Abstract
Histiocytic sarcoma (HS) is a rare hematopoietic neoplasm with an aggressive clinical course and a poor response to conventional chemotherapy. Currently, no standard treatment paradigms are available. Herein, we present a case of de novo HS treated with pembrolizumab combined with a GDP regimen (gemcitabine, cisplatin, and dexamethasone) that resulted in sustained complete remission with progression-free survival exceeding 4 years. Immunohistochemical analysis demonstrated significant overexpression of programmed death ligand 1 (PD-L1) on biopsy samples. Additionally, fluorescence in situ hybridization (FISH) with a JAK-2 probe indicated 9p24.1 amplification, suggesting reliance on the JAK-STAT pathway. Polymerase chain reaction (PCR) analysis did not reveal any BRAF-V600 mutations. Consequently, an immune checkpoint inhibitor (ICI) was administered alongside chemotherapy, resulting in sustained complete remission and progression-free survival for over 4 years. Our findings suggest that a combination of ICI and chemotherapy could represent a promising therapeutic approach for HS.
Introduction
Histiocytic sarcoma (HS) is a rare malignant neoplasm with dismal clinical outcomes. The Surveillance, Epidemiology, and End Results survey reported a median overall survival of 6 months among a cohort of 158 patients with HS. 1
Currently, there is no consensus regarding the treatment of HS. Recent genetic analysis has identified recurrent mutations involving the RAS-MAPK signaling pathway (MAP2K1, KRAS, NRAS, BRAF, and PTPN11), among which BRAF V600E was most frequently detected. Some mutations activating the PI3K signaling pathway (PTEN, MTOR, PIK3R1, and PIK3CA) were also found in some patients with HS. 2 There are case reports that showed favorable responses by targeting the RAS/RAF/MEK pathway with BRAF and/or MEK inhibitors, such as trametinib, 3 vemurafenib, or cobimetinib. 2 However, the significant expression of PD-L1 in proliferative histiocytosis may represent the basis for using ICIs as a treatment.
Herein, we present a case of de novo HS treated with pembrolizumab combined with a GDP regimen (gemcitabine, cisplatin, and dexamethasone) that resulted in sustained complete remission.
Case
A 66-year-old female was referred to our institution presenting with chest pain, paroxysmal atrial fibrillation, persistent high fever reaching 41°C, and notable weight loss over the past 4 months. Chest computed tomography (CT) revealed a mass in the lower lobe of the right lung, extending into the hilar and mediastinal regions.
Under general anesthesia, the patient underwent endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA). Histopathological analysis revealed an infiltrate of large, pleomorphic, and atypical histiocytoid cells with abundant cytoplasm and associated necrosis, consistent with the diagnosis of HS. Immunohistochemical staining demonstrated that the tumor cells were positive for CD68, CD163, and lysozyme but negative for CD20, CD3, CD30, CD15, CD1a, Langerin, CD4, CD5, CD7, CD8, HMB45, ALK1, and SMA. In addition, in situ hybridization of EBV EBER yielded negative results. The Ki-67 proliferation index was 30% (Figure 1).

Immunostaining of biopsy specimens positive for CD68, CD163, LCA, lysozyme, and PD-L1 (VENTANA SP263, Roche).
Given the absence of a universally accepted treatment protocol for HS, we implemented a poly-chemotherapy induction regimen comprising a Cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) cycle followed by a Cyclophosphamide, doxorubicin, vincristine, prednisone (CHOPE) cycle. Post-induction chemotherapy, her fever subsided but recurred within 2-3 weeks. Positron emission tomography/computed tomography (PET/CT) indicated partial remission of the lesion (Figure 2B). Considering the suboptimal response, we proceeded with consolidation using high-dose chemotherapy involving two cycles of the IMOPE regimen (ifosfamide, mitoxantrone, vincristine, etoposide, and prednisone). The patient developed severe myelosuppression, presenting as grade IV hematological toxicity and febrile neutropenia, necessitating an extended hospital stay. Nevertheless, interim PET-CT revealed no improvement in the residual lesion and identified new hypermetabolic activity in the T4 thoracic spine, indicative of disease progression (Figure 2C).
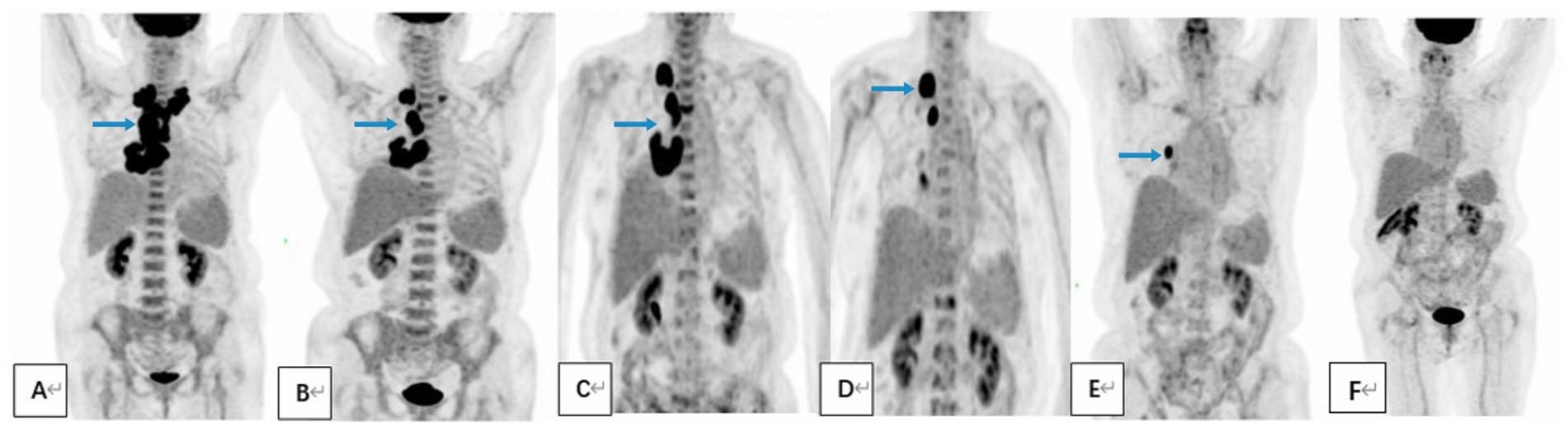
A comprehensive 18F-FDG-PET scan of the entire body reveals a substantial decrease in lesion size following treatment with immune checkpoint inhibitors (A) Lesions observed at initial diagnosis; (B) Partial lesion remission post-induction protocol; (C) Disease progression after multiple conventional chemotherapy regimens; (D and E) Efficacy observed with ICI in combination with GDP chemotherapy; (F) Sustained complete remission exceeding 4 years at the latest follow-up.
Considering the ineffectiveness of the previous chemotherapy and the patient’s poor tolerance to intensive regimens, targeted therapy was evaluated. However, PCR analysis did not reveal any oncogenic BRAF V600E mutations, leading us to conclude that MEK or BRAF inhibitors are likely to be ineffective.
Several studies and articles in the literature have documented high levels of PD-L1 expression in histiocytic neoplasms. Immunostaining revealed that more than 90% of tumor cells in the initial biopsy specimen exhibited significant PD-L1 overexpression (refer to the list of papers in Figure 1). Additionally, FISH analysis identified copy number amplification of JAK-2 in these tumor cells (Supplementary Figure 1). The patient was treated with pembrolizumab in combination with a GDP regimen (gemcitabine, cisplatin, and dexamethasone) every 3 weeks. After four chemotherapy cycles, the patient’s overall condition improved, B symptoms resolved, and PET-CT scans indicated an effective response (Figure 2D and E). However, treatment was complicated by severe thrombocytopenia and extensive purpura. Considering both economic and therapeutic safety, the patient was treated with sintilimab (a PD-1 inhibitor) at a dosage of 3 mg/kg every 3 weeks, in conjunction with thalidomide (100 mg/day), without encountering significant adverse events. The patient survived and remained symptom-free. PET-CT scans at the latest follow-up confirmed ongoing complete remission for over 4 years (Figure 2F).
Discussion
Histiocytic sarcoma is an extremely rare malignant neoplasm of mature histiocytes with an aggressive clinical course and poor response to chemotherapy. Currently, there is no widely accepted standard treatment approach due to the low frequency of the disease and the lack of clinical studies.
Owing to its rarity and poor prognosis, we reviewed the literature on the successful treatment of HS. Several cases documented favorable responses to MEK and/or BRAF inhibitors, including trametinib, vemurafenib, cobimetinib, and tyrosine kinase inhibitors such as imatinib. 4 For conventional chemotherapy, a combination of cladribine and high-dose cytosine arabinoside or CLAG-M has been reported as a promising treatment approach. 5 Successful treatment of HS with alemtuzumab, dose-escalated CHOP plus etoposide, and pediatric abbreviated high-risk leukemia chemotherapy was also documented. 6
The present case did not harbor BRAF V600E oncogenic mutations with disease progression after multiple lines of chemotherapy and did not tolerate intensive chemotherapy because of severe myelosuppression toxicity. Accordingly, establishing optimal management is challenging. In their study, Gatalica et al 7 first found a significant expression of PD-L1 in proliferative histiocytosis. A few case reports have demonstrated the promising efficacy of ICIs.8,9 Therefore, we attempted to combine PD-1 inhibitors with chemotherapy. Given the patient’s lack of prior exposure to platinum, relatively low risk of cross-resistance, and minimal myelosuppressive toxicity, we administered a combination of pembrolizumab and a GDP regimen for salvage therapy, resulting in complete remission. Since it has been reported that thalidomide could result in durable long-term survival, 10 we selected a PD-1 inhibitor combined with thalidomide for maintenance.
Fluorescence in situ hybridization revealed JAK-2 copy number amplification in primary tumor cells. This indicates that the relationship between 9p24.1 copy number amplification and PD-L1 overexpression in HS parallels that observed in nodular sclerosing classic Hodgkin lymphoma (cHL) and primary mediastinal large B-cell lymphoma. 11 We speculated that the amplification of 9p24.1 and the related JAK-STAT pathway similarly enhanced PD-L1 expression in HS.
In conclusion, we propose that integrating ICI with chemotherapy could be a promising therapeutic strategy for HS. Nevertheless, further case studies are necessary to validate the efficacy of ICI-based treatment modalities for this condition.
Supplemental Material
sj-tiff-1-hic-10.1177_23247096241274561 – Supplemental material for Pembrolizumab Combined With GDP Regimen Inducing Sustained Remission in Histiocytic Sarcoma: A Case Report
Supplemental material, sj-tiff-1-hic-10.1177_23247096241274561 for Pembrolizumab Combined With GDP Regimen Inducing Sustained Remission in Histiocytic Sarcoma: A Case Report by Jinjie Gao, Min Li, Cuiling Liu and Hongmei Jing in Journal of Investigative Medicine High Impact Case Reports
Footnotes
Acknowledgements
We express our gratitude to the patient and her son for providing written informed consent for the use of the off-label anticancer medications and for their approval to publish this case report.
Author Contributions
All authors took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; we all have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the special fund of the National Clinical Key Specialty Construction Program, P. R. China (2023).
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
