Abstract
Guillain-Barré Syndrome (GBS) is an acute immune-mediated polyneuropathy causing rapidly progressive muscle weakness and often respiratory failure, requiring mechanical ventilation in 30% of cases. Successful weaning and extubation are crucial, focusing on readiness for spontaneous breathing trials, optimal timing, and minimizing complications such as ventilator-associated pneumonia and extubation failure. This case report describes an 83-year-old male with multiple comorbidities who was intubated after his negative inspiratory force dropped below −30 cm H₂O and treated with plasmapheresis, leading to partial improvement. Despite not meeting standard extubation criteria, he was successfully extubated, maintained stable respiratory function, and was safely discharged. This case highlights the challenges of extubation in GBS patients, where advanced age, decreased physiological reserve, and comorbidities increase the risks of prolonged intubation and complications. This case also highlights the need for individualized extubation strategies in GBS, particularly in elderly patients who may not meet standard criteria. Tailored approaches can still lead to successful outcomes. Based on our institutional experience, we propose factors that predict the success or failure of extubation in these patients. Further research is needed to refine predictive markers and improve extubation success in this population, ultimately enhancing outcomes and reducing ICU and hospital stays.
Keywords
Introduction
Guillain-Barré Syndrome (GBS) is an acute autoimmune polyneuropathy characterized by progressive muscle weakness, which can necessitate mechanical ventilation (MV) in up to 30% of patients due to severe respiratory failure.1-3
A study identifies key predictors of MV in patients with GBS. Bulbar dysfunction, rapid disease progression, autonomic instability, and respiratory muscle weakness—reflected by the 20/30/40 rule; vital capacity (VC) <20 mL/kg, maximum inspiratory pressure (PImax) <30 cm H₂O, and Maximum Expiratory Pressure (PEmax) <40 cm H₂O—are strong indicators of respiratory failure. 4
PImax and PEmax are measures of respiratory muscle strength, with PImax reflecting inspiratory muscle function (primarily the diaphragm) and PEmax indicating expiratory muscle strength (mainly the abdominal and intercostal muscles). Both are crucial for assessing respiratory failure risk in conditions such as GBS. 4 The Erasmus GBS Respiratory Insufficiency Score (EGRIS) predicts the risk of MV based on 3 key factors: onset-to-admission time, facial/bulbar weakness, and limb weakness severity. A low EGRIS score is associated with a 1% MV risk corresponding to a more prolonged onset of weakness after admission, often exceeding 7 days, no facial/bulbar weakness, and mild limb weakness. Conversely, an EGRIS score of 6 or higher is associated with a 90% MV risk reflecting a rapid disease progression (onset of weakness <3 days to admission), facial/bulbar weakness, and severe limb weakness. By quantifying these clinical indicators, EGRIS aids in the early identification of high-risk patients, allowing for timely ICU resource allocation and respiratory management. 5
However, deciding when to extubate these patients remains challenging in their management. The timing of extubation is critical, as early extubation has been associated with improved outcomes, while delayed extubation may lead to prolonged MV, increased risk of complications like pneumonia, and a more extended recovery period. 6 Extubation in patients with GBS is complicated by several factors, including the degree of respiratory muscle weakness, the patient’s age, comorbid conditions, and the presence of autonomic dysfunction or other critical illnesses. Although traditional markers such as VC and negative inspiratory force (NIF) are commonly used to determine readiness for extubation, applying these criteria in GBS patients is not well defined. 6
This case report focuses on a unique instance of successful early extubation in a patient with GBS. It highlights the complexity of clinical decision-making, where comprehensive respiratory and neuromuscular function assessments are critical in determining extubation readiness.
Case Presentation
An 83-year-old patient with a past medical history significant for aortic stenosis, coronary artery disease (CAD) status post-coronary artery bypass grafting, and chronic obstructive pulmonary disease (COPD) initially presented with bilateral lower extremity weakness, which resulted in a fall at home. He reported developing ascending and progressive weakness, leading to a complete inability to move his legs on the morning of his presentation. In addition, he noted the hoarseness of the voice. He denied any inciting events and was fully functional before his presentation. Initially, he had stable vital signs, and his physical examination revealed mildly decreased reflexes, which were more pronounced in the lower extremities.
The initial workup revealed a head CT scan with no acute abnormalities. A CT scan of the abdomen and pelvis demonstrated moderate interstitial and patchy alveolar infiltrates in the lung bases, more prominent on the right than the left, consistent with an infectious or inflammatory process. Laboratory results showed a creatinine level of 1.5 mg/dL, BUN of 22 mg/dL, lactic acid of 2.7 mmol/L, AST of 60 U/L, ALT of 16 U/L, and a urinalysis consistent with urinary tract infection (UTI). The patient was started on antibiotic therapy.
On the second day of his presentation, the patient started having worsening neurological symptoms, with loss of lower extremity motor function. He was also found to have absent patella, ankle, and plantar reflexes in his bilateral lower extremities. Due to the progressive ascending worsening of neurologic symptoms, the decision was made to transfer the patient to ICU. His NIF dropped below −30 cm H20. EGRIS score is estimated to be +6, predicting a high risk of respiratory failure in the 1st week of admission (65% in the derivation data). Therefore, it was decided to intubate the patient.
Further workup included a lumbar puncture and cerebrospinal fluid (CSF) analysis consistent with albuminocytologic dissociation, confirming the GBS diagnosis (Table 1).
Comprehensive Spinal Fluid Analysis Results.
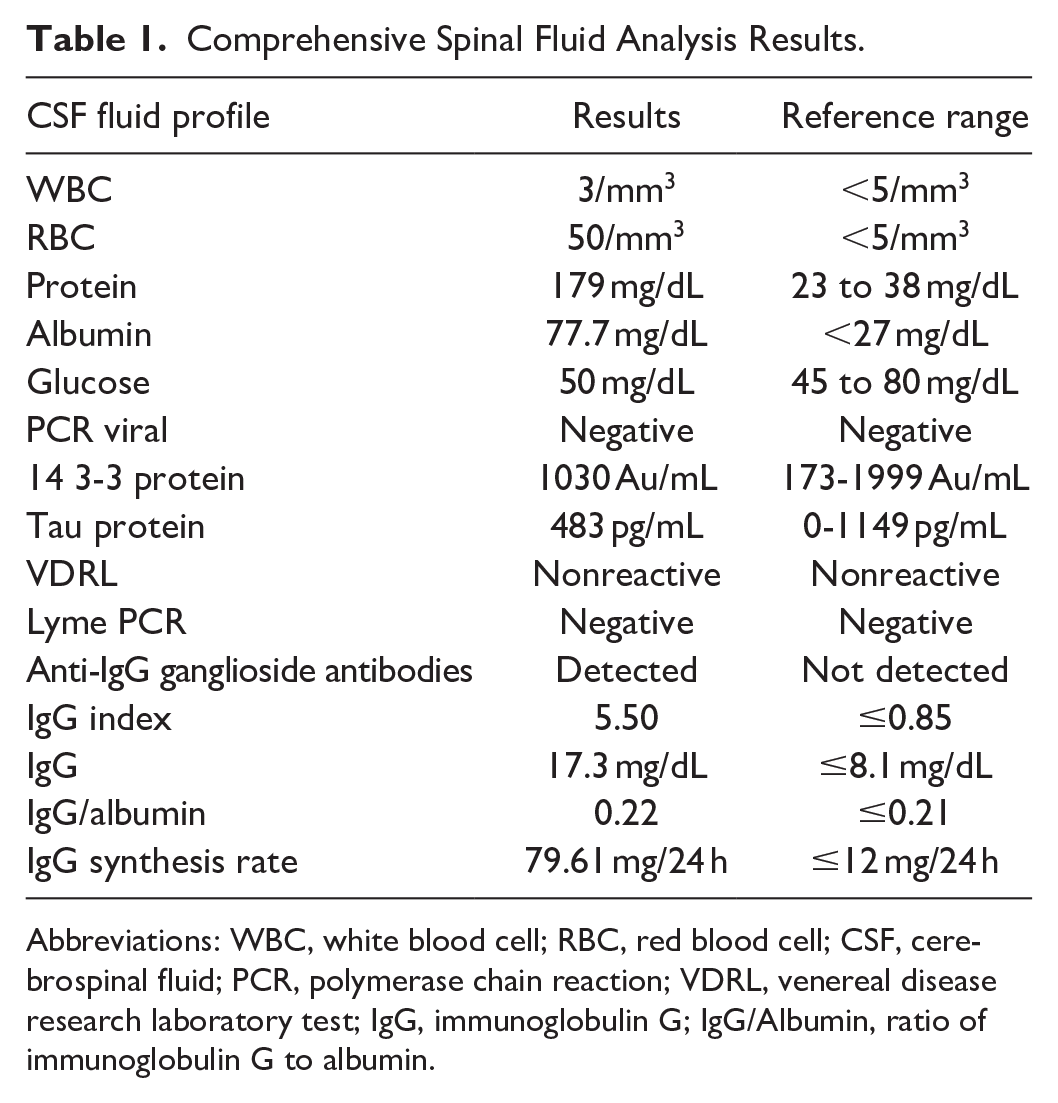
Abbreviations: WBC, white blood cell; RBC, red blood cell; CSF, cerebrospinal fluid; PCR, polymerase chain reaction; VDRL, venereal disease research laboratory test; IgG, immunoglobulin G; IgG/Albumin, ratio of immunoglobulin G to albumin.
Treatment was initiated with plasmapheresis, which the patient underwent 5 times. Intravenous immunoglobulin was considered; however, it was not initiated due to his favorable response to plasmapheresis, which included increased muscle strength, stabilization of autonomic parameters, and minimal ventilatory support requirements. During his ICU stay, he was closely monitored for autonomic instability, though no significant dysautonomia was observed.
Despite not meeting standard extubation criteria (VC > 15 mL/kg and NIF > −30 cm H₂O), the multidisciplinary team, including intensivists, neurologists, and respiratory therapists, agreed that the patient’s overall clinical course supported a trial of extubation. The decision was based on stable hemodynamics, absence of autonomic dysfunction, and an effective cough, all indicating adequate airway protection and respiratory effort. At the time of extubation, the patient’s VC was 3.6 mL/kg, and NIF was −14 cm H₂O, values below standard thresholds. However, extubation was performed given the patient’s clinical stability and ability to clear secretions. The patient was successfully extubated to 5 L O₂ via nasal cannula, and close post-extubation monitoring was planned to ensure prompt intervention in case of respiratory failure recurrence. Post-extubation arterial blood gas analysis showed a pH of 7.38, PaCO₂ of 42 mmHg, and PaO₂ of 88 mmHg. He was closely monitored in the ICU for signs of respiratory distress. Chest physiotherapy and incentive spirometry were initiated to prevent atelectasis and promote lung expansion. No desaturation or respiratory distress episodes were observed during the subsequent 24 hours. Physical rehabilitation commenced on the first day post-extubation to address the patient’s profound muscle weakness and prevent complications of immobility. A tailored rehabilitation program was developed, including passive range-of-motion exercises with gradual progression to active-assisted exercises as his strength improved. The patient tolerated the rehabilitation sessions well, demonstrating improved mobility in his upper extremities by the third day. By the time of transfer to the floor service, he could perform limited daily activities with minimal assistance. The rehabilitation team also provided education on fall prevention strategies, which were critical given his history of aortic stenosis and CAD. Table 2 summarizes the clinical course.
Timeline of the Patient’s Clinical Course.

Discussion
The decision to extubate patients with GBS remains challenging due to the profound respiratory muscle weakness that can impact the success of spontaneous breathing trials (SBT) and subsequent extubation. 7 This challenge is particularly pronounced in elderly patients with GBS, who have reduced physiologic reserve, lower muscle mass, and a higher incidence of MV dependence and pneumonia compared to younger individuals. 8 Careful assessment of respiratory muscle strength and overall clinical stability is crucial to optimizing extubation outcomes in this population. This is especially true when combined with any critical illness neuromuscular abnormalities. Thus, early interventions may assist with difficult extubation in associated muscle weakness. 7
Earlier extubation is associated with better outcomes in GBS, and patients should be assessed for extubation as early as possible. Prolonged intubation correlates with a prolonged recovery period, as do ICU complications, such as pneumonia. 9 Pneumonia is a significant complication for intubated GBS patients, with a reported incidence of about 50%.9,10 ICU complications are uncommon if the total stay is under 3 weeks; however, longer ICU stays increase the risk of systemic infections. 10 During SBT, minimal ventilator settings assess readiness for breathing without MV. Each day of failure increases mortality risk. 11 The need for reintubation is also associated with increased ICU length of stay, rates of nosocomial pneumonia, and mortality overall. 12
Some predictors of successful extubation have been studied in the general ICU population but not patients with GBS. However, based on our experience, they could still be applied to patients with GBS. One measurement that may be helpful is rapid shallow breathing, which is the ratio of respiratory frequency to tidal volume (f/VT). Yang et al. found this to be the most accurate predictor of MV weaning, with the absence of rapid shallow breathing proving to be the most precise predictor of successful MV weaning. 13 A randomized, multicenter Spanish trial found improved rates of extubation without the need for reintubation when patients were given a 1-hour rest with ventilator support after successful SBT before being extubated. 14 Figure 1 summarizes predictors of success or failure of extubation in patients with GBS.

Factors predicting success or failure of extubation in patients with Guillain-Barré syndrome.
Several factors have been associated with successful extubation in GBS. Nguyen et al. found that a lower (more negative) NIF and higher VC on the day of extubation correlated with extubation success and a change in the VC of >4 mL/kg pre-intubation compared to pre-extubation correlated with a 90% positive predictive value for extubation success. A higher VC at extubation also correlated with success with multivariate analysis. Extubation failure in GBS patients was associated with pulmonary comorbidities (79% vs. 36%, P = .008) and autonomic dysfunction (73% vs. 27%, P = .008). Additionally, patients who failed extubation or required tracheostomy had a more extended ICU stay (21.5 ± 11.1 vs. 12.5 ± 8.7 days, P = .005). 15 Although our patient had chronic medical conditions such as COPD and CAD, he did not demonstrate features of autonomic dysfunction or experience a prolonged stay in the ICU. He was extubated, with measured VC remaining below 15 mL/kg and an NIF of less than −30 mmHg. Following extubation, he was closely monitored for signs of increased work of breathing, mental status changes, the effectiveness of his cough, and hypercapnia through ABGs.
It is generally recommended that patients have VC > 10 mL/kg and NIF ≤ −20 to −30 in the assessment of readiness for SBT.16,17 SBT assesses the patient’s breathing ability while receiving minimal or no ventilator support. However, because of a lack of studies that have compared its duration directly, the recommendations on how long an SBT should last are understandably less incisive, suggesting that SBTs be conducted for either 30 minutes or 60 to 120 minutes depending on the patient’s risk for extubation failure. The patient can breathe through either a T-tube circuit of a ventilatory circuit with continuous positive airway pressure of 5 cm H2O or pressure support ventilation of less than 7 cm H2O. 18 Our patient had SBT, which lasted for 60 minutes before proceeding with extubation.
The Spontaneous Awakening Trial-Spontaneous Breathing Trial (SL-SBT) trial compared 30-minute versus 120-minute SBTs in patients with difficult weaning. The study found no significant difference in extubation success rates or secondary outcomes, suggesting that longer SBTs do not improve weaning outcomes. These findings support using shorter SBTs to minimize patient discomfort and resource use without compromising safety. 19 Extubation in a patient with GBS involves specific considerations that differ from the general population studied in the SL-SBT and similar trials. Patients with GBS often have neuromuscular weakness and autonomic dysfunction, which can complicate the weaning process. 6 For GBS patients, neurally adjusted ventilatory assist has shown promise in improving patient-ventilator synchronization and facilitating the weaning process. 20 Continuous monitoring of the diaphragm’s electrical activity (Edi) for pressure support titration can particularly benefit these patients.20,21
A study performed in surgical ICU patients found that an NIF more negative than −25 cm H2O had an 83% positive predictive value for weaning success. 22 The WIND study proposes additional criteria for SBT in the neurocritical care unit, especially in myasthenia gravis or GBS, including FVC >10 mL/kg or improved VC by >4 mL/kg compared with the previously recorded measurement. 11 It also suggests a maximum inspiratory pressure of −20 cm H2O as a threshold for extubation. 11 Additional monitoring methods such as end-tidal CO2 (ETCO2) to monitor for hypercapnia are suggested. 18 While ETCO2 is a standard noninvasive ventilation assessment tool, its role in GBS remains unclear and lacks specific validation in GBS patients.
Accepted standards state that patients who tolerate an SBT for 30 to 120 minutes be considered for discontinuation of MV. 23 Little data is available for a more extended breathing trial in patients who have failed previous attempts. Patients at high risk of extubation failure after being mechanically ventilated for over 24 hours are recommended to extubate to noninvasive ventilation. 18 Clinicians are often reasonably hesitant to extubate when NIF and VC are low, but there is minimal information on extubation criteria considering these 2 measures in the GBS population. A study of 38 GBS patients by Ropper et al. describes extubation attempts when VC reaches >8 to 10 mL/kg. With this approach, 12 patients (31.5%) in the study required tracheostomy. 24 Another study found that weaning trials in GBS patients with a VC < 7 mL/kg were unsuccessful and used VC ≥ 15 mL/kg as their extubation threshold. 25 This study observed that patients with VC < 7.0 mL/kg body weight (BW) could not tolerate even 15 minutes of spontaneous breathing with a T-piece, confirming that this threshold indicates poor weaning tolerance. The mean VC for all patients was 7.1 ± 1.2 mL/kg BW (median 6.5 mL/kg BW), reinforcing the critical role of VC in assessing readiness for weaning. In contrast, extubation was safely undertaken when minute ventilation (VE) exceeded 15 mL/kg BW, with a mean VE of 15.1 ± 0.9 mL/kg BW (median 15.0 mL/kg BW), a value significantly higher than VE at the beginning of the weaning period (P < .0001). These findings highlight the importance of both VC and VE as key predictors of successful extubation, suggesting that VC < 7.0 mL/kg BW predicts weaning failure, while VE > 15 mL/kg BW supports safe extubation decisions. 25 Nguyen et al. also found similar results; successfully extubated patients had a higher VC (21.9 ± 8.4 mL/kg) than those who failed (13.0 ± 5.9 mL/kg). 15
After meeting all the other widely accepted extubation criteria, our patient was successfully extubated on an MV on day 9. Prior to extubation, his NIF reached −14 cm H2O and his VC was 3.6 mL/kg (0.265 L, 73.2 kg). He continued to improve and was ultimately discharged with stable pulmonary function and no supplementary oxygen.
Conclusion
This case features the complexity of managing respiratory failure in GBS, particularly in elderly patients with comorbidities. It highlights the importance of individualized extubation strategies and the potential for successful outcomes even when standard criteria are unmet. This case also emphasizes the need for further research to refine standardized predictive markers of extubation failure and develop extubation criteria, prioritizing early extubation in this unique population to improve outcomes and reduce complications.
Footnotes
Acknowledgements
We would like to express our sincere appreciation to the Research Committee of the Internal Medicine Department at Marshall University, WV, for their invaluable support and guidance during the development of this manuscript. Their assistance has been instrumental in our work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Submissions/Publications
The abstract of this manuscript has been accepted for publication in the ATS meeting in May 2025.
