Abstract
Immune checkpoint inhibitors have become essential antineoplastic agents in medical oncology over the past decade. However, they are associated with potentially fatal multisystem abnormalities, with increasing concern in gastrointestinal tract and its associated organs. We present a patient with advanced renal cell carcinoma, who presented with acute liver failure after the first dose of combined immunotherapy with nivolumab and ipilimumab. A thorough evaluation for viral, metabolic, and autoimmune causes was unremarkable. He was managed with steroids and made significant improvement. To our knowledge, this is the first documented case of acute liver failure following ipilimumab and nivolumab.
Introduction
Immune checkpoint inhibitors (ICIs) have now become the standard of care in management of oncological diseases. ICIs inhibit programmed cell death receptor (PD-1), its ligand, programmed cell death ligand-1 (PDL-1), or cytotoxic T-lymphocyte-associated protein-4 (CTLA-4), and relieve tumor-induced T-lymphocyte inactivation. Nivolumab is a monoclonal antibody against programmed cell death receptor-1 and ipilimumab is a monoclonal antibody against cytotoxic T-lymphocyte antigen 4. Through the above mechanism of action, unfortunately, they may go on to attack virtually innate cells in all organs, including the digestive system and thus lead to adverse events.1,2 These adverse effects are occasionally life threatening, leading to increased mortality and morbidity in patients with cancer.
Acute liver failure (ALF) is defined as liver injury with an international normalized ratio (INR) over 1.5 and hepatic encephalopathy in the absence of prior liver injury in the past 6 months. While liver injury due to ICI is relatively common, ALF from ICI is rare. Only 4 cases of ICI-induced ALF have been described in the literature.3-6
We report a case of an otherwise healthy man with metastatic renal cell carcinoma with normal liver function who developed ALF 2 weeks after initiation of combination immunotherapy, successfully treated with steroids.
Case Presentation
A 72-year-old male presented with fever, chills, nausea, vomiting, and confusion. He was initially diagnosed with left renal cell carcinoma and underwent left radical nephrectomy 7 months prior. He was monitored with Positron emission tomography and found to have metastatic renal cell cancer to lungs, bone, and adrenals. He received nivolumab and ipilimumab 2 weeks before presentation and liver function test before initiation of immunotherapy was unremarkable (Figure 1).

Trend of liver function since presentation.
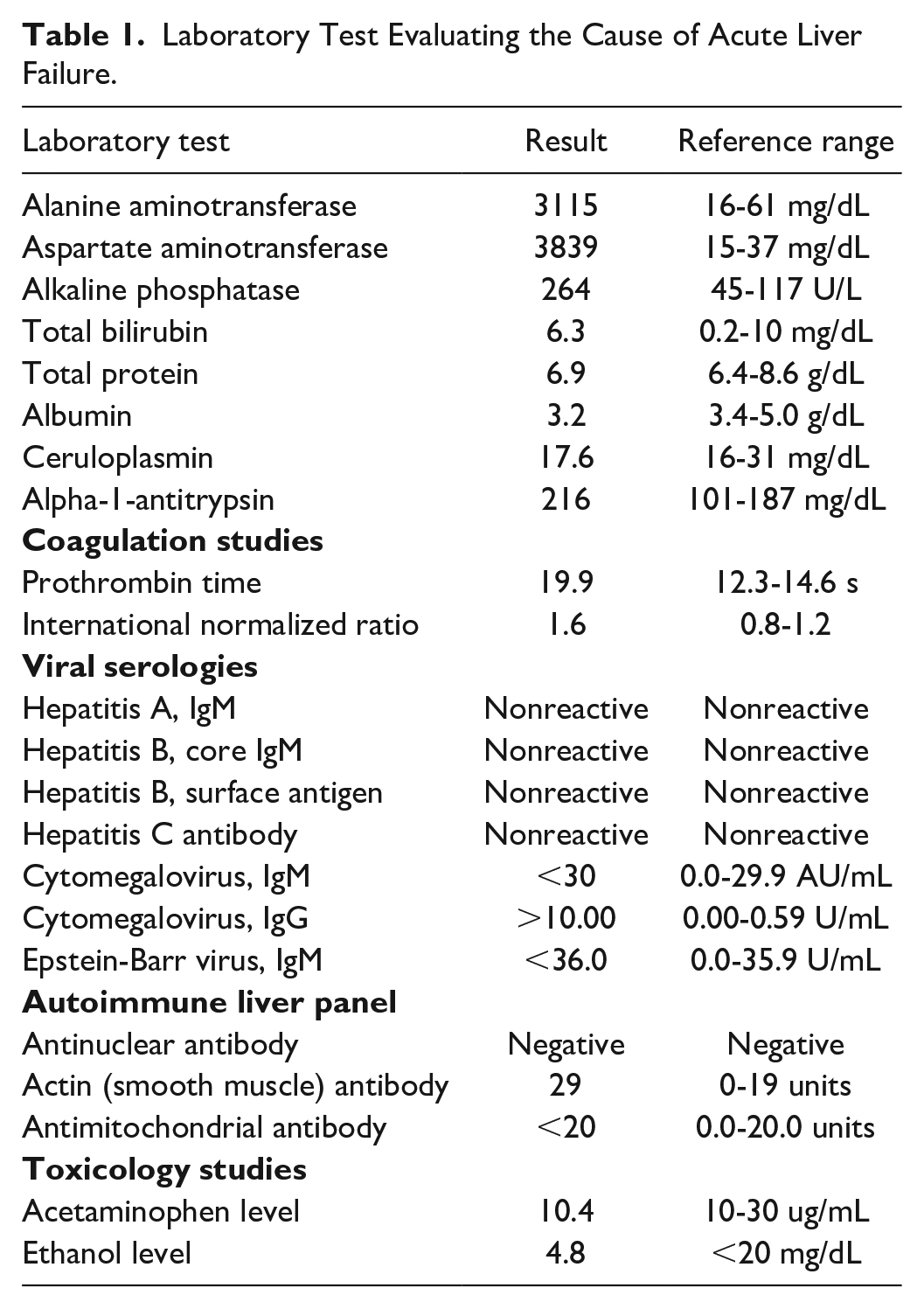
His temperature on presentation was 97.8 mmHg and other vital signs were unremarkable. On physical examination, he had conjunctival icterus and tenderness over the right upper quadrant without rebound rigidity, and abdomen was nondistended. Laboratory evaluation showed significantly deranged liver function (Table 1). Evaluation for other causes including viral panel, creatine kinase, acetaminophen and alcohol level, ceruloplasmin, alpha-1-antitrypsin, cytomegalovirus, Epstein-Barr virus, antinuclear, and antimitochondrial antibodies were negative. Actin (smooth muscle) antibody was weakly positive. Abdominal ultrasound and noncontrast computed tomography were unremarkable.
Laboratory Test Evaluating the Cause of Acute Liver Failure.
Following oncology and gastroenterology evaluation, he was admitted for ALF due ICI hepatic injury and received intravenous methylprednisolone 1 g twice daily and intravenous fluids. Liver function significantly improved, and he was discharged on hospital day 4 with a tapered dose of oral prednisone over 28 days. On follow-up in the outpatient clinic 2 weeks later, liver function continued to drop (improve).
Prednisone was reduced from 120 mg daily after 7 days to 60 mg daily. Five days later, patient had fever and rash at home. Laboratory evaluation at this time showed a sharp rise in Alanine aminotransferase (ALT)/aspartate aminotransferase (AST), so prednisone was increased to 80 mg daily and tapered by 20 mg every 7 days. Fever and rash resolved and liver function steadily improved toward baseline.
Discussion
We have presented a case of an elderly male with ALF following combination ICI therapy. To the best of our knowledge, this is the first case of ALF following the combination of ipilimumab and nivolumab.3-6 Prior cases have followed pembrolizumab, nivolumab, atezolumab, and combination of atezolizumab and tiragolumab. While ipilimumab has been associated with higher risk of hepatoxicity compared to other ICI, they are usually limited to mild hepatic injury. 2 Risk factors appears to mirror that of liver injury including the use of combination therapy, female sex, and advanced age. 7
Presentation of liver injury in ICIs is usually hepatocellular rather than cholestatic or mixed within few weeks of initiation of therapy which is consistent with our case. Auto antibodies are infrequently present as in our case with weakly positive antismooth muscle antibody, making it occasionally difficult to distinguish from autoimmune hepatitis. 4 In most cases, liver dysfunction first developed following the initial dose of immunotherapy and ALF presented after about 2 weeks. This suggests that evaluation at 1 to 2 weeks after initiation of therapy may help in early identification and treatment of individuals at risk before the development of ALF.
Diagnosis is commonly established with clinical history after ruling out close differentials including autoimmune hepatitis. Liver biopsy is not routinely required and only performed when the diagnosis is unclear. When performed, centrilobular necrosis with lobular inflammation and acidophil cells are commonly seen in histology.
Transaminase levels are elevated in ALF. Elevation in ALT and AST is generally defined as levels greater than 3× the upper limit of normal. However, levels in ALF are usually significantly higher as in our patient and even higher levels have been reported.
While acute liver injury is defined as transient in ALT/AST greater than 3 times the upper limit of normal, ALF combines the above with coagulopathy and change in mentation. While our patient meets diagnostic criteria for ALF, liver injury could present similarly especially with milder elevation in INR.
Treatment of ALF from ICI has evolved over the years. High dose intravenous steroid is recommended. As demonstrated by this case, we suggest gentle tapering (by about 20 mg) of steroid along with close monitoring of liver function to detect a plateauing or rise in LFTs. In patients who do not respond to steroids, immunosuppression with mycophenolate mofetil, azathioprine, cyclosporine, or tacrolimus is recommended. 8 As demonstrated by Renault et al, some patients may need plasma exchange for rapid symptomatic improvement. 3
In conclusion, ALF from ICI commonly presents with hepatocellular pattern of injury soon after initiation of therapy. Treatment is commonly with steroids and patients should be monitored closely. In this group of patients, ICI should be discontinued permanently.
Footnotes
Author Contributions
CE wrote the first draft of the manuscript. OU and AJ revised the manuscript. DL and RC reviewed the manuscript and made final corrections. RC is the article guarantors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Guarantor
Ryan Chauvin, MD
