Abstract
Central nervous system (CNS) toxoplasmosis is a life-threatening opportunistic infection most often reported in patients with HIV/AIDS and solid organ transplantation. Its occurrence in immunocompetent patients is rare and even less commonly reported in those receiving single-agent immunosuppressive therapy. We present a unique case of CNS toxoplasmosis in a 68-year-old seropositive female with autoimmune hepatitis maintained on long-term mycophenolate mofetil (MMF) monotherapy. She presented with a 2-day history of confusion and expressive aphasia, without fever or focal neurologic deficits. Brain MRI revealed multiple ring-enhancing lesions, prompting a broad differential diagnosis including malignancy, abscess, and demyelination. A stereotactic biopsy confirmed necrotic tissue harboring Toxoplasma gondii tachyzoites and cysts. Her serum Toxoplasma IgG was markedly elevated, indicating reactivation rather than primary infection. Treatment with high-dose sulfamethoxazole-trimethoprim led to radiologic and clinical improvement. MMF was discontinued, and the patient remained stable off immunosuppression under close hepatic monitoring. This case underscores the underestimated risk of opportunistic infections associated with chronic MMF monotherapy in non-HIV, nontransplant populations. As similar cases begin to emerge in the literature, clinicians must recognize that MMF can sufficiently impair immune defenses to permit CNS toxoplasmosis. This report contributes to a growing body of evidence advocating for broader awareness of atypical presentations of opportunistic neuroinfections in autoimmune disease management.
Keywords
Introduction
Toxoplasma gondii is a protozoan parasite with a global seroprevalence estimated to range from 10% to 60%, varying by region, diet, and host immune status. 1 While most infections in immunocompetent individuals are subclinical or self-limited, reactivation of latent infection can lead to severe manifestations, including central nervous system (CNS) toxoplasmosis, particularly in immunocompromised populations. 2 CNS toxoplasmosis is most commonly reported in patients with AIDS, where it remains a leading cause of intracerebral mass lesions, as well as in transplant recipients and those receiving cytotoxic chemotherapy. 3 However, reports of CNS toxoplasmosis in patients without HIV or solid organ transplantation are exceedingly rare. Mycophenolate mofetil (MMF), an immunosuppressant widely used in autoimmune diseases and transplant medicine, has been associated with opportunistic infections due to its selective inhibition of lymphocyte proliferation.4-6
This report presents a rare case of CNS toxoplasmosis in a non-HIV, nontransplant patient on long-term MMF for autoimmune hepatitis. It underscores the need for heightened clinical suspicion for toxoplasmosis in atypical settings and highlights the diagnostic and therapeutic challenges in such presentations.
Case Description
A 68-year-old female presented to the emergency department with a 2-day history of intermittent confusion and expressive speech difficulties. She had been at her baseline cognitive and functional status until symptom onset. She denied fever, seizure activity, visual disturbances, or recent travel. Her past medical history was significant for autoimmune hepatitis, managed with MMF 500 mg twice daily for 11 years, and type II noninsulin-dependent diabetes mellitus. She was diagnosed with autoimmune hepatitis ~12 years prior to this encounter and was initially started on prednisone and azathioprine. She had completed a 2-month prednisone taper. She was found to be severely anemic and thrombocytopenic at her 6-month follow-up, with a hemoglobin level of 4.5 g/dL and a platelet count of 25 000/μL. As a result, her regimen was transitioned to MMF, which she has continued since that time. She has not received any other forms of immunosuppressive therapy. Her diabetes has been reasonably well controlled with weekly subcutaneous dulaglutide 3 mg injections, and her most recent hemoglobin A1C was 6.2%. There was no history of HIV, malignancy, or organ transplantation.
On examination, she was alert, oriented, and cooperative, with no focal neurological deficits. Her vital signs were stable, and there were no signs of meningismus, systemic infection, or manifestations of cirrhosis. Initial laboratory work up including complete blood cell count, renal function panel, hepatic function panel, hepatitis panel, and fourth-generation HIV1/2 immunoassay were unremarkable.
A noncontrast head computed tomography (CT) revealed vasogenic edema within the posterior right frontal periventricular white matter (Figure 1). Brain MRI with contrast showed multiple ring-enhancing lesions, the largest measuring 1.7 × 1.7 cm in the right frontal lobe, with associated edema (Figure 2). The imaging differential included metastatic disease, abscess, lymphoma, or atypical demyelinating lesions.

Noncontrast CT brain shows hypodense vasogenic edema in the right frontal periventricular white matter without mass effect (blue arrows), suggestive of an inflammatory, or infectious etiology in an immunocompromised host.

Axial FLAIR MRI images showing multiple hyperintense lesions in the bilateral frontal and parietal lobes with surrounding edema (blue arrows). Lesions demonstrate asymmetrical involvement of periventricular white matter.
To establish a definitive diagnosis, she underwent a stereotactic right frontal brain biopsy, which demonstrated necrotic brain parenchyma with scattered tachyzoites and rare cysts with bradyzoites, morphologically consistent with T. gondii (Figure 3). Serum Toxoplasma IgG was markedly elevated at >400 IU/mL (normal <7.20 IU/mL), confirming chronic latent infection with reactivation.

(A) Low magnification view shows a necrotizing lesion with fibrinoid and dirty necrosis. (B) High magnification shows numerous clusters of tachyzoites within a background of necrosis. These tachyzoites may be interpreted as nuclear debris on low magnification, especially in a background of necrosis.
Due to concerns about the affordability of pyrimethamine and the need for sulfadiazine to be taken 4 times daily, sulfamethoxazole-trimethoprim (SMX-TMP) was selected over the pyrimethamine-sulfadiazine-leucovorin regimen. Treatment was initiated with intravenous SMX-TMP, later transitioned to high-dose oral SMX-TMP 1600 to 320 mg twice daily. Mycophenolate was discontinued, and a multidisciplinary team decided against initiating alternative immunosuppressive therapy, opting instead for close hepatic monitoring. The patient tolerated therapy well, and a 4-week follow-up MRI showed reduction in lesion size and vasogenic edema (Figure 4). After 6 weeks of high-dose therapy, she was transitioned to maintenance SMX-TMP 800 to 160 mg twice daily for secondary prophylaxis. She was initially monitored every 2 weeks with renal and hepatic function panels for the first 6 weeks. Follow-up was then spaced to monthly visits for the next 3 months, and subsequently every 3 months. At her 9-month follow-up, she remained clinically stable without any adverse effects from SMX-TMP and without any deterioration in liver function. The hepatic function panels at presentation, and at 3-, 6-, and 9-month follow-ups are summarized in Table 1. The current plan is to continue SMX-TMP 800 to 160 mg twice daily indefinitely, with close monitoring of hepatic function while off MMF. MMF therapy will be reinstated if there is evidence of progressive liver function deterioration.

Follow-up axial FLAIR MRI images showing resolution and improvement of periventricular and cortical hyperintensities (blue arrows), indicating radiological improvement of CNS toxoplasmosis following antimicrobial therapy, CNS, central nervous system.
Hepatic Function Panels.

Abbreviations: ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate transaminase; INR: international normalized ratio.
Discussion
T. gondii is a globally prevalent protozoan parasite with the remarkable ability to infect a wide range of warm-blooded animals, including humans. Infection in humans primarily occurs through the consumption or handling of undercooked or raw meat, particularly pork or lamb that contains tissue cysts or through ingestion of food or water contaminated with oocysts shed in the feces of infected cats. Because these exposures are often unrecognized, the specific route of transmission is frequently indeterminate. 7 The wide variations in T. gondii seroprevalence across populations appear to reflect variations in dietary habits and sanitation practices, supporting the idea that the oral route is the predominant pathway of infection. Outbreaks of toxoplasmosis in both humans and livestock have been linked to contact with environments contaminated by infected cats, highlighting the role of feline oocyst shedding in disease transmission. Additionally, multiple human outbreaks have also resulted from consuming untreated water sources. Current evidence shows no transmission occurs through breastfeeding or direct human-to-human contact, with the notable exception of vertical transmission from infected mothers to their unborn children during pregnancy. 8
T. gondii’s neurotropism is well documented, with latent cysts persisting in the CNS of seropositive individuals. In most, these remain clinically silent; however, their reactivation is often indicative of profound impairment in cell-mediated immune function. CNS involvement is most often documented in immunosuppressed individuals, particularly those with HIV/AIDS, hematological malignancies, or organ transplants.6,9 However, cases in patients receiving immunosuppressive therapy for autoimmune or inflammatory diseases, without HIV infection are less frequently reported. This case report contributes to the growing recognition of CNS toxoplasmosis as a possible, albeit rare, complication in non-HIV immunosuppressed patients and the potential for reactivation of latent toxoplasmosis in the CNS among patients on MMF therapy.
Mycophenolate serves as an essential immunosuppressive medication for preventing organ transplant rejection and managing various autoimmune and rheumatologic conditions. As a prodrug, it converts to mycophenolic acid, which selectively and reversibly blocks inosine monophosphate dehydrogenase, a critical enzyme required for de novo guanine nucleotide production. Through this mechanism, mycophenolic acid prevents T and B lymphocyte proliferation and development, resulting in suppressed cellular and humoral immunity. Unlike other transplant medications, MMF possesses the distinctive capability to simultaneously inhibit both T-cell and B-cell functions.10,11 Beyond its effects on lymphocytes, MMF also directly affects endothelial cells by interfering with leukocyte adhesion processes and reducing the migration of lymphocytes and monocytes into sites of inflammation.11,12
Although MMF’s immunosuppressive properties prove advantageous for managing autoimmune and rheumatological disorders, these same effects compromise the body’s natural defense mechanisms against pathogens, creating potential risks. Studies examining smaller patient cohorts have reported infection rates between 16% and 44% among MMF users.11,13 It is well established that higher doses of mycophenolate are associated with an increased risk of infections. However, the relationship between the duration of therapy and infection risk remains unclear. Cytomegalovirus and herpes zoster represent the most frequently observed infections in patients receiving this medication. 13
The medical literature contains <10 reported instances of CNS toxoplasmosis linked to MMF therapy, with the majority occurring in patients receiving multiple immunosuppressive medications simultaneously.14,15 Notably, only 1 documented case involved patient using MMF as their sole immunosuppressive treatment, demonstrating that even MMF monotherapy can predispose individuals to developing CNS toxoplasmosis. 15 A summary of the reported cases is provided in Table 2.6,14-19
Reported Cases of Central Nervous System Toxoplasmosis Associated With Mycophenolate Mofetil Therapy.
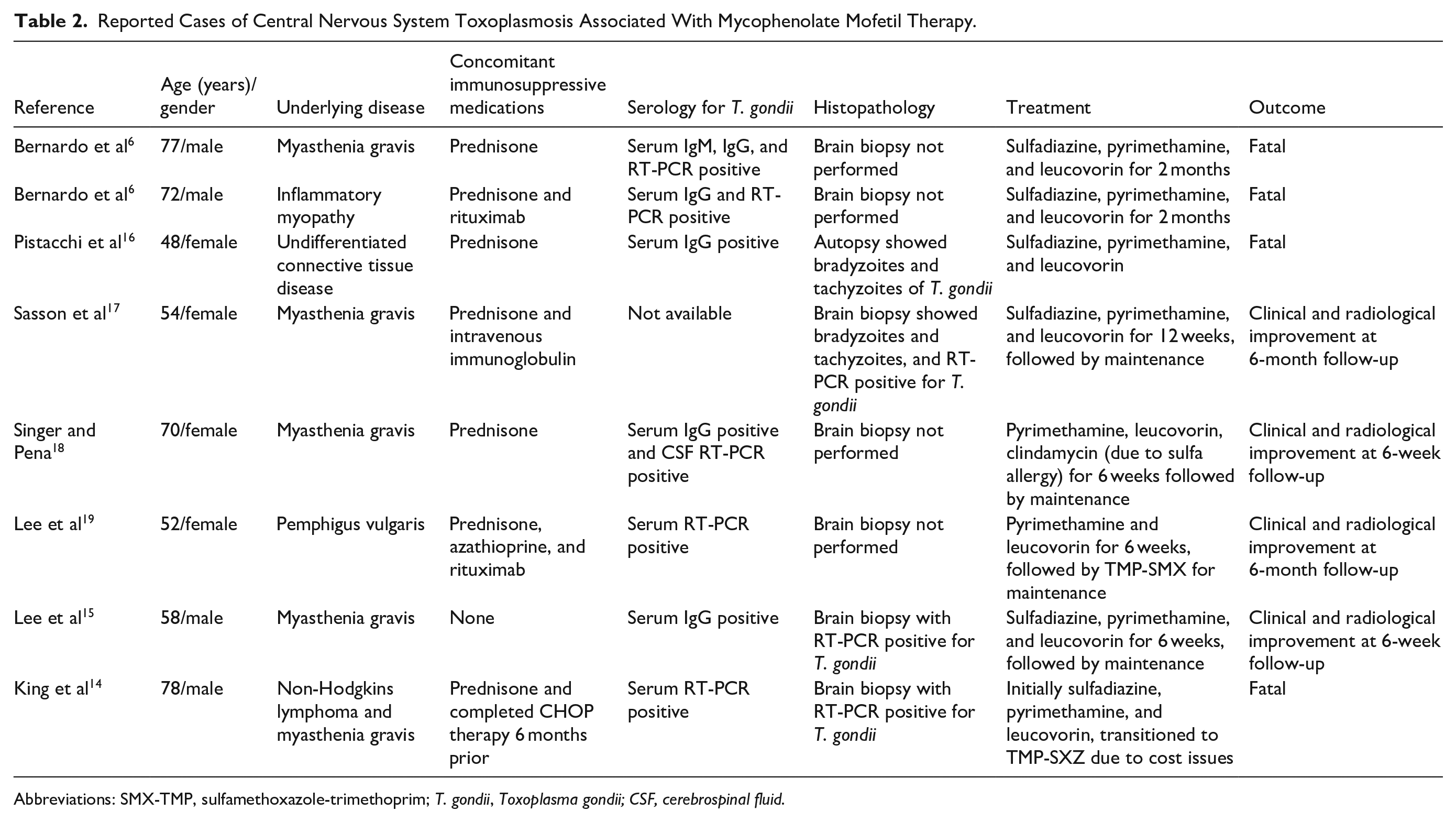
Abbreviations: SMX-TMP, sulfamethoxazole-trimethoprim; T. gondii, Toxoplasma gondii; CSF, cerebrospinal fluid.
Lee et al reported the case of a 58-year-old male with type II diabetes mellitus and myasthenia gravis, undergoing treatment with MMF. The diagnosis was primarily supported by MRI findings of ring-enhancing brain lesions, elevated Toxoplasma IgG levels, a positive PCR for Toxoplasma on brain biopsy, and a favorable response to Toxoplasma-targeted therapy. Interestingly, histopathological examination did not reveal tachyzoites or cysts containing bradyzoites. 15 It is important to note that a positive PCR result from brain tissue alone is not definitive for active Toxoplasma infection, as the CNS can harbor latent bradyzoites in individuals with prior exposure.20,21 Although the described patient lacked a definitive diagnostic test for cerebral toxoplasmosis, the strong clinical improvement following empirical anti-Toxoplasma treatment supported the diagnosis. Our case appears to be the first documented instance of CNS toxoplasmosis definitively diagnosed in the context of MMF monotherapy, with brain biopsy confirming the presence of T. gondii tachyzoites.
Treatment in this case appropriately diverged from HIV-centered protocols. SMX-TMP was chosen over pyrimethamine-based regimens to optimize efficacy while also considering patient compliance, affordability, and tolerability. Discontinuation of MMF was critical, and the patient’s improvement supports the decision to withhold further immunosuppression. In settings of autoimmune disease, this can be a delicate risk-benefit calculus, but in this case, the priority was clearly control of the infection.
Conclusion
This case underscores the importance of maintaining a heightened clinical vigilance for Toxoplasma infection, even in the absence of traditional risk factors. MMF, even when used as a single-agent immunosuppressant, can cause sufficient immune suppression to allow for reactivation of latent toxoplasmosis. As the use of immunosuppressive therapies in autoimmune conditions continues to rise, there remains a significant gap in understanding the prevalence of toxoplasmosis and how to assess risk in this population. Currently, there are no established guidelines for screening latent toxoplasmosis prior to initiating immunosuppressive therapy, and the potential role of chemoprophylaxis in these patients has yet to be defined. 22
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
