Abstract
Immune Checkpoint Inhibitors (ICIs) are monoclonal antibodies that block inhibitory immune targets, such as cytotoxic T lymphocyte antigen 4 (CTLA-4), programmed cell death protein 1 (PD-1), and programmed death ligand 1 (PD-L). Pembrolizumab targets the PD-1 receptor of lymphocytes in lung cancer treatment. ICI checkpoint blockade enhances immunity against cancer cells. However, loss of immunoregulatory control can cause autoimmune reactions in various organs, leading to immune-related adverse events (irAEs). The most common irAE is ICIs-induced colitis, which usually develops 6–8 weeks after ICI initiation and can involve any part of the gastrointestinal system. Herein, we report a presentation of pembrolizumab-induced colitis in a female patient with metastatic lung cancer and review the most recent findings in the model of checkpoint-induced colitis. It was interesting to learn that the colon mucosa may show normal macroscopic findings, but microscopically, immunotherapy-induced autoimmune colitis could be present. Additionally, patients with grade 2 or higher symptoms should have a colonoscopy, receive systemic corticosteroids as treatment, and, based on their response, receive biologic therapy. Here, we present a case report of in a 45-year-old female who has been a smoker for 25 years, without comorbidities, and with metastatic lung cancer who developed colitis after the seventh cycle of pembrolizumab. This case presentation highlights the importance of early recognition and appropriate intervention in order to prevent permanent interruption of treatment with checkpoint inhibitors, as well as prevention of colitis complications.
Keywords
Introduction
Since 2011, the FDA (Food and Drug Administration) has approved several ICIs, including CTLA-4 inhibitors (ipilimumab), PD-L1 inhibitors (atezolizumab, avelumab, and durvalumab), and PD-1 inhibitors (nivolumab and pembrolizumab). These ICIs showed efficacy for many cancers, including advanced-stage melanoma, renal cell carcinoma, triple negative breast cancer (TNBC), gastrointestinal cancers and non-small cell lung carcinoma (NSCLC). 1 Immune checkpoint inhibitors (ICIs) are linked to organ-specific adverse events (AEs), which differ from those associated with conventional chemotherapies and multikinase inhibitors. 2 These adverse events present as autoimmune or inflammatory disease-like reactions and are termed immune-related adverse events (irAEs). 3 IrAEs occur as a result of overstimulation of the immune system by blockade of ICIs. The occurrence of irAEs depends on the tumor type, agent, dose, and patient characteristics. The median of IrAEs is about 40 days and they may be described as “acute” if they develop during treatment, “delayed” if they appear following ICI completion, and “chronic” if they persist beyond 12 weeks after discontinuation of therapy. 4 Any organ system can be affected by IrAEs, therefore iAEs such as pneumonitis, myocarditis, pericarditis, hepatitis, pancreatitis, vasculitis, acute coronary syndrome, myositis and adrenal insufficiency as well as skin toxicities. 5 Additionaly, haematological toxicities, neurological toxicities have been noted so far. 1 One of important IrAEs is colitis which could be one of the leading causes of hospitalization and quality of life deterioration during ICI treatment. 6 Pembrolizumab, a monoclonal humanized anti-PD-1 antibody, is used to treat metastatic NSCLC and other conditions like TNBC, melanoma, and gastrointestinal cancers. 6 Researchers report a higher incidence of ICI-induced colitis with a combination of ICI therapy. 4 The incidence of colitis due to the use of PD-1 inhibitors, in the first place pembrolizumab, is 1.2%–0.2%, and the incidence due to the use of PD-L1 inhibitors is 0.3%–0.004%. Among PD-1 inhibitors, such as pembrolizumab, and PD-L1 inhibitors, the incidence of mild colitis is more frequent using PD-1 than PD-L1 (1.2% vs 0.3%); for severe colitis, there is no significant difference between pembrolizumab and PD-L1 inhibitors. 4 In ICI-induced colitis, CD8+ Tissue Resident Memory T cells (TRM CD8+ cells) differentiate into cytotoxic T lymphocytes (TLy CD8+) that can release interferon-gamma (IFNγ) and Tumor Necrosis Factor—alpha (TNF-α). This damages and inflames the intestinal epithelial barrier. 7 Colonization of the mucous membranes by TLy CD8+ explains why colitis is a common and early adverse event during immunotherapy. The increased concentration of these lymphocytes in the intestinal mucosa could explain the sudden response to the corticosteroid, which induces apoptosis of activated T lymphocytes. 7 Diarrhea/colitis is the most common adverse effect and the most common reason for discontinuing ICI therapy. 8 After the first administration of ICI, the median interval to onset of diarrhea is approximately 4–8 weeks. 8 Some patients experience symptoms as early as 1 week after exposure. Some patients developed symptoms months or even 2 years after discontinuation of the therapy. 8 Clinical presentations are usually non-specific and include fever (46%), hematochezia (64%), abdominal pain (82%), diarrhea (92%), and vomiting.8,9 Pembrolizumab and others ICIs can cause either mild, moderate, or severe forms of colitis. Some patients could experience rapid progression to serious complications including bowel perforation and even death.4,8,9 Experts commonly use the Common Terminology Criteria for Adverse Events, Version 5, to assess and grade the severity of the disease. Grade 1 denotes mild symptoms, while grade 5 signifies the patient’s death due to ICI-induced colitis. 10 Grades 1, 2, and 3 of diarrhea differ in the number of stools: less than four, between four and six, and over seven stools per day (Table 1). Life-threatening consequences (intestinal ischemia, necrosis, bleeding, toxic megacolon, perforation, and systemic shock) characterize Grade 4, indicating the need for urgent intervention 11 (Table 1). Once infection, inflammatory bowel disease (IBD) and tumor metastasis are ruled out, Checkpoint Inhibitor Induced Colitis (CIC) can be diagnosed. Before starting treatment, the gold standard for diagnosis is endoscopy with biopsy.12,13 Endoscopic findings may vary from normal-appearing mucosa to edema and ulcerations. Additionally, endoscopic findings serve as the primary predictors in determining the response of ICI-induced enterocolitis to treatment. 13 Pathohistological patterns of CIC could be classified as follows: acute active colitis, chronic active colitis, microscopic (collagenous and lymphocytic) colitis, graft-versus-host disease-like (GVHD), and other types (mixed type, increased apoptosis, ischemic colitis-like, and non-specific inflammatory reactive changes (NSIRC)) 9 (Table 2).
CRP: C reactive protein; FCP: fecal calprotectin; PCR: polymerase chain reaction; HBP Ag: helicobacter pylori antigen.
LP: lamina propria; Ly-s: lymphocytes; GVHD: Graft-Versus-Host Disease-like.
It is stated that the use of budesonide formulated to be released specifically in the colon may play a role in the treatment of patients with microscopic ICI-induced colitis. 7
Timely diagnosis and adequate treatment of ICI-induced colitis are crucial for an optimal outcome. 15
Global perspective of immune-checkpoint inhibitor toxicity over a decade
We summarized irAEs from VigiBase, the international pharmacovigilance database, reported from 2008 until January 2023, to characterize trends in irAEs reporting, clinical features, risk factors, and outcomes16,17 and to update the information previously described by Wang at al. 18
Each case was examined for its administrative data (date, reporter qualifications, and reporting region), patient demographics (sex, age, cancer type), treatment characteristics (immune checkpoint inhibitors and other anticancer medications, prescription indications, initiation and termination dates, administration route), and details of adverse reactions (reported terms, onset and end dates, severity, outcomes including fatalities and progression following drug discontinuation, and any rechallenge).
Population
The most prevalent cancer types associated with irAE were non-small cell lung cancer (15,397/44,589, 34.5%), melanoma (14,630/44,589, 32.8%), and renal cancer (4265/44,589, 9.6%). In 62.4% of patients (31,409/50,347), anti-PD1 or anti-PDL1 monotherapies were administered, while 12.8% (6422/50,347) received anti-CTLA4 + antiPDL1 therapies. A combination of anti-CTLA4 + antiPD(L)1 therapies was included in 16.3% of patients (8215/50,347). The evolution of ICI regimen and cancer categories associated with irAE varied significantly over the four studied time periods of reporting in VigiBase (⩽2016, 2017–2018, 2019–2020, 2021–2022). The proportion of anti-CTLA4 monotherapy-associated irAE almost vanished representing 47% of prescriptions of ICI before 2017 versus 1.6% in 2021–2022 (P < 1 × 10−160), in contrast to combination of anti-CTLA4 + anti-PD(L)1 in the same time periods (8.9% vs 20%, P = 2 × 10−90).
The prevalence of irAE
In total, 60,323 distinct irAE were identified in 50,347 cases. Skin reactions (22.9%, 11,537/50,347), pneumonitis (18.5%, 9317/50,347), enterocolitis (14.4%), and thyroiditis (12.1%, 6070/50,347) were the most reported irAEs. The proportion of gastro-intestinal irAE (i.e. esogastro-enterocolitis) decreased significantly, representing 23% of total irAE before 2017 compared to 12% in 2021–2022 (P = 7 × 10−53). This was in contrast to a significant increase in myotoxicities (i.e. myocarditis, myositis, or myasthenia gravis, 3.7% vs 8.3%, P = 6 × 10−43) and pancreatico-hepatic irAE during the same time periods (8.2% vs 11%, P = 6 × 10−15).
Time to onset and fatality
The median TTO of irAE from the initiation of ICI therapy was 1–9 months. The median TTO for myotoxicities was the shortest, with a range of 22–60 for myasthenia gravis, 31 days for myositis, and 33 days for myocarditis and 65 days for colitis. Severe cutaneous adverse reactions (SCAR) also had one of the shortest TTOs (40 days, IQR = 14–119). Except for arthritis (104 days, IQR = 31–224), diabetes (114 days, IQR = 45–243), pancreatitis (121 days, IQR = 41–261), esogastritis (126 days, IQR = 45–295), sarcoidosis (141 days, IQR = 75–274), vitiligo (170 days, IQR = 89–326), and skin bullous auto-immune reactions (273 days, IQR = 93–487), the majority of other irAE had a median TTO within 1 and 3 months.
In the narrow irAE population, death occurred in 5709 out of 50,347 individuals, representing 11.3%. The highest overall irAE fatality rates were observed in myocarditis (27.6% and 19.2%, respectively), myasthenia gravis (23.1% and 13.6%), SCAR (22.1% and 12.3%), myositis (21.9% and 11.3%), pneumonitis (21.0% and 10.4%), and encephalomyelitis (18% and 10.9%). The overall reported fatality rates for most life-threatening irAEs decreased over time, with the exception of SCAR and myasthenia gravis which compares rates across the periods ⩽2016, 2017–2018, 2019–2020, and 2021–2022. The overall fatality rates for most other irAEs ranged from 5% to 15%, while the irAE-related fatality rates ranged from 2% to 5%. Arthritis, hypophysitis, diabetes, vitiligo, skin bullous reactions, uveitis, and sarcoidosis exhibited the lowest overall fatality rates (<5%) and very low irAE-related fatality rates (<2%). In the multivariate analysis, myotoxicities, pneumonitis, SCAR, and encephalomyelitis demonstrated a stronger association with overall mortality (OR = 1.7–3.3) relative to other irAE types. In addition to irAE types, factors contributing to increased mortality included age (OR = 1.16, 95% CI = 1.1–1.2 for individuals aged ⩾65 years compared to those aged <65 years), geographic location (eastern Asia versus the rest of the world, OR = 1.9, 95% CI = 1.7–2.0), and specific cancer types, with odds ratios ranging from 1.5 to 2.7 for urothelial, gastro-oesophageal, head and neck, lung, and liver cancers, listed in order of increasing OR. Factors linked to reduced fatality rates included female gender (OR = 0.85, 95% CI = 0.8–0.9 compared to male) and the most recent reporting period (OR = 0.53, 95% CI = 0.5–0.6 for 2017–2022 compared to ⩽2016).
Drug discontinuation and rechallenge
Among the 60,323 identified specific irAE within the 50,347 narrow cases, 27,471 irAE within 23,942 cases had documented outcomes regarding both ICI discontinuation and irAE resolution. A total of 88.1% of irAEs were managed through ICI discontinuation, with 73.9% of cases classified as resolved. The resolution rate for each irAE (in the subset of non-fatal cases) varied between 32.5% and 88.5%. The resolution rate for most irAEs was 70%, with the exceptions of peripheral neuropathy at 65% and vitiligo at 32.5%, which exhibited lower resolution rates. The recurrence rate for specific irAEs, based on 10 available cases, varied between 9.5% and 62.5%. The highest recurrence rates were noted for skin bullous reactions (62.5%), nephritis (36.8%), myocarditis (37.5%), enterocolitis (35.6%), and pneumonitis (32.2%).
Gastrointestinal irAEs induced by ICIs
The latest EudraVigilance and VigiAccess databases indicate that colitis (12,581) and diarrhea (12,108) are the most frequently reported gastrointestinal adverse events. 19 Furthermore, in both databases, the highest number of adverse drug reaction (ADR) reports is linked to nivolumab and pembrolizumab. Durvalumab (ROR: 3.96, 95% CI: 3.65–4.28), ipilimumab (ROR: 1.95, 95% CI: 1.89–2.01), nivolumab (ROR: 1.05, 95% CI: 1.02–1.07), and atezolizumab (ROR: 1.04, 95% CI: 1.01–1.07) exhibited increased risks of gastrointestinal events relative to other immune checkpoint inhibitors. The analysis of EudraVigilance revealed dysphagia, ascites, hematochezia, and gastroesophageal reflux disease as potential signals linked to ICI therapy. A significant proportion of ADR reports (87.2%) consisted of serious gastrointestinal adverse events, with a subset resulting in fatal outcomes (14.5%). Atezolizumab (14.9%), and pembrolizumab (11.9%) demonstrated a greater incidence of fatal outcomes relative to other immune checkpoint inhibitors.
Accordingly, we emphasize the paramount importance of our case report, especially in terms of the need to understand the management of pembrolizumab-induced colitis.
Known facts
Checkpoint inhibitors like pembrolizumab, atezolizumab, and durvalumab have been approved for the treatment of advanced NSCLC in patients whose tumor cells have 50% or higher levels of PD-L1 expression. If tumor cells have PD-L1 protein between 1% and 49%, pembrolizumab and other checkpoint inhibitors are used in combination with chemotherapy to treat NSCLC. 1
Pembrolizumab can cause immune-related adverse events such as pneumonitis, hepatitis, dermatitis, hypophisitis, pancreatitis, and colitis. 20
A new point of view
The patterns of ICI reintroduction included the same ICI utilized before, the same type of ICIs, and the switching of ICIs of distinct types. The safety and efficacy of rechallenging ICIs in NSCLC patients, as reported in previous studies. They discovered that switching ICI class therapy produced a superior outcome, with over 50% of patients experiencing stable or partial regression disease. 21
Gobbini et al. 22 demonstrated that NSCLC patients discontinuing for irAEs had more prolonged survival than those discontinuing for disease progression.
ICI-induced colitis is an exclusion diagnosis. Corticosteroids are the current first-line treatment, followed by infliximab for steroid-refractory colitis. Vedolizumab and fecal microbiota transplantation are promising options for treatment-refractory ICI-induced colitis. 8
Biological agents (IFX and vedolizumab) in the treatment of refractory microscopic colitis induced by ICIs
The corticosteroid response for grade 2–4 toxicity should be evaluated early in the treatment of immune-mediated colitis, after 2–3 days. Escalation to a biological agent is recommended for non-responders in the absence of peritonitis, hepatitis, or perforation (infliximab (IFX) 5 mg/kg). Researchers have confirmed that IFX, a TNF-α antagonist, significantly enhances tumor immunity and mitigates resistance to PD-1 inhibitors. 23 In instances where IFX is either ineffective or contraindicated, vedolizumab may be employed. Vedolizumab, an antagonist of a4b7 on CD4 + T cells, prevents T cells from aggregating and returning to the inflammatory intestinal mucosa. 300 mg infusions are administered to treat steroid-refractory cases at 0, 2, and 6 weeks, or until laboratory or clinical improvement. 9 Vedolizumab also induces a response in CIC patients with microscopic colitis confirmed by histopathology. The potential risk of tumor progression in patients with lymph node metastasis may be mitigated by the gastrointestinal selectivity of vedolizumab. 9 In addition, the risk of secondary tumors in susceptible individuals was not elevated by the use of vedolizumab, which possesses the same potential as IFX. Abu-Sbeih et al. 24 discovered that over half of the 179 CIC patients who received IFX or vedolizumab experienced a significantly lower hospitalization, shorter symptom duration, shorter steroid taper/taper attempt, a higher success rate of steroid taper, decreased fecal calprotectin levels, a promoted histological regression rate, and a decreased risk of infection.
Gastrointestinal microbiota as an important factor in the development of ICI-induced colitis
The development of CIC and the response of tumors to ICI therapy may be associated with gut microbiome characteristics. In ICI-induced colitis, the microbiota diversity was significantly reduced, particularly in certain genera within the Firmicutes phylum, such as Faecalibacterium prausnitzii (F. prausnitzii) and Bacteroides fragilis (B. fragilis). The presence of colitis may be linked to the enrichment of F. prausnitzii. F. prausnitzii’s typical function is to maintain the integrity of the colonic mucosa as an obligate anaerobe. 9 The recruitment of Tregs and a4b7 T cells was facilitated by the increase of F. prausnitzii, which was associated with T-cell proliferation in the gastrointestinal mucosa. Furthermore, it has the potential to increase the concentration of CTLs in the tumor microenvironment, thereby extending the progression-free survival (PFS) and overall survive (OS). The protective factor of CIC is the increase in Fragilis. It functions as an anti-inflammatory agent in the gastrointestinal tract. 9 Similar experiments conducted with PD-L1 inhibitors demonstrated that Bifidobacterium species could mediate antitumor efficacy. 25 This bacterium has the potential to reduce gastrointestinal inflammation that is induced by Tregs without compromising the antitumor response. 25 Consequently, the development of CIC may be influenced by the species and relative abundance of intestinal microbiota. The inflammation initiation may be orchestrated by certain microbial species, while other subsets may contribute to the perpetuation of CIC. Additional efforts should be made to further understand the intricate symbiosis between bacteria and ICI-induced colitis, with the ultimate objective of manipulating the microbiota to achieve a balance between the tumor response and irAEs. It is hypothesized that the intestinal microbiome is a critical factor in the development of CIC. Numerous gut microbiomes are associated with the induction or alleviation of ICI-induced colitis. Fecal transplantation has been suggested as an optional therapeutic approach for patients with severe or refractory CIC, as it contributes to the reconstitution of the gut microbiome and the relative increase of regulatory T cells within the colon mucosas.4,8,9
Patients with IBD and risk from development of ICI-induced colitis
In comparison to non-IBD patients, patients with IBD (Crohn’s disease and ulcerative colitis) are at an elevated risk for GI irAEs. However, these events can be controlled, and these patients necessitate appropriate monitoring. The median time to exacerbation that necessitated corticosteroids in a recently published case series of 13 IBD patients was 5 months following the initiation of the ICI. In a retrospective study, the effects of ICIs on tumor response were consistent between patients with and without IBD. 26
Case presentation
Here, we describe a female patient with metastatic lung cancer who developed colitis after the seventh cycle of pembrolizumab. The patient did not suffer from IBD and it was exclusion criteria in this case.
Case description
In August 2023, a 45-year-old female presented to us with metastatic lung cancer. The first complaints in the form of frequent headaches, forgetfulness, and uncontrollable hand tremors appeared in July 2023. The patient had Eastern Cooperative Oncology Group Performans Status 1 (ECOG PS 1). She has been a smoker for 25 years, averaging 20 cigarettes a day, without comorbidities. The patient`s family history was positive for malignancy (her mother suffered from lung and cervical cancer). We performed an Magnetic resonance imaging (MRI) of the endocranium to establish a diagnosis. MRI of the endocranium (07/23) has shown an expansive frontal lesion in diameter of 3.9 × 3.4 × 3.8 cm, bordered by extensive perifocal vasogenic edema with a compressive effect on the surrounding brain parenchyma, and a smaller cerebellar lesion on the left hemisphere of the brain (Figure 1). On the initial Multislice Computed Tomography (MSCT) of the chest (08/23), a spiculated tumor of 28 × 31 × 29 mm was described (on the left at the level of the anterior segment of the upper lobe, posteriorly along the aorta, left a pulmonalis, and along the incision). Mediastinal lymphadenopathy was also described as draining lymph nodes, the largest diameter of which is about 25 × 16 mm (Figure 2). According to the eighth world TNM classification, the tumor belongs to T4N2M1, and it is staged as the 4th clinical stage. Operation of the tumor was performed in the area of the frontal region on the left hemisphere of the brain. The tumor on the left in the area of the cerebellum was treated with the X-knife.

MRI of endocranium (07/23). The first and second MRI images of this figure depict an expansive frontal lesion with a diameter of 3.9 × 3.4 × 3.8 cm, surrounded by extensive perifocal vasogenic edema that exerts a compressive effect on the surrounding brain parenchyma. The third MRI image of this figure depicts a smaller cerebellar lesion on the left hemisphere of the brain.

The initial MSCT of the chest (08/23) described a spiculated tumor of 28 × 31 × 29 mm on the left at the level of the anterior segment of the upper lobe.
The tumor biology
Pathohistological analysis of brain metastasis revealed primary lung adenocarcinoma.
Molecular testing:
– PD-L1—TPS (Tumor Proportion Score) 55%
– Anaplastic Lymphoma Kinase (ALK) expression—absent
– Epidermal Growth Factor Receptor (EGFR)—no changes were detected in the EGFR gene; wild type EGFR (wtEGFR gene).
A PD-L expression of more than 50% prompted the start of pembrolizumab treatment. Following four cycles of pembrolizumab, we conducted the first evaluation of the disease. Follow-up MSCT of the chest and abdomen (10/23) showed the first immune unconfirmed progressive disease (iUPD) (in the upper left lobe, the tumor had dimensions of 32 × 68 × 70 mm; Lymph Nodes (LNs) conglomerate from group 4L and 5 measured 37 × 45 × 40 mm; pericardial effusion was 14 mm; Figure 3a). Endocranial MRI (10/23) showed stable disease (SD). We performed another control scan after the seventh cycle of pemrolizumab. The MSCT of the chest and abdomen (01/24) revealed immune Stabile Disease (iSD), with the tumor in the upper left lobe measuring 51 × 33 × 60 mm, the LNs conglomerate from group 5 measuring 36 × 30 mm, and the LNs from group 4L measuring 11 mm, all without pericardial and pleural effusion (Figure 3b).
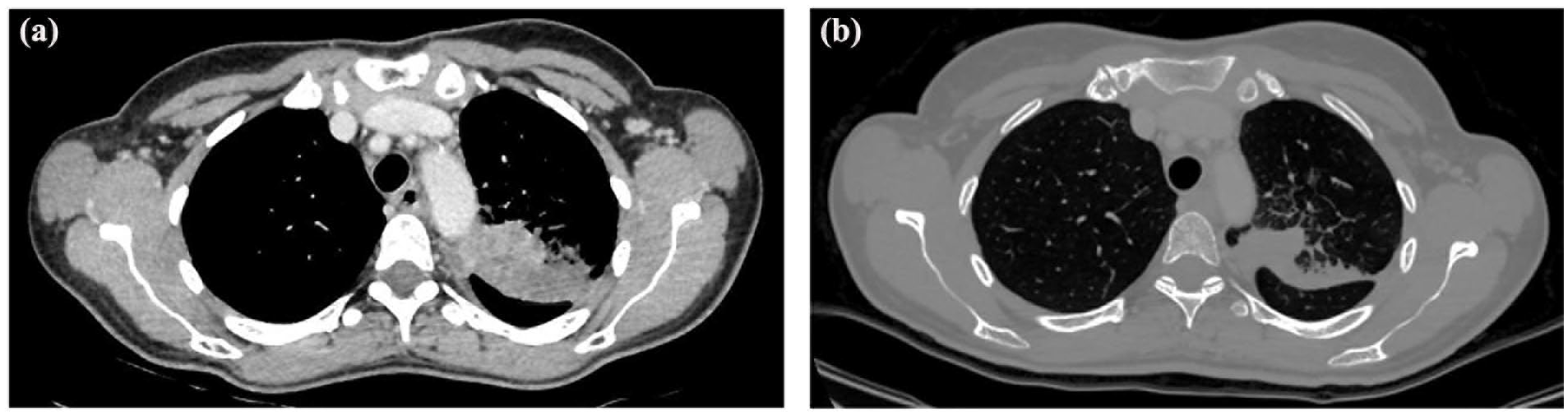
MSCT of the chest and abdomen. (a) On the left figure of the computed tomography (CT) scan (10/23) is shown iUPD (in the upper left lobe, the tumor had dimensions of 32 × 68 × 70 mm; LNs conglomerate from group 4L and 5 measured 37 × 45 × 40 mm; pericardial effusion was 14 mm). (b) On the right figure of the CT scan (01/24) is shown iSD with the tumor in the upper left lobe measuring 51 × 33 × 60 mm.
After the 7th cycle of pembrolizumab, due to persistent diarrhea, laboratory analyses were performed, as well as stool analysis (Table 3). Among the complaints, the patient mentioned six loose stools per day, malaise, and a depressed mood phase. The planned eighth cycle of immunotherapy was postponed.
Laboratory and stool analyses.

Ig: immunoglobulin; C3 and C4: complement component 3 and 4; TSH: thyroid-stimulating hormone; FT4: thyroxine; RF: rheumatoid factor; ANA: antinuclear antibody.
The patient was admitted to the Center for Internal Oncology’s chemotherapy department at the beginning of March 2024 to confirm the diagnosis of suspected autoimmune colitis caused by pembrolizumab.
Colonoscopy and macroscopic finding of the colon mucosa
During the hospital stay, a colonoscopy revealed a normal macroscopic finding of the colon mucosa (Figure 4). We took polytopic biopsies of the colon.

Colonoscopy, demonstrating a normal macroscopic finding of the colon mucosa.
Pathohistological analysis of the colon mucosa
The colon’s mucosa, which covered the epithelium and superficial parts, remained intact, showing no histological signs of cryptitis or crypt abscesses. The stroma was swollen and filled with a mild to moderate inflammatory infiltrate made up of lymphocytes, plasma cells, and a few eosinophilic granulocytes. We concluded with a diagnosis of mild to moderate colitis interstitialis—autoimmune (Figure 5).

Microscopic colitis interstitialis (autoimmune), mild to moderate degree: the colon’s mucosa remains intact, a mild-to-moderate diffuse inflammatory infiltrate, consisting of lymphocytes, plasma cells, and rare eosinophilic granulocytes, permeates the edematous stroma. Original magnification: (a) ×40, (b) ×100, and (c) ×200.
Treatment
In the treatment of our patient, we are guided by European Society for Medical Oncology (ESMO) recommendations. As our patient has grade 2 diarrhea, treatment involves the use of corticosteroid therapy such as prednisolone at a dose of 1 mg/kg. The patient received parenteral corticosteroid therapy (amp. Dexamethasone N0II/12h), potassium supplementation, infusion solutions, probiotic therapy, Loperamide tbl per the scheme, and proton pump blockers (PPI) at the time of hospitalization. A few days after applying the parenteral corticosteroid therapy, the diarrhea subsided. The patient’s clinical symptoms improved, leading to a switch to corticosteroids (Prednisolone) for 4 weeks, followed by a gradual taper and PPI during corticosteroid therapy. The patient is currently free of complaints, has normal laboratory and clinical findings, and continues treatment with pembrolizumab. The last follow-up scan described the disease as stable.
Discussion
ICIs can cause either mild, moderate, or severe forms of colitis. 13 Our patient developed grade 2 colitis with 4–6 liquid stools per day, after the seventh cycle of Pembrolizumab. We made the diagnosis of CIC by ruling out infection, IBD, and tumor metastasis. Stool analysis for various infectious agents was negative. There were no significant deviations in the blood tests except for hypokalemia. The results of immunological and immunochemical analyses, as well as biohumoral markers of inflammation, were within the acceptable range. Endoscopy with biopsy is the gold standard for diagnosis, and microscopically, the acute colitis pattern of injury is the most common finding. 26 The macroscopic examination of our patient’s colonoscopy was normal. Pathistological analysis of polytopic biopsies of the colon confirmed that the condition was a mild to moderate case of microscopic interstitialis, also known as autoimmune colitis. For grade 2 diarrhea or colitis, systemic corticosteroids (prednisone or intravenous methylprednisolone of 1–2 mg/kg/day) should be initiated until the symptom improves to grade 1 or less. After that, the gradual tapering of steroids is recommended for at least 4–6 weeks.2,8,9 In the treatment of our patient, we are guided by ESMO recommendations. Our patient had grade 2 diarrhea with 4–6 liquid stools per day, so treatment involved the use of corticosteroid therapy such as prednisolone at a dose of 1 mg/kg. During the patient’s hospitalization, we administered parenteral corticosteroid therapy (amp. Dexamethasone N0II/12h), along with potassium supplementation, infusion solutions, probiotic therapy, Loperamide tbl as per the scheme, and proton pump blockers (PPI). A few days after applying the parenteral corticosteroid therapy, the diarrhea subsided. Due to the improvement of clinical symptoms, the patient was treated with gradual tapering of steroids per os for at least 4–6 weeks with PPI all the time corticosteroid therapy. The patient is currently free of complaints, has normal laboratory and clinical findings, and continues treatment with the 15th cycle of pembrolizumab. The latest multi-slice chest scan has shown immunostable disease (iSD). Some retrospective studies reported that patients who developed ICI-induced diarrhea or colitis had a superior OS than those without gastrointestinal toxicity, 22 as it happened in our case. Early diagnosis and appropriate treatment of ICI-induced colitis is crucial for an optimal result.
Conclusion
Endoscopic findings are the most important factor in predicting how ICI-induced colitis will respond to treatment. Macroscopic findings of the colon mucosa may be normal, but microscopically, it may be autoimmune colitis caused by immunotherapy. Good cooperation between oncologists, gastroenterologists and pathologists is necessary in order to provide timely and adequate treatment of ICI-induced colitis. It is important to note that patients who developed ICI-induced diarrhea/colitis had a superior OS than those without gastrointestinal toxicity. The most important and effective approach to managing adverse events of immunotherapy treatment is early recognition and intervention in order to prevent complications of immune related colitis and prevention of permanent interruption of treatment with Immune check point inhibitors. Vedolizumab and fecal microbiota transplantation are promising options for treatment-refractory ICI-induced colitis.
Footnotes
Author contributions
Marina Markovic: Conceptualization; supervision; Writing – original draft; Writing – review & editing. Danijela Niciforovic: Conceptualization; visualization; Writing – original draft; Writing – review & editing. Violeta Mladenovic: Conceptualization; visualization; Writing – original draft; Writing – review & editing. Dragica Pavlovic: Conceptualization; visualization; Writing – original draft; Writing – review & editing. Dragana Papic: Conceptualization; visualization; Writing – original draft; Writing – review & editing. Katarina Milojevic; visualization; Writing – original draft; help in providing case for MRI and MSCT images. Dalibor Jovanovic: visualization; Writing – original draft; provided case for microscopic images. Marija Spasojevic: visualization; Writing – original draft; provided case for microscopic images. Rade Milic: visualization; Writing – original draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethicals approval
Ethical approval was not sought for the present study because *REASON*. Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from all subjects before the study.
Consent to participate
The patient’s written consent was obtained for the participation in the study.
Consent for publication
The patient’s written consent was obtained for the writing and publication of the case, including the publication of images.


