Abstract
To investigate the effects of total flavonoids from
Introduction
Diabetic nephropathy (DN) is one of the most severe chronic microvascular complications of diabetes. It is a major cause of death in diabetes patients and the major cause of end-stage renal disease. The mechanisms underlying DN development are complex. Reported changes involved in DN development include epigenetic changes,1,2 glucose metabolism disorders,3,4 changes in renal hemodynamics,5,6 insulin resistance,7,8 and abnormalities in inflammatory responses and cytokines.9,10 In recent years, the phenomenon of inflammation has received increasing interest in the field, with suggestions that DN is an immune-mediated inflammatory disease. Studies have revealed that the inflammatory cytokines angiotensin (Ang) II, monocyte chemoattractant protein (MCP)-1, intracellular adhesion molecule (ICAM)-1, nuclear factor-(NF)-κB, vascular cell adhesion molecule (VCAM)-1, interleukin (IL)-6, IL-8, and transforming growth factor (TGF)-β1 are closely related to DN.11–13 The majority of these inflammatory cytokines are primarily secreted by immune cells such as monocytes/macrophages and lymphocytes. 14 These cytokines induce fibroblast activation and the secretion of extracellular matrix (ECM) components, such as collagen, fibronectin, and laminin, which are important factors implicated in DN development and progression.
Furthermore, these inflammatory factors interact with multiple signaling pathways in the kidneys to form a complex network that promotes the development of DN. Among the various signaling pathways, the Janus kinase (JAK)/signal transducer and activator of transcription (STAT) pathway plays a dual role in signal transduction and activation of gene transcription. It exerts biological effects by mediating intracellular signal transduction processes of multiple cytokines and activating the corresponding target genes. JAK/STAT is the key signaling pathway involved in the development of DN. In vivo studies have confirmed that JAK/STAT signaling activation results in AngII-mediated mesangial cell proliferation and stimulation of TGF-β production, leading to a further increase in ECM secretion, and aggravates the pathology of DN.15–17 Therefore, inhibiting the JAK/STAT pathway can suppress intracellular signal transduction processes of inflammatory cytokines, thereby attenuating DN development. The suppressors of cytokine signaling (SOCS) family of proteins are negative regulators of the JAK/STAT pathway. They are involved in the regulation of the wound-healing process in various diseases that may occur in various diseases such as rheumatoid arthritis and other immune diseases, cerebral hypoxia, heart failure, and tumors.18,19 Recently, the SOCS family was found to relieve the damaging effect of DN by suppressing JAK/STAT signaling. 19
Numerous studies have shown that plant flavonoids demonstrate significant efficacy in improving diabetes-related complications.20,21 Their effects include the lowering of blood sugar and blood lipids, improvement of renal function, and inhibition of glomerular hypertrophy and mesangial matrix expansion. Some of these effects have been observed clinically.22–24 Recent studies have shown that total flavonoids from
Materials and methods
Experimental preparation
Experimental animals
Fifty 8-week-old male KK-Ay mice and ten 8-week-old male C57BL/6j mice were purchased from Beijing HFK Bioscience (Beijing, China), and all of these are specific-pathogen-free (SPF) animals.
Reagents
Hematoxylin and eosin (H&E) stain kit (C0105; Beyotime), horseradish peroxidase (HRP)-labeled goat anti-rabbit secondary antibody (A0208; Beyotime), radioimmunoprecipitation assay (RIPA) lysis buffer (P0013C; Beyotime), TRIzol (9109; Takara), PrimeScript RT Reagent Kit with gDNA Eraser (RR047A; Takara), real-time PCR (qPCR) kit (RR820A; Takara), rabbit anti-SOCS-1 polyclonal antibody (GTX100443; Genetex), rabbit anti-SOCS-3 polyclonal antibody (GTX100034; Genetex), rabbit anti-phosphorylated (p)-JAK2 polyclonal antibody (GTX41286; Genetex), rabbit anti-p-STAT1 polyclonal antibody (GTX37694; Genetex), anti-JAK2 (ab108596; Abcam), anti-STAT1 (ab31369; Abcam), SOCS-2 (Abcam; ab3692), total-STAT3 (Abcam; ab119352), p-STAT3 (Abcam; ab76315), TGF-β1(Abcam; ab92486), fibronectin(Abcam; ab2413); laminin(Abcam; ab11575), collagen type IV(Abcam; ab6586).
Experimental methods
Model establishment
Eight-week-old KK-Ay mice were used as the model control group, and C57BL/6J mice were used as the non-treat control group. The mice in the model and control groups were fed a high-fat and a normal diet, respectively. After 4 weeks of the diet, blood glucose was measured; the DN model was considered to be successfully established if the blood glucose stability was ⩾16.7 mmol/L compared with the control group.
Grouping and dosing
Total flavonoids from
Sample collection
At the end of treatment week 4, the mice were fasted but allowed free access to water. After, 24 h urine samples were collected and stored at −20°C for further testing. Blood was collected from the tail vein; the serum was separated and stored at −20°C. After blood collection, the mice were euthanized. The kidneys were separated and dissected longitudinally into several sections, which were stored in 4% paraformaldehyde or liquid nitrogen based on the detection method used in subsequent analyses.
Assessment of general conditions and body weight
The general condition of the mice, amount of water and food intake, body weight, and random blood glucose levels were monitored. The body weights were recorded before and 0, 2, and 4 weeks after model establishment.
Detection of blood glucose and urinary albumin (UAlb)
Twenty-four-hour urine was collected using metal metabolic cages. The urine volume was recorded. Blood glucose was measured in blood sampled from the tail vein. UAlb and blood glucose levels were measured before and at 0, 2, and 4 weeks after the model was established.
Serum biochemical parameters
Changes in serum creatinine (Cre), blood urea nitrogen (BUN), and uric acid (UA) levels were measured after 4 weeks of treatment using an automatic biochemical analyzer.
Glomerular tissue morphology and evaluation
H&E staining was used to observe pathological changes in the renal tissues. Briefly, the renal tissue samples were fixed in 4% paraformaldehyde for 1 week, dehydrated using an alcohol gradient, embedded in wax, and cut into 4 μm slices after drying. The tissue slices were mounted and hydrated by reversing the aforementioned dehydration steps. The sections were then stained with hematoxylin at room temperature for 5 min, followed by washing and color separation with a hydrochloric acid–alcohol solution. The tissue sections were washed, stained with eosin for 30 s, and dehydrated according to the aforementioned procedure until transparent. Finally, the sections were sealed for observation. The histological was evaluated according to Lu et al. 28 The degree of damage in each glomerulus was assessed using a semi-quantitative scoring method as follows: grade 0, normal glomeruli (no damage); grade 1, mesangial expansion area, up to 25% (minimal damage); grade 2, 26%–50% expansion (moderate damage); grade 3, 51%–75% expansion (moderate-to-severe damage); and grade 4, 76%–100% expansion (severe damage). The glomerular matrix expansion index (GMI) was then calculated by the following formula: GMI = (1 × n1 + 2 × n2 + 3 × n3 + 4 × n4)/(n0 + n1 + n2 + n3 + n4).
Reverse transcription-qPCR
RNA was extracted from kidney tissues stored in liquid nitrogen using TRIzol the manufacturer’s instructions. After removal of genomic DNA and RNA quantification and quality check, the RNA was reverse transcribed into cDNA. The cDNA was used as a template for qPCRs. The thermal cycles were as follows: pre-denaturation at 95°C for 10 min, 40 cycles of 95°C for 15 s, 60°C for 15 s, and 72°C for 30 s. Relative target gene expression was calculated as 2–ΔΔCt. Primer sequences are shown in Table 1.
Primer sequences.

MCP-1: monocyte chemoattractant protein-1; NF-κB: nuclear factor; IL-6: interleukin-6; TGF-β1: transforming growth factor; JAK: Janus kinase; STAT: signal transducer and activator of transcription; SOCS: suppressor of cytokine signaling.
Western blotting
Fresh renal tissue was collected, washed with phosphate-buffered saline (PBS), and cold RIPA buffer was added to the tissue on ice. After lysis, the sample was transferred to a new centrifuge tube and the lysis reaction was continued for 30 min. Then, the tube was centrifuged at 13,000 r/min, the supernatant was collected, and the protein concentration was determined using bicinchoninic acid kit. Proteins were resolved using 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The samples were subjected to the following procedures: electrophoresis, membrane transfer, blocking, overnight incubation with primary antibody, washing with PBS, secondary antibody incubation at room temperature for 1.5 h, washing with PBS, visualization using enhanced chemiluminescence, X-ray film development, and image capturing.
Cell culture experiment in vitro
Glomerular mesangial cells were cultured in RPMI-1640 medium, penicillin and streptomycin were added to make their final concentration of 100 U/mL. The cells were purchased from China Center for Type Culture Collection (CCTCC) and stored in our laboratory. According to the experimental requirements, prepare a normal culture medium containing 5 mmol/L glucose, and 30 mmol/L high-glucose medium for use. MC was cultured at 5 mmol/L and 30 mmol/L, respectively. The high-glucose group was divided into five groups, model, pioglitazone (10µg/mL), low dose flavonoids of
Assessment of ECM accumulation
The experimental method was based on Pugliese et al. 29 and was further improved. Media were collected and 10% carbonate–bicarbonate buffer, pH 9.6, protease inhibitors was added to prevent protein degradation, whereas cells were extracted with 0.5 N NaOH containing 2 mM phenylmethanesulfonyl fluoride (PMSF) and sonicated. The NaOH extracts referred to as cells actually included cells, intracellular spaces, and pericellular regions. Media and cell extracts were stored at −20°C until assayed C-IV, FN, and laminin.
Statistical methods
The Statistical Package for the Social Sciences (SPSS) 21.0 statistical software package was used for statistical analyses. The results are presented as the mean ± standard deviation. One-way analysis of variance was used to compare means among multiple groups, while a least significant difference
Results
Effect of total flavonoids from O. falcata on general conditions and changes in body weight in DN mice
Compared with the non-treat control group, after establishment of the diabetes model, the KK-Ay mice showed increases in food intake, water intake, urine output, and body weight. In control C57BL/6J mice, body weight increased steadily and the magnitude of the increase remained constant over the 4-week experimental period. Mice treated with total flavonoids from

Effects of total flavonoids from
Effects of total flavonoids from O. falcata on serum Cre, BUN, and UA in DN mice
Serum Cre, BUN, and UA levels in C57BL/6J control mice remained at normal levels over the 4 week experiment. Compared with the control group, the serum levels of Cre, BUN, and UA in the model mice were significantly (
Effects of total flavonoids from O. falcata on serum Cre, BUN, and UA.
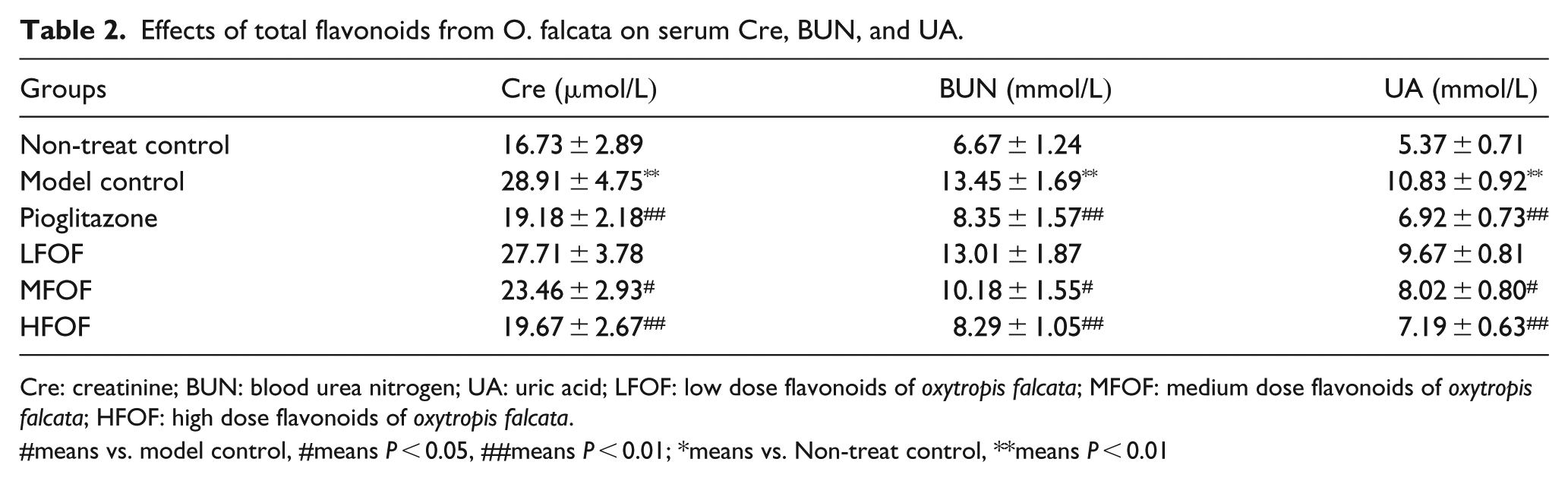
Cre: creatinine; BUN: blood urea nitrogen; UA: uric acid; LFOF: low dose flavonoids of
#means vs. model control, #means
Effects of total flavonoids from O. falcata on glomerular histomorphology in DN mice
Compared with that in C57BL/6J control mice, the number of glomeruli in the kidney tissues of the model mice significantly decreased 4 weeks after model establishment, whereas pyknosis, glomerular cell number, and ECM significantly increased. Compared with that in the non-treated model mice, the number of glomeruli in the kidneys significantly increased in the pioglitazone-treated positive control group and the group treated with the high dose of total flavonoids from

Effects of total flavonoids from
Effects of total flavonoids from O. falcata on mRNA expression in the kidneys of DN mice and Glomerular mesangial cells
Compared with that in C57BL/6J control mice, renal mRNA levels of
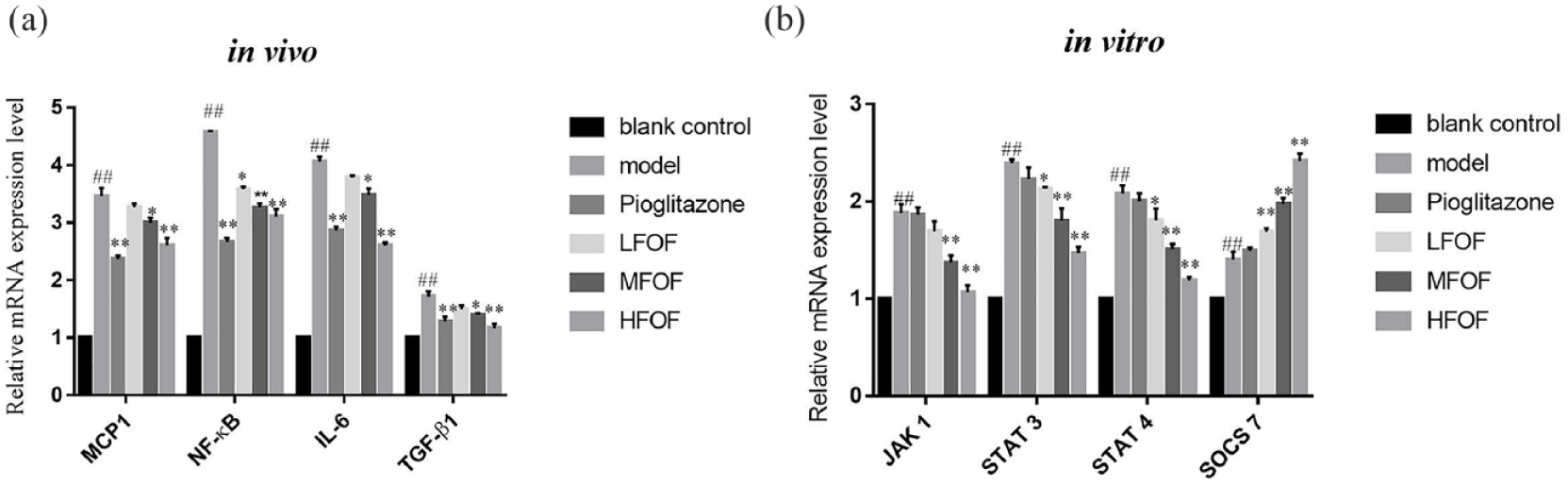
Effects of total flavonoids from
Effects of total flavonoids from O. falcata on the protein levels in the kidneys of DN mice and Glomerular mesangial cells
Compared with C57BL/6J control mice, p-JAK 2 and p-STAT 1 levels in the kidneys of the model group significantly increased 4 weeks after model establishment, whereas the SOCS-1 and SOCS-3 protein levels were significantly reduced. Compared with the model control mice, these responses were significantly suppressed in model mice treated with medium and high doses of total flavonoids from
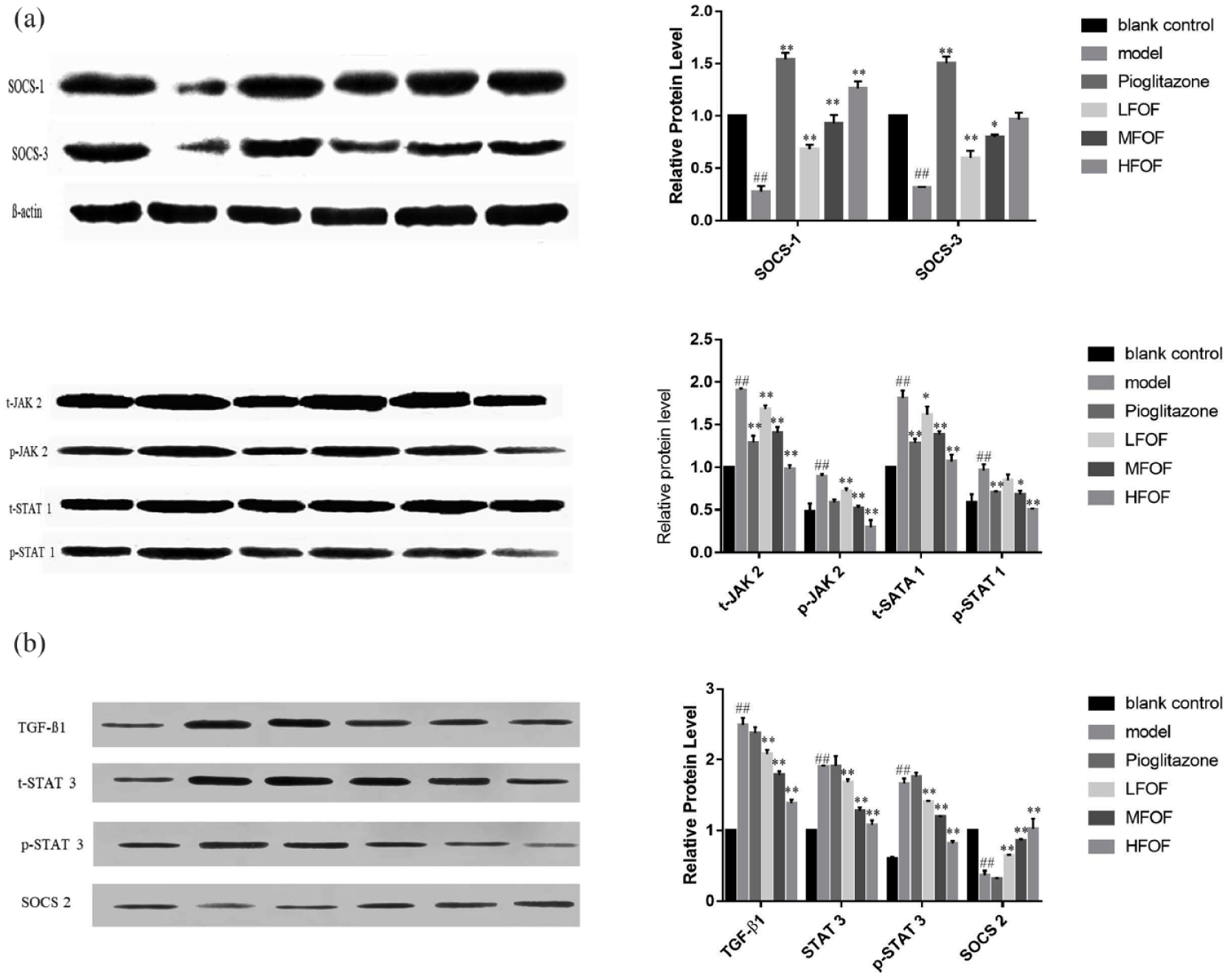
Effects of total flavonoids from
The effect of total flavonoids from O. falcata on ECM accumulation in vitro
Compared with control group, model group of fibronectin, laminin, and collagen type IV expression on protein and RNA levels was significantly increased; positive control was no significant difference with model group. With the increase of total flavonoids dosage, the degree of renal mesangial cell fibrosis gradually decreased. Total flavonoids can reduce the accumulation of ECM, and it is speculated that total flavonoids act directly on renal cells to protect the kidney (Figure 5).

The effect of total flavonoid on ECM accumulation. (a) The western blot of Fibronectin, laminin and Collagen type. (b) The RNA expression level of Fibronectin, laminin and Collagen type.
Discussion
This study investigated the protective effect of total flavonoids from
Most patients with type 2 diabetes die from complications as their disease progresses, and DN, which is a key cause of death in these patients, is the major cause of end-stage renal disease. DN is a chronic microvascular disorder of the kidneys induced by high glucose. Pathological changes induced by DN include an increased glomerular filtration rate accompanied by mesangial proliferation, ECM thickening, glomerulosclerosis, and interstitial fibrosis of renal tubules. As DN progresses, the glomerular filtration rate continues to gradually decrease, accompanied by diffuse thickening of the glomerular basement membrane and matrix proliferation, and the formation of typical K-W nodules. This is followed by renal tubular atrophy and renal dysfunction and, eventually, end-stage renal disease.31,32
In the present study, 24 h UAlb levels in the KK-Ay type 2 DN model mice increased over time under high-fat diet. Blood levels of Cre, BUN, and UA, which are important indicators of renal functions,33,34 significantly increased at the end of week 4 after DN model establishment. Increases in UAlb, Cre, BUN, and UA indicate that the molecular sieve that serves as a barrier for glomerular filtration is disrupted. The negative charge in the glomerular filtration membrane is reduced, and the glomerular charge barrier and mechanical barrier are impaired. 35 In the present study, as the duration of high-fat and high-glucose diet feeding increased, the renal function of mice with type 2 diabetes gradually deteriorated, eventually leading to DN. Pathological changes in the renal tissues, including glomerular atrophy and ECM thickening, confirmed the development of DN.
Treatment of mice with type 2 diabetes with total flavonoids from
Hyperglycemia is the underlying cause of DN, and direct control of the aberrant increase in blood glucose can effectively limit DN development. This may explain how total flavonoids from
In conclusion, total flavonoids from
Footnotes
Acknowledgements
Thanks to National natural science foundation (81560764), Gansu Longyuan Youth Innovation and Entrepreneurship Talent Project, Tibetan Medicine Regional Collaborative Innovation Center Project (2017XTCX013), and thanks to everyone who contributed to this article project.
Author’s Note
Lili Wu, Cuiyan Lv and Tonghua Liu are also affiliated with Tibetan University of Tibetan Traditional Medicine, Lhasa.
Author contributions
LY and JX are first co-authors; they contributed equally to the article. LW and CL are the corresponding authors; all authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
This study was approved by the animal experiment ethics of Gansu University of Traditional Chinese Medicine. Experimental animals were killed and their bodies were disposed of: the cervical vertebra was dislocated after anesthesia with 3% pentobarbital sodium intraperitoneal injection. The carcasses were delivered to the animal center of Gansu University of Traditional Chinese Medicine for innocuous treatment at the hazardous waste disposal center of Gansu province (no. 2016-165).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National natural science foundation (81560764), Gansu Longyuan youth innovation and entrepreneurship talent project, Tibetan medicine regional collaborative innovation center project (2017XTCX013).
