Abstract
Immunotherapy-induced pseudomembranous colitis (PMC) is an uncommon but increasingly recognized adverse effect of immune checkpoint inhibitors, particularly in patients with advanced malignancies. We present a case of a 68-year-old male with gastric adenocarcinoma undergoing treatment with immunotherapy and chemotherapy, who developed symptoms of PMC. Workup for Clostridium difficile and other common etiologies was negative. Colonoscopy revealed severe mucosal congestion and yellowish-green exudates, consistent with PMC. Based on the biopsy results and clinical presentation, after excluding common etiologies, immunotherapy-induced PMC was suspected. The patient responded to steroid therapy, with gradual improvement and a tapering regimen upon discharge. This case underscores the diagnostic challenges in identifying the etiology of PMC, particularly when it presents with diffuse involvement of the colon, which is an uncommon presentation for immunotherapy-related colitis. The overlap in clinical, endoscopic, and histopathological findings with other forms of colitis, such as Clostridium difficile infection (CDI) and inflammatory bowel disease, highlights the need for heightened awareness among clinicians. This case highlights the diagnostic challenges in recognizing immunotherapy-induced PMC, particularly with atypical, diffuse colonic involvement. The overlapping features with other colitis make timely diagnosis difficult. Further research is needed to refine diagnostic criteria and management strategies for immunotherapy induced colitis (IMC).
Introduction
Pseudomembranous colitis (PMC) is a severe inflammatory disease of the colon, typically characterized by yellow-white plaques forming pseudomembranes on the colonic mucosa. PMC is most commonly associated with Clostridium difficile infection, which occurs due to an imbalance in gut microbiota following prior antibiotic use. 1 Over the years, the incidence of PMC has increased, and several other etiologies have been identified, beyond C. difficile infection. These include ischemic colitis, severe collagenous colitis, inflammatory bowel disease, and infections due to other bacterial, viral, and parasitic organisms such as Strongyloides stercoralis and Entamoeba histolytica. 2 Other less common causes include Behcet’s disease, chemotherapy or immunotherapy, and toxins. 3
Immune checkpoint inhibitors (ICIs) have emerged as a widespread form of immunotherapy. With the increasing use of immunotherapy and its diverse applications, PMC is now recognized as being associated with ICIs. The presentation of PMC due to immunotherapy closely resembles that of C. difficile-induced PMC, and more often than not, immunotherapy-induced PMC is diagnosed after excluding other obvious causes. 4 In this article, we present a case of PMC initially presumed to be caused by C. difficile, but later concluded to be likely immunotherapy-induced colitis based on biopsy and endoscopic findings.
Case Description
We present a 68-year-old male patient with a past medical history of grade IV gastric adenocarcinoma (on active chemotherapy and immunotherapy), chronic lymphocytic leukemia, and recently diagnosed deep venous thrombosis. The patient initially presented to the hospital with 2 episodes of bright red blood per rectum and cramping abdominal pain. On admission, vital signs were stable: a temperature of 97.7 °F, heart rate of 71 beats/minute, blood pressure of 132/76 mmHg, respiratory rate of 19/minute, and oxygen saturation of 99% on room air. Physical examination revealed palpable purpura on the abdomen, diffuse abdominal tenderness, and maroon-colored stool on digital rectal exam. Laboratory results showed a hemoglobin level of 12.5 g/dL, platelet count of 151 K/cmm, white blood cell count of 41 000/cmm (with an absolute neutrophil count of 1000), and normal liver function tests. Imaging studies, including a Computed tomography (CT) scan of the abdomen and pelvis with oral contrast, revealed pancolonic circumferential wall thickening consistent with colitis, as well as small-volume abdominopelvic ascites, mesenteric edema, and unchanged gastric wall thickening (Figures 1 and 2).

CT imaging showing pancolonic circumferential wall thickening likely suggestive of colitis with arrows pointing the thickened bowel wall.
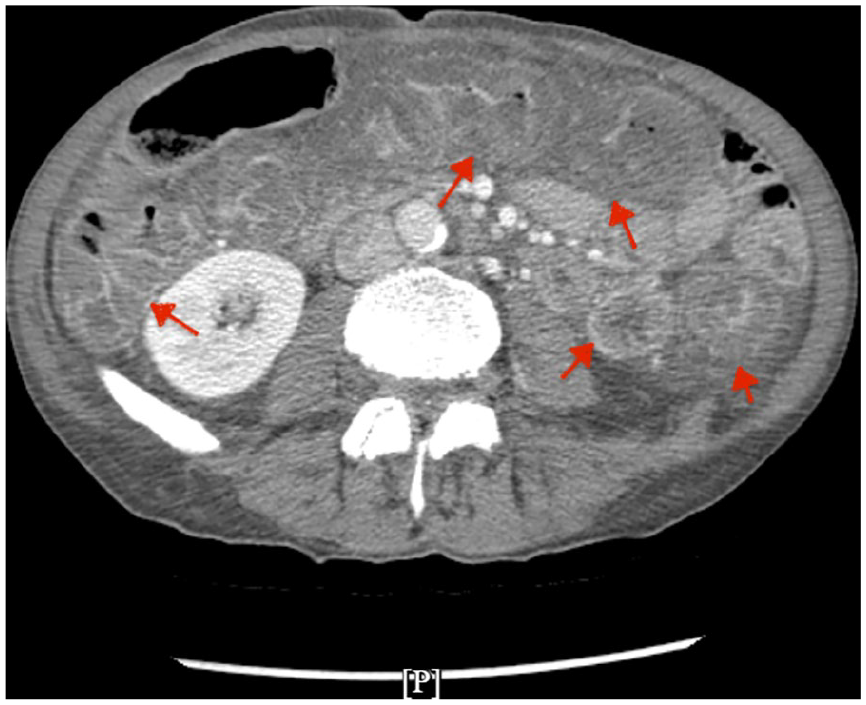
Transverse section of CT abdomen CT showing circumferential wall thickening in colon, indicated by red arrows pointing the bowel wall.
The patient was admitted, anticoagulants were held, and piperacillin-tazobactam was started empirically based on the imaging findings suggestive of colitis. A colonoscopy was performed the following day after bowel preparation and it revealed severe mucosal changes, including congestion (edema) and yellowish-green exudates, suggestive of PMC in the sigmoid and descending colon, starting at 30 cm from the anal verge (Figures 3–5). Normal mucosa was noted in the rectum and rectosigmoid colon. The scope was not advanced further due to severe inflammation, and superficial biopsy samples were obtained. Fecal calprotectin and stool microbiology (including tests for common gastrointestinal pathogens Escherichia coli, Salmonella, Shigella, Campylobacter, Trichomonas, Cyclospora, and also C. difficile, Epstein–Barr virus, and cytomegalovirus PCRs) were sent, and a vasculitis workup was performed due to the presence of palpable purpura and the findings of colitis on colonoscopy.

Colonoscopy image of the sigmoid colon showing yellowish pseudomembranes (white arrows) widely across the colonic mucosa (represented by pink mucosa in the red circle).

Colonoscopy image of the descending colon showing yellowish pseudomembranes (white arrows) circling almost entirely through the descending colon with sparse colonic mucosa (red circle).

Colonoscopy image. Descending colon showing pseudomembranes (indicated by white arrows).
Initially, C. difficile was considered the leading differential diagnosis due to the extensive pseudomembrane formation and significant leukocytosis. The low suspicion for ischemic colitis was based on the absence of a cardiac history and classic CT abdomen findings. Given the colonoscopy results, inflammatory bowel disease and collagenous colitis were also excluded. The patient was started on oral vancomycin and metronidazole while waiting for biopsy and stool microbiology results. After a few days, stool microbiology, including tests for C. difficile, returned negative, and fecal calprotectin was also negative.
After excluding all common causes, immunotherapy-induced PMC became the leading differential diagnosis. The patient was receiving nivolumab, a PD-L1 inhibitor, for his gastric adenocarcinoma, in combination with chemotherapy (5-Fluorouracil, Leucovorin, Oxaliplatin, and Nivolumab) which was started in January 2024. Biopsy results showed benign colonic mucosa with mild hyperplastic changes, fresh hemorrhage, and edema, but no signs of active inflammation. Immunotherapy-induced PMC was strongly considered based on the clinical presentation, the timeline of the presentation and after exclusion of other causes. The patient was started on weight-based oral prednisone (1 mg/kg/day) for moderate ICI-induced colitis, and symptoms improved. Steroid therapy was gradually tapered as the patient’s condition improved dramatically. The patient was discharged with a tapering steroid regimen and advised to hold immunotherapy following shared decision-making with the oncology team. The patient was followed up outpatient 1 week after discontinuation of the therapy and reported complete resolution of his symptoms including diarrhea. Patient followed up with his oncologist outpatient, and they decided to completely discontinue the Nivolumab and optimize his chemotherapy.
Discussion
PMC is a severe manifestation of colonic disease, most commonly associated with C. difficile infection, but can be caused by various other etiologies. 5 Pathologically, PMC is triggered by local mucosal damage, leading to small areas of epithelial necrosis. The eruption of neutrophils, nuclear debris, and other inflammatory elements from the lamina propria onto the epithelium results in pseudomembrane formation.2,3
Common symptoms of PMC include watery diarrhea with pus or mucus in the stool, abdominal pain/cramps, and fever, with leukocytosis being a frequent sign. Symptoms can appear as early as a few days and up to 6 weeks after starting antibiotics due to alterations in the gut flora. 3
There are various causes of PMC, including C. difficile infection, ischemic disease, inflammatory bowel disease, infections from bacterial organisms (eg, Staphylococcus aureus, Klebsiella oxytoca, E. coli), viral infections (eg, Cytomegalovirus (CMV), coronavirus), and parasitic infections (eg, S. stercoralis, E. histolytica). 2 Immunotherapy-induced PMC is a rare but significant cause.
ICIs include several types of drugs such as anti-CTLA-4 agents (eg, ipilimumab, tremelimumab), anti-PD-1 agents (eg, nivolumab, pembrolizumab), and anti-PD-L1 agents (eg, avelumab, atezolizumab, and durvalumab).4,6 These drugs increase T-cell activity and enhance the immune response against neoplastic cells. They are increasingly used in the treatment of advanced-stage malignant tumors, but they also interfere with the host immune system, including the gastrointestinal system, leading to adverse effects ranging from nausea and diarrhea to colitis, which can be a severe manifestation.7-10
Immunotherapy-induced colitis occurs due to the activation of tissue-resident CD8+ T cells in the colon, which is triggered by checkpoint inhibitor treatment. This activation recruits additional CD4+ and CD8+ T cells, leading to rapid proliferation and inflammation. Elevated levels of IFNγ contribute to this response by promoting immune cell recruitment and amplifying inflammation, explaining the rapid onset and severity of colitis in these patients. 11
This case presents an atypical presentation of PMC involving diffuse colonic inflammation, which is rare in immunotherapy-induced colitis. Previous reports of immunotherapy-induced colitis exist in the literature,4,5,7-11 but the presentation often varies, and there is diagnostic uncertainty as the clinical, endoscopic, and histopathological findings can overlap with other etiologies such as infections, medications, and inflammatory bowel disease. 5 In immunocompromised patients, particularly those on chemotherapy, C. difficile infection is often the first differential when presented with a high white blood cell count and PMC.
Immunotherapy-induced PMC is rarely considered a differential diagnosis because the other etiologies for PMC dominate. The diagnosis is often made clinically by the treating oncologist based on symptom presentation, and corticosteroids are typically initiated without histological confirmation or use of noninvasive biomarkers such as fecal calprotectin or lactoferrin. 12 While previous cases of immunotherapy-induced colitis have been reported, this case is particularly unique in that it presents with diffuse PMC, which is not typical and not often considered as a primary differential following endoscopy.
PMC solely attributed to immunotherapy is extremely rare. Only 1 previously documented case of immunotherapy-induced PMC exists, involving pembrolizumab in the treatment of hepatocellular carcinoma. 6 To our knowledge, our case represents the first documented instance of nivolumab-induced PMC. The challenge of diagnosing immunotherapy-induced colitis lies in its overlap with other causes, and timely recognition is critical to avoid severe complications.
Conclusion
This case highlights the challenging nature of diagnosing immunotherapy-induced PMC (IMC), particularly when it presents with atypical, diffuse involvement of the colon. Although PMC is most commonly attributed to C. difficile infection or other infectious and inflammatory causes, this case underscores the need for clinicians to consider immunotherapy as a potential cause, especially in patients undergoing treatment with ICIs. As the first documented instance of nivolumab-induced PMC, this case contributes to the growing body of literature and calls attention to the importance of considering less-common etiologies in immunocompromised patients presenting with colitis. The overlapping clinical, endoscopic, and histopathological features between various forms of colitis necessitate a high index of suspicion, and further research is needed to better define diagnostic criteria and optimize management strategies for IMC. This case serves as a reminder of the evolving spectrum of side effects associated with modern cancer immunotherapy and the need for vigilance in diagnosis and treatment.
Footnotes
Acknowledgements
We would like to express our gratitude to the clinical staff at The Brooklyn Hospital Centre for their exceptional care and support during the management of the patient described in this case report. We also thank our colleagues for their insightful discussions and contributions to the development of this manuscript. Lastly, we appreciate the guidance and resources provided by the Institutional Review Board, which facilitated the ethical approval for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Permission to report this case was granted by the Institutional Review Board of The Brooklyn Hospital Center number: 2297805-1.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
