Abstract
Introduction
Guillain–Barré syndrome (GBS) is an inflammatory polyradiculoneuropathy characterized by the sudden onset of symmetric muscle weakness, unstable walking, and hypo- or areflexia. It is believed to be an autoimmune disorder that often follows a symptomatic bacterial or viral infection, such as Campylobacter jejuni, cytomegalovirus, Epstein–Barr virus, Mycoplasma pneumoniae, or influenza. 1 While GBS generally has a favorable prognosis, it can be a devastating condition with a mortality rate of approximately 10% and a significant disability rate of around 20%. 1 GBS has been observed as a rare adverse event associated with various viral vaccinations, including polio, influenza, hepatitis A and B, rabies, and the COVID-19 vaccine. 1 During the global COVID-19 immunization campaign, several uncommon vaccine-related adverse events with the potential for fatality have been reported. These include Vaccine-Induced Thrombotic Thrombocytopenia (VITT), thromboembolic events, and GBS. The necessity of vaccinating a large population against COVID-19 to prevent severe outcomes has brought about concerns regarding the potential impact on the incidence and prevalence of Guillain–Barré syndrome.
The COVID-19 vaccination has been associated with various neurological complications, including GBS, encephalitis, seizures, acute stroke, delirium, and meningoencephalitis. However, the precise nature of the association between COVID-19 vaccination and GBS, as well as the specific characteristics of GBS in this context, remains unclear. Recent data from the Vaccine Safety Datalink, analyzed by the Centers for Disease Control and Prevention (CDC), revealed that the incidence of GBS during the first 3 weeks following COVID-19 immunization with the Johnson & Johnson vaccine was 21 times higher compared to the mRNA vaccines from Pfizer, BioNTech, or Moderna.2,3
Methods
A comprehensive search was conducted on PubMed, Google Scholar, Cochrane, and Embase databases to identify relevant case reports published up until July 2022. The search terms used were “Guillain-Barré Syndrome” or “GBS,” “COVID-19 Vaccines” or “COVID-19,” “vaccination” or “vaccine,” and “Case report.” Initially, the screening process involved reviewing the titles and abstracts of the articles. Subsequently, case reports that focused on presenting symptoms, pathophysiological mechanisms, diagnosis, and treatment were considered.
Results
In our study, we analyzed 42 case reports published in peer-reviewed journals. These reports involved a total of 67 individuals who developed GBS following COVID-19 vaccination. Among the cases reviewed, the highest number of GBS cases, 36 individuals, were reported after receiving the AstraZeneca (Vaxzevria) vaccine. This was followed by 12 cases after Pfizer vaccination, 6 cases after Johnson & Johnson vaccination, 5 cases after Sputnik vaccination, 5 cases after Moderna vaccination, 2 cases after Sinopharm vaccination, and 1 case after Corbevax COVID-19 vaccination, as depicted in Figure 1. Number of people developing GBS after COVID-19 vaccination.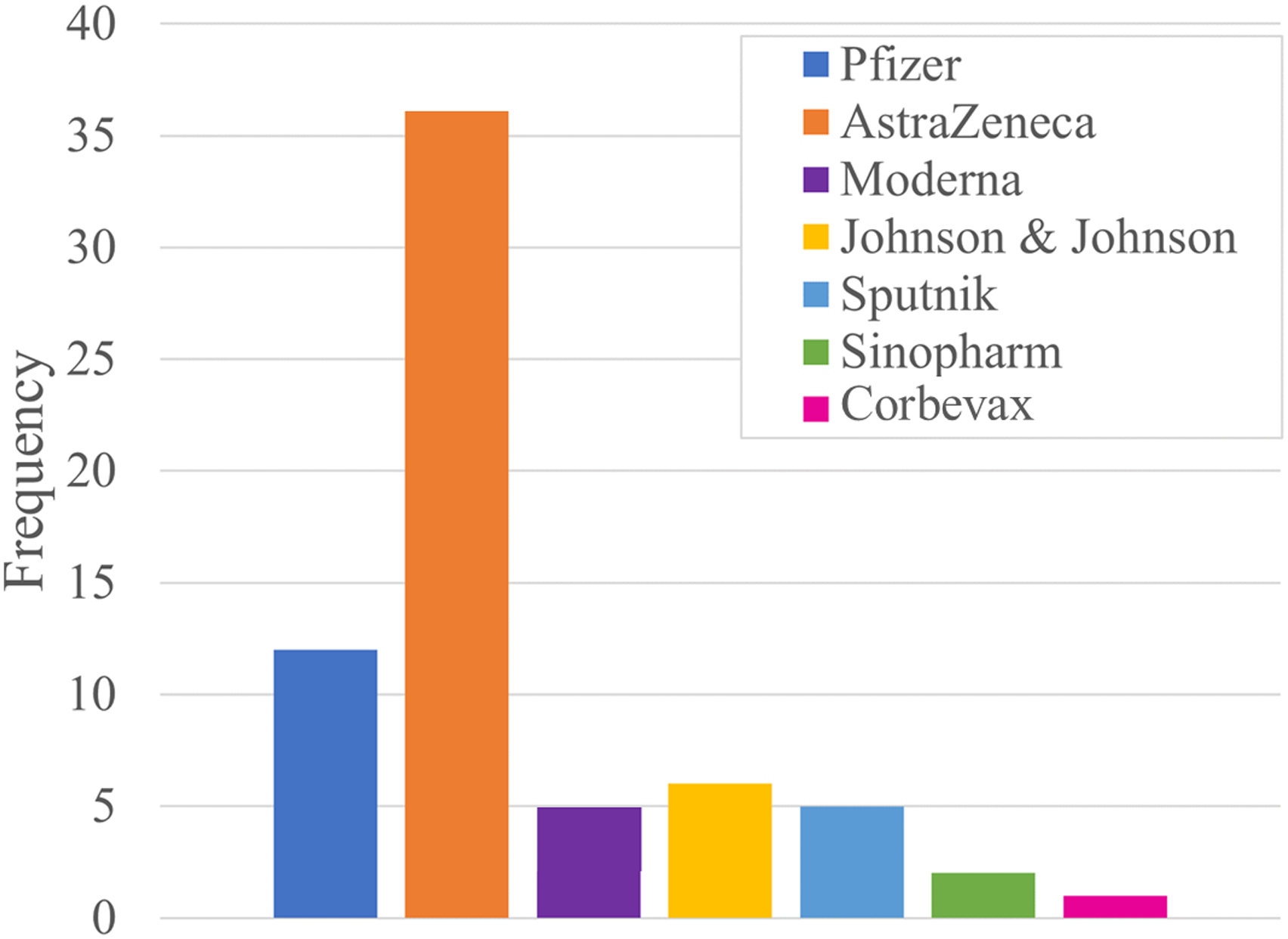
Full texts from the case reports were reviewed, and extraction of relevant data was then conducted and organized into tables [Supplemental tables 1–2].
The incidence of GBS development following COVID-19 vaccination was reported in case reports from 16 countries and regions. Among these countries, the highest number of cases (15) was reported in the USA, followed by 9 cases in Argentina, and 8 cases each in India and the UK. The remaining cases were reported from various other countries, as depicted in Figure 2. Incidence of GBS in different countries and regions following COVID-19 vaccination.
A wide range of age groups were observed among the individuals affected by GBS following COVID-19 vaccination. The highest number of individuals affected had an age range of 45–65 years. The average age at which GBS occurred after receiving the respective COVID-19 vaccine was determined to be 51.66 years, as illustrated in Figure 3. Furthermore, a notable male predominance of 61.2% (41 out of 67 individuals) was observed, while females accounted for 38.8% (26 out of 67 individuals). The average duration between receiving the COVID-19 vaccine and the onset of GBS symptoms was recorded as 12.67 days (Figure 4). Occurrence of GBS following COVID-19 vaccination in different age groups.
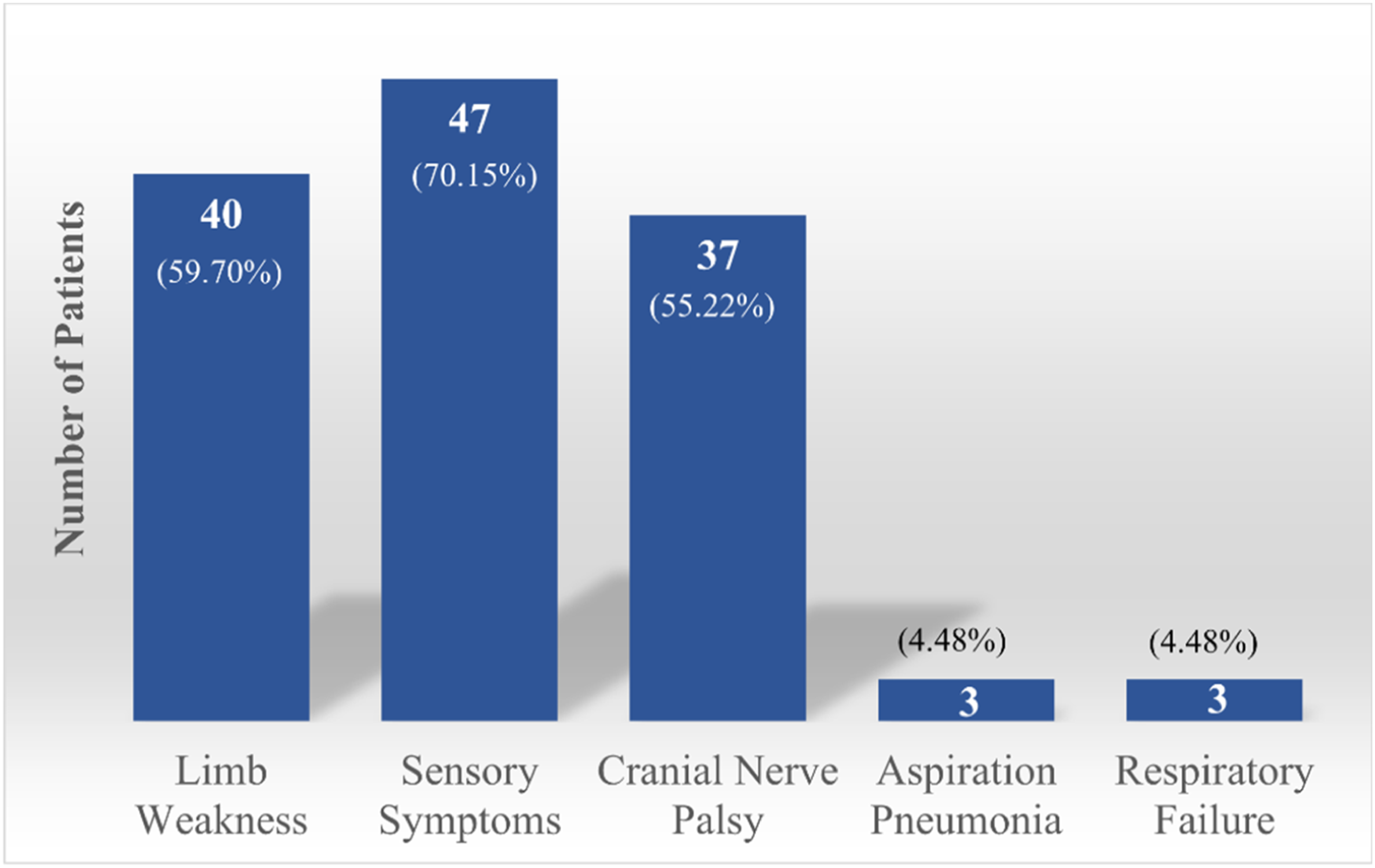
The initial presentations of GBS included paresthesia (47/67), limb weakness (40/67), cranial nerve palsy (37/67, mostly affecting facial nerve followed by oculomotor, trigeminal, and vagus nerves), myalgia, and headaches, and symptoms of dysautonomia such as constipation, nocturia, and urinary retention were also observed during hospitalizations (Figure 5). Interestingly, in eight cases, patients experienced sudden onset back pain before weakness in the lower limbs. The latency period between the administration of the vaccine and the appearance of GBS symptoms varied from 1 day to 60 days. Due to the lack of specific and sensitive disease biomarkers, the diagnosis of GBS relies on clinical history, examination, and supplementary investigations such as cerebrospinal fluid (CSF) analysis and electromyography studies. A total of 63 patients received lumbar puncture; 58 had manifestations of albuminocytological dissociation (characterized by an increased protein level without pleocytosis) in the CSF. Moreover, electromyography and nerve conduction studies revealed demyelinating polyneuropathy in most patients. Distribution of onset of symptoms days after vaccination. Most frequently observed features in patients.
The most frequent treatment used as the first-line therapy was intravenous immunoglobulin (IVIG) (n = 47, 70.1%). Plasmapheresis (PLEX) was performed in 14.9% of the cases (n = 10, four cases received PLEX solely and six received PLEX along with IVIG). After five to seven sessions of plasmapheresis, notable clinical improvement was reported. PLEX was associated with hypotension in a patient requiring treatment with midodrine. 4 Eleven patients have received corticosteroids (16.4%). Nine patients received mechanical ventilation (13.4%). Two patients required prolonged mechanical ventilation via tracheostomy.5,6 Nearly half of the patients experienced favorable outcomes (n = 30, 44.68%), about half of the patients (n = 34, 50.07%) had partial improvement requiring long-term rehabilitation, and two of the patients’ condition had improved initially but worsened later due to nosocomial infection. One patient died following sudden onset of arrhythmia and cardiac arrest before receiving any treatment. 7
Discussion
There is emerging evidence of the epidemiological link between Guillain–Barré syndrome and COVID-19 vaccination. GBS is an autoimmune disorder of the peripheral nervous system that typically manifests as a rapidly progressive ascending paralysis. 8 The study is of prime importance to promote early diagnosis and initiation of treatment because complications in GBS can cause severe morbidity and death. 9 This article focused on exploring the association between GBS and COVID-19 vaccination. The findings from the reviewed case reports provide important insights into the clinical features, diagnostic investigations, and treatment outcomes of GBS following COVID-19 vaccination. The reported cases demonstrate a clear temporal relationship between the administration of COVID-19 vaccines and the onset of GBS symptoms. Patients experienced ascending progressive paresthesia in the distal lower and upper limbs, along with muscle weakness and decreased or absent reflexes. This pattern of clinical presentation aligns with the typical manifestations of GBS, indicating a potential causal link between vaccination and GBS development.
Notably, our study revealed a predominance of GBS cases in the adult population, with an average age of 51.66 years. This is consistent with previous studies indicating that older age is a risk factor for developing severe COVID-19 and may also contribute to an increased incidence of GBS. The male predominance observed in our study further highlights the importance of considering demographic factors when assessing the risk of GBS following vaccination.
The time interval between vaccination and symptom onset averaged 12.67 days, suggesting a relatively short latency period for GBS development. This temporal association supports the hypothesis that COVID-19 vaccination may trigger an immune response leading to the development of GBS. However, it is worth noting that the underlying mechanisms linking vaccination and GBS remain unclear and require further investigation.
Treatment options for GBS following vaccination included intravenous immunoglobulin (IVIG), steroids, and plasmapheresis. IVIG was commonly used and showed effectiveness in halting the progression of neuropathy and improving muscle strength. With regard to outcomes, the favorable outcome was achieved in less than half the cases; in most cases, patients had partial recovery. Nine patients required mechanical ventilation. Further research is needed to determine the optimal treatment approach for GBS following COVID-19 vaccination and to assess the long-term outcomes of these interventions.
Rare occurrences of central nervous system autoimmune diseases have been reported in connection with the COVID-19 vaccine. 11 The potential association between autoimmune diseases and vaccination can be attributed to various mechanisms. These include the similarity of vaccine epitopes with myelin or axon epitopes, which may trigger immune responses at the cellular and humoral levels. Additionally, direct exposure of vaccine virus or vaccine-related components could potentially lead to the degradation of axon or myelin membranes. Genetic predisposition may also play a role in the development of autoimmune diseases following vaccination. 10
While the presence of a temporal association between vaccination and Guillain–Barré syndromedoes not provide sufficient evidence for a causal relationship, it does suggest a potential link. However, the wide range of time intervals observed in this study, ranging from less than a day to 60 days between vaccination and the onset of GBS symptoms, indicates a complex multifactorial relationship rather than a direct cause-and-effect connection between the two. The current COVID-19 vaccination programs, administered to billions of individuals worldwide, also warrant close monitoring and investigation to better understand the risk–benefit profile and to ensure the safety of vaccinated individuals.
One of the main limitations of this review article is the potential for selection bias due to the reliance on published case reports. This may result in an overrepresentation of severe or unique cases, potentially skewing the understanding of the overall incidence and prevalence of Guillain–Barré syndrome following COVID-19 vaccination. Reporting bias is also a concern, as positive findings are more likely to be published, leading to an overestimation of the association. The lack of a control group limits the ability to establish a causal relationship, while the heterogeneity of data from various sources makes generalization challenging. These limitations emphasize the need for more comprehensive studies with larger sample sizes and rigorous designs to accurately assess the relationship between COVID-19 vaccination and GBS.
Conclusion
While emerging evidence points to a potential temporal link between Guillain–Barré syndrome and COVID-19 vaccination, further comprehensive research is needed to establish a definitive causal relationship. The reported cases offer valuable insights into clinical patterns and treatment outcomes, emphasizing the importance of early diagnosis and intervention. Ongoing monitoring and robust studies are crucial to better understand the complex interplay between COVID-19 vaccination and GBS, ensuring the safety of global vaccination efforts.
Supplemental Material
Supplemental Material - Association of Guillain–Barré syndrome following COVID-19 vaccination
Supplemental Material for Association of Guillain–Barré syndrome following COVID-19 vaccination by Selia Chowdhury and Samia Chowdhury in International Journal of Immunopathology and Pharmacology
Supplemental Material
Supplemental Material - Association of Guillain–Barré syndrome following COVID-19 vaccination
Supplemental Material for Association of Guillain–Barré syndrome following COVID-19 vaccination by Selia Chowdhury and Samia Chowdhury in International Journal of Immunopathology and Pharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
