Abstract
We report a case of a 26-year-old type 1 diabetic woman presenting with acute lower abdominal pain, bloating, and vomiting. Initial examination revealed right lower quadrant tenderness and a suprapubic mass. Computed tomography (CT) imaging identified a 12-cm cystic structure suggestive of a dilated bowel loop with an adjacent inflamed 7-cm small bowel segment. Surgical exploration uncovered a Merkel’s diverticulum–associated duplication cyst originating from the Meckel’s diverticulum. Subsequent complications included an anastomotic leak, requiring relook laparotomy and the formation of a double-barrel stoma. The patient recovered and was discharged on day 13. This case highlights the diagnostic challenge of Meckel’s diverticulum-associated duplication cysts, emphasizing the need for vigilance in managing complex abdominal presentations.
Introduction
Meckel’s diverticulum (MD), a congenital gastrointestinal (GI) anomaly resulting from the persistence of the vitelline duct, is a well-recognized entity. 1 However, the coexistence of MD and enteric duplication cysts, particularly Merkel’s diverticulum–associated duplication cysts, represents a rare and challenging diagnostic scenario. This article reviews the literature on Merkel’s diverticulum–associated duplication cysts, shedding light on their clinical nuances and emphasizing importance of surgeons to be aware of this condition.
Case Presentation
We present an extraordinary case of a 26-year-old type 1 diabetic female who presented to the emergency department with acute lower abdominal pain and associated symptoms of abdominal bloating and vomiting. Upon review, she was tachycardic, normotensive, and afebrile. Physical examination revealed a soft abdomen with tenderness in the right lower quadrant, accompanied by a mass-like fullness at the suprapubic region. The inflammatory markers were unremarkable and was admitted to the surgical ward overnight for observation.
Following consultant ward round review, the following morning she underwent computed tomography (CT) imaging of the abdomen and pelvis. Radiological evaluation identified a 12-cm cystic structure in the mid-pelvis, indicative of a grossly dilated and distended loop of bowel with mesenteric swirl with suspicion of an internal hernia (Figures 1 and 2). The radiologist also made comment on an adjacent 7-cm segment of small bowel with features of inflammation. Given there had been no improvement in the patient’s symptoms and concern for threatened bowel, she proceeded to surgery. This involved a diagnostic laparoscopy with findings of a large cystic mass intimate with the terminal ileum. Complete inspection of the small bowel was unable to be performed through laparoscopy owing to the large mass. No inflamed appendix was found and pelvic organs appeared normal on inspection. Decision was made to convert to lower midline laparotomy to facilitate inspection of the small bowel mass. The 12-cm mass was fluid-filled and had a thick fibrous connecting cord to the antimesenteric aspect of terminal ileal segment associated with MD (Figure 3). This was found 30-cm proximal to the ileocaecal valve. The small bowel proximal and distal to the affected segment appeared normal. Thus, an ileocolic resection with end-to-end anastomosis was done.

CT abdomen and pelvis (axial view) of 12-cm cystic mass in the mid-pelvis.

CT abdomen and pelvis (coronal view) of 12-cm cystic mass in the mid-pelvis.
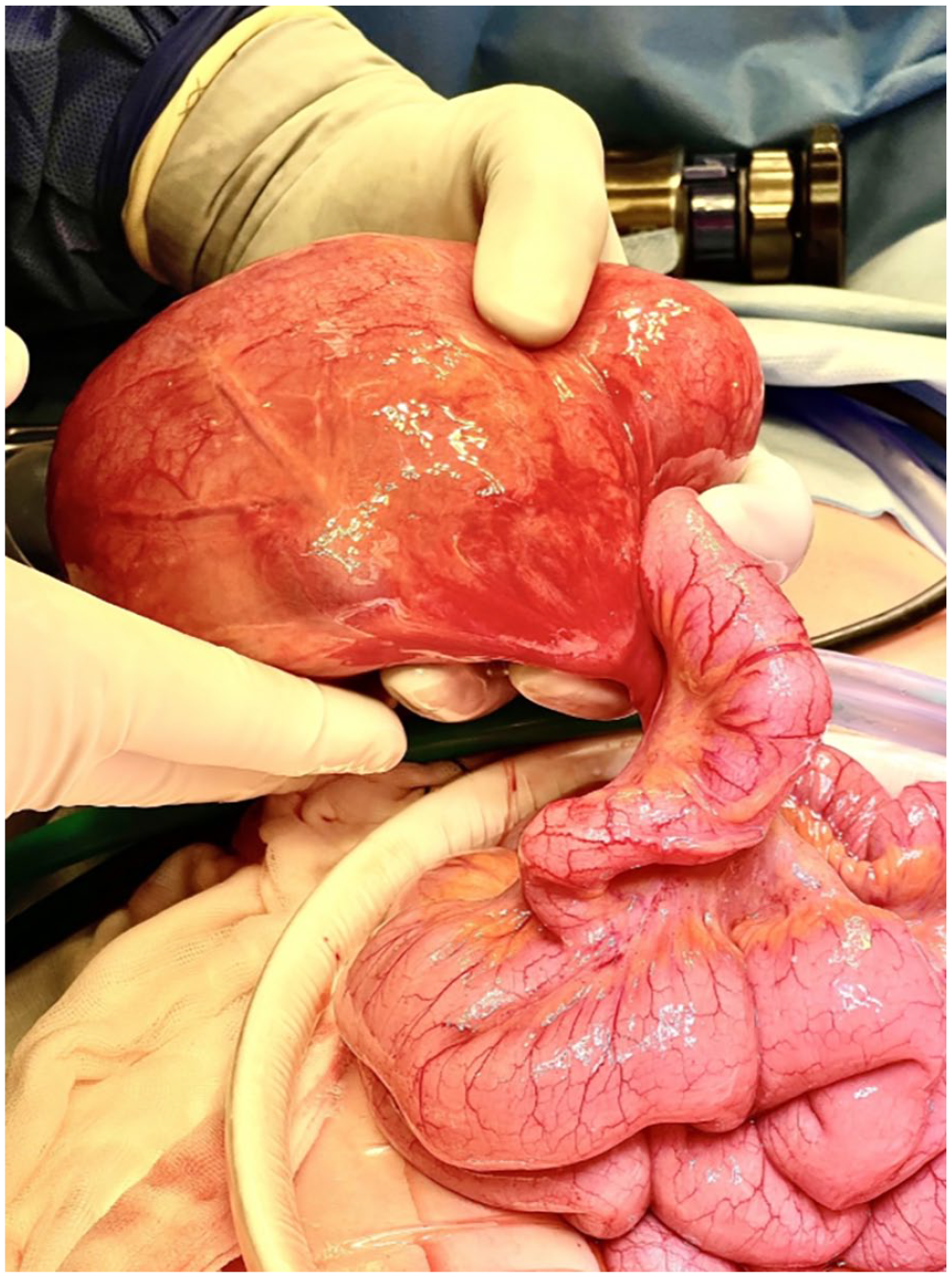
12-cm fluid-filled mass.
Histopathological examination revealed a Merkel’s diverticulum measuring 70 × 20 mm with a narrow luminal connection to a 12-cm cystically dilated bowel segment displaying active chronic inflammation and ischemic changes. The attached cystic structure exhibited colonic histology with confirmed mucosa, submucosa, muscularis propria and serosal layers.
Microscopy demonstrated a predominant crypt-type architecture, without overt villous features.
Postoperatively the patient had a marginal recovery in the first 72 hours. She was managed with an insulin infusion on the surgical ward and oral intake was titrated. Her progress included passing flatus on day 2 postoperatively. The patient remained tachycardic on the ward and underwent CT-Pulmonary Angiogram (CTPA) to exclude a pulmonary embolism.
Concurrent imaging of the abdomen confirmed an ileus. Her clinical condition improved over the subsequent 72 hours with bowels open on day 4. Unfortunately, on day 7 her condition deteriorated as she was tachycardic and spiked temperature in the ward. Further cross-sectional imaging demonstrated a likely anastomotic leak at the ileocolic anastomosis. This was confirmed upon return to theater for a relook laparotomy and necessitated formation of Abcarian (double-barrel) stoma. The patient improved and was suitable for discharge on day 13 of admission and was subsequently followed-up by the Colorectal Surgical team as an outpatient.
Discussion
Meckel’s diverticulum is the most frequently encountered congenital anomaly of the GI tract and is the result of the incomplete obliteration of the vitelline duct during intestinal development. This results in the formation of a true diverticulum within the small intestine, typically located within 100 cm of the ileocecal valve. 1 MD has prevalence of 2% in the general population, with a mean size of 3 cm. 2 Enteric duplication cysts, in contrast, can occur anywhere along the GI tract. These anomalies are characterized by a smooth muscle coat, an epithelial lining resembling intestinal mucosa, and their proximity to the GI tract, frequently on the mesenteric aspect. 3 These cysts may share a common wall with the main alimentary tract lumen and often share a common blood supply to the adjacent intestine. However, it is exceedingly rare for a duplication cyst to originate directly from an MD. Diagnosing MD-associated enteric duplication cysts can be challenging, particularly when they arise directly in communication with an MD, as seen in our case.
Conclusion
The rarity of this presentation, combined with difficulties in reaching a diagnosis on conventional CT imaging, provides a particularly challenging clinical scenario of the acute abdomen. Surgical resection remains the mainstay of treatment, particularly when symptomatic. Clinicians must remain vigilant to the spectrum of intestinal development anomalies that can manifest in unexplained abdominal symptoms.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Central Adelaide Local Health Network Human Research Ethics Committee (CALHN HREC).
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
