findings were more suggestive of SRC. Finally, diagnosis of aHUS requires exclusion of other causes of TMA, 13 which in this case was SRC.
Case reports of seronegative SRC have been reported but are rare. 14 In a study by Salazar et al of 3249 scleroderma patients, 6.4% were ANA negative. The study further showed that these patients had fewer vascular sequelae, were more likely to be male, and had a higher rate of gastrointestinal complications. 15
The pathogenesis of SRC remains unclear, although research suggests that intimal proliferation from an endothelial insult leads to luminal narrowing of small renal arteries and arterioles. 2 Reduced renal blood flow resulting from hypovolemia, for instance, is known to precipitate SRC. 3 In this case, NSAIDs or relative hypovolemia associated with trauma may have helped provoke SRC, a TMA-like process. Sustained hypertension probably stemming from SRC or monoclonal gammopathy could have also helped trigger immune-mediated TMA. Monoclonal gammopathy may serve as a potential trigger for TMA by activating the alternative complement pathway (indirect mechanism) or injuring endothelial cells within capillaries (direct mechanism), though the mechanism is not fully understood.6 -8
The relationship between scleroderma and MGUS is presently unclear. A study by Ughi et al 16 showed that patients with SSc have a similar prevalence of MGUS to that of the general population below the age of 60. Interestingly, however, there are case reports implicating scleroderma as a paraneoplastic syndrome with multiple myeloma. Many of these cases describe patients with diffuse skin involvement.17,18 Scleroderma serology in most cases is negative. Treatment of myeloma generally results in improvement of scleroderma features, supporting the notion of a paraneoplastic syndrome. Scleroderma appears to be intricately linked with the development of malignancy. In a systematic review by Bonifazi et al, 19 lung and hematologic malignancies were most common. Evidence for the risk of other forms of malignancy including gastrointestinal, genitourinary, and skin is conflicting. The presence of RNA polymerase III antibodies confers an increased risk. 20 A proposed pathophysiologic mechanism for this phenomenon is that the immune system develops a response to mutated antigens cancer cells which results in cross-reactivity with host cell antigen. 21
Conclusion
This case of scleroderma with negative serology yet manifesting with significant organ involvement, particularly renal involvement, illustrates the importance of judging the diagnosis based on the salient clinical features of the case, even when laboratory data might indicate otherwise. Further research is needed to investigate the risk of renal failure and other complications in this subset of patients.
Footnotes
Authors’ Note
This case was presented as an abstract by Dr Parth Visrodia at American Society of Nephrology Kidney Week, November 2019.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information and images to be published in this article. Unfortunately, the patient has since died.
