Abstract
Several adverse effects have been reported following the use of the Pfizer-BioNTech mRNA vaccine against COVID-19 infection with acute pancreatitis being one of those. However, a direct relationship between the vaccine and the occurrence of acute pancreatitis has not been confirmed. Hereby, a case of an acute necrotizing pancreatitis resulting in fatal outcome following the administration of the COVID-19 BioNTech Pfizer vaccine is presented. A 70-year-old male presented with abdominal pain 4 days after receiving his fifth dose of the COVID-19 BioNTech Pfizer vaccine. Laboratory examination showed lipase levels of 2000 U/L. A CT scan was performed and showed necrotizing pancreatitis. After detailed evaluation, no etiological cause of pancreatitis could be found in this patient. The patient was admitted to the intensive care unit. After a disease course of ~6 weeks, the patient died due to multiorgan failure. This case presentation takes into account previously reported cases of acute pancreatitis following COVID-19 vaccination. To our knowledge, this is the first case of acute pancreatitis with fatal ending following a COVID-19 vaccination. This highlights the importance of thorough evaluation of patients presenting with abdominal pain following the administration of a COVID-19 vaccine.
Keywords
Introduction
The COVID-19 BioNTech Pfizer vaccine came on the market in December 2020, when the COVID-19 virus was the cause of a pandemic.1-6 Although the amount of data on adverse effects following vaccination with the Pfizer vaccine is rather limited, several case reports propose an association between vaccination with the COVID-19 BioNTech Pfizer vaccine and the occurrence of acute pancreatitis. This case report with analysis of several other case reports further supports this association. The presented case distinguishes itself from previously reported cases since the patient in this case succumbed due to the complications of a severe acute necrotizing pancreatitis.
Case Presentation
We present the case of a 70-year-old man with a history of arterial hypertension and coronary artery disease and therefore stenting of the LAD in 2010, there was no history of (acute) pancreatitis. The patient’s medication list consisted of acetylsalicylic acid, a combination preparation of enalapril and lercanidipine, a combination preparation of bisoprolol and hydrochlorothiazide, and a combination preparation of rosuvastatin and ezetimibe.
The patient presented to the Emergency Department (ED) of another hospital in October 2023, 4 days after receiving his fifth dose of the COVID-19 BioNTech Pfizer vaccine. He was feeling unwell since a few days and started developing abdominal pain on the morning of presentation to the ED. There was no history of vomiting, diarrhea, or fever. Laboratory examination showed lipase levels of 2000 U/L (reference levels 13-60 U/L). Triglyceride levels were negative namely 115 mg/dL with the reference level being under 150 mg/dL. IgG4 levels were also negative namely 0.683 g/L (reference levels 0.03-2.01 g/L). A Computerized Tomography (CT) scan was performed and showed necrotizing pancreatitis (Figure 1A). The patient was first admitted to the gastroenterology ward for IV fluids and analgesics. After 2 days, the patient was transferred to the intensive care unit (ICU) ward because of multiorgan failure. There, Meropenem and dialysis were started. The patient was also intubated because of respiratory insufficiency.
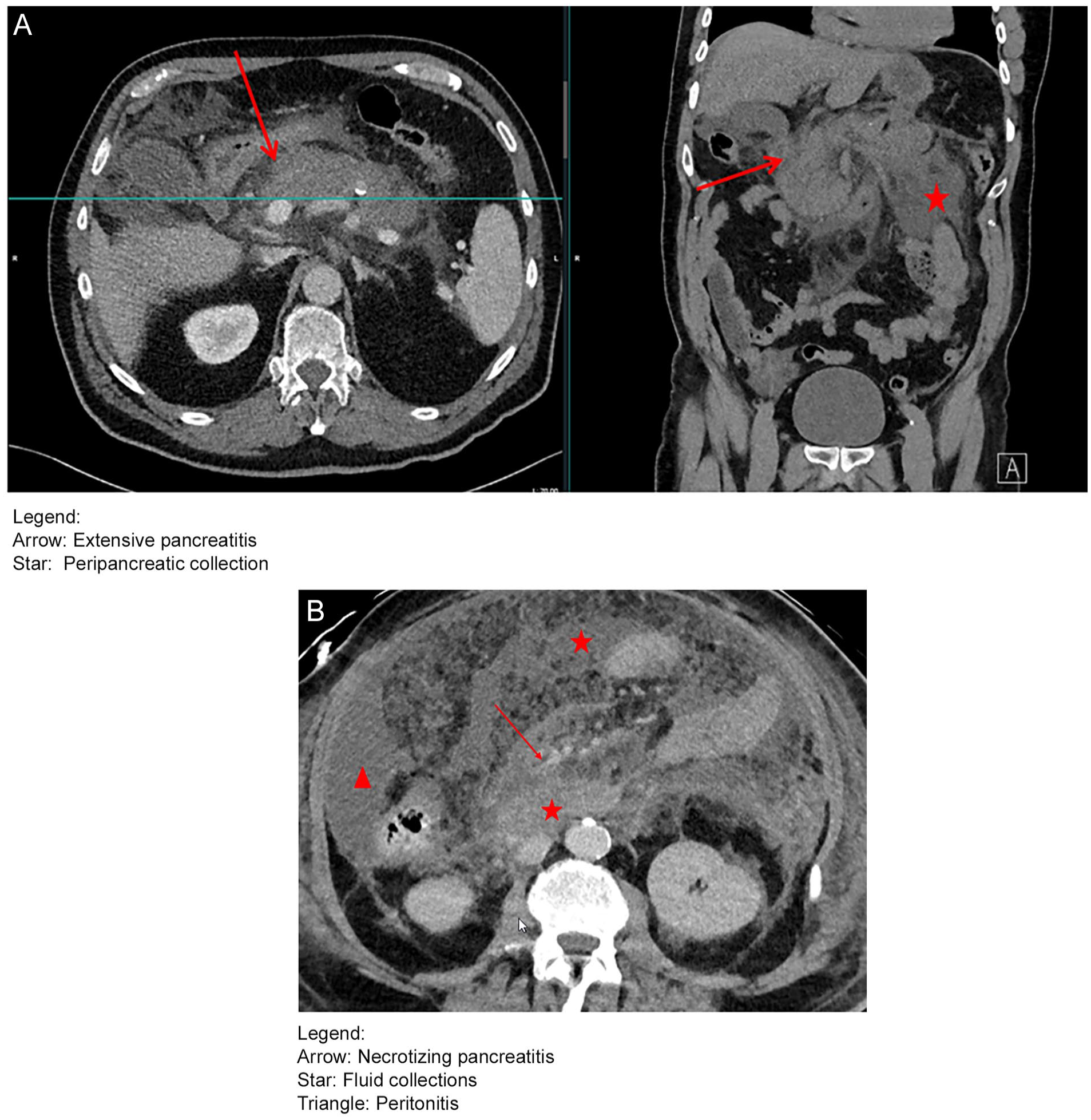
(A) CT scan of the necrotizing pancreatitis at the moment of presentation: day 0. Arrow: extensive pancreatitis. Star: peripancreatic collection. (B) CT scan of the necrotizing pancreatitis on day 18. Arrow: necrotizing pancreatitis. Star: fluid collections. Triangle: peritonitis.
On day 12, the patient was transferred to our ICU because of increasing intra-abdominal pressure and difficult ventilation. At the time of arrival in our hospital, the abdominal pressures had normalized and after consulting with the abdominal surgeons, a conservative management was pursued. A new CT showed multiple fluid collections (Figure 1B). Therefore, a transgastric drainage with stenting was performed. Also, a percutaneous suprapubic drainage of fluid was performed. Culture of the transgastric drainage was positive for Candida glabrata, and thus, Caspofungine was started. Meropenem was switched to Tazocin because of thrombocytopenia and Vancomycine was associated because of increasing inflammatory parameters. Caspofungine was switched to Voriconazole because of positive Galactomannan via broncho-alveolar lavage. On day 38, the intra-abdominal pressure increased, and ventilation became more difficult with increasing respiratory pressure. An urgent decompressive laparotomy was performed because of an acute abdominal compartment syndrome, but it was impossible for the necrotic collections to be drained because of their hardness. Ventilation could also not be improved. Eventually, the patient died on day 39.
Discussion
Most cases of acute pancreatitis in developed countries are due to alcohol, gallstones, or hypertriglyceridemia. Endoscopic retrograde cholangiopancreatography, certain drugs, hypercalcemia, viral infections, trauma, tumors, and anatomical variants can also cause acute pancreatitis. In our case, triglyceridemia was not elevated at the moment of presentation, there were no gallstones, and the use of alcohol was denied. Three preparations of the patient’s medication can be associated with acute pancreatitis, but the patient was already taking these medications for more than 5 years, making this rather unlikely. Other possible etiological factors were also not found. A possible association could be the COVID-19 vaccination 4 days prior to presentation. Acute pancreatitis after certain vaccinations has been described. Searching the literature, the mechanism by which vaccines could induce acute pancreatitis remains unclear; however, molecular mimicry is postulated as a plausible explanation. According to this hypothesis, an autoimmune reaction targeting pancreatic acinar cells occurs due to structural similarities between the viral antigens and host self-antigens.7,8
After searching the PubMed and Google abstract databases, there were 20 case reports found proposing an association between COVID-19 vaccine and the occurrence of acute pancreatitis. Sixteen of these cases occurred after Pfizer vaccination, in 1 article the vaccine type was not specified, in another case acute pancreatitis occurred after vaccination with Johnson & Johnson, in another case after the Sputnik V and in the last case after the Sinopharm BIBP COVID-19 vaccine (Table 1).
Analysis of Other Case Reports.

Abbreviations: CAD: coronary artery disease; DES: drug eluting stent; DRESS: drug rash with eosinophilia and systemic symptoms; ERCP: Endoscopic retrograde cholangiopancreatography; F: female; GERD: gastroesophageal reflux disease; HUS: hemolytic uremic syndrome; ICU: intensive care unit; IV: intravenous; M: male; NS: not specified; NSAID: nonsteroidal anti-inflammatory drug; PPI: proton pump inhibitor; SLE: systemic lupus erythematosus; U: unit.
To our knowledge, this is the first case report describing acute pancreatitis with fatal outcome after COVID-19 vaccination. When searching the European database of suspected adverse drug reaction, it reports 737 cases of pancreatitis following COVID-19 Pfizer vaccination. 9 Of these cases, 15 were reported to be fatal. Since reporting of adverse drug reactions is not obligatory, this is probably an underestimation. Pancreatitis after other COVID-19 vaccines has also been reported, especially after vaccination with Moderna (280 cases) and AstraZeneca (181 cases).
When the COVID-19 Pfizer vaccine was approved, in December 2020, COVID-19 was the cause of a pandemic where the prevalence of COVID-19 infection was high. 10 In January 2021, there were 4 million new cases of COVID infections every week, with ~76 000 deaths/week, corresponding with a mortality of 1.9%. 11 In November 2023, there were ~130 000 new cases a week and 600 deaths/week, and thus a lower mortality rate of 0.4%. The number of hospitalizations due to COVID-19 infection has also grossly decreased. 12
Apparently, the amount of COVID infections and deaths has largely decreased since the use of COVID-19 vaccination. The Sciensano database, showing the epidemiological situation in Belgium, demonstrates that the amount of booster vaccinations is declining while the number of hospitalizations due to COVID-19 is not rising.
To conclude, the advantageous effects of vaccination in the perspective to the possible complications are always to be considered in a benefit/risk analysis. Furthermore, in an individual patient, one should be aware of the possibility of acute pancreatitis after vaccination.
Footnotes
Acknowledgements
We would like to thank the Department of Intensive Care Unit of Roeselare for their valuable support in the preparation of this case report. We also extend our sincere gratitude to the family of the patient for consenting to share this case for educational purposes.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Verbal informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
