Abstract
Merkel cell cancer (MCC) is a rare cutaneous malignancy arising from neuroendocrine cells. This rare but lethal malignancy (mortality greater than 30%) has also tripled in incidence over the last two decades. The role of immunodeficiency in pathogenesis of this rare malignancy is well established, with elucidation of viral pathogenesis by Merkel cell polyoma virus (MCPyV), a novel polyoma virus, in a majority of patients. Viral neoantigens while playing an important role in oncogenesis can also aid in early immunologic detection of recurrent disease. Viral neoantigens can also be targets for immunotherapy. Immune checkpoint inhibitors (ICI) have established role in frontline therapy in addition to recurrence of metastatic MCC. ICI therapy is being explored in adjuvant and neoadjuvant settings as well. We would like to illustrate curative potential of early diagnosis of recurrence and prompt use of ICI in treatment of oligometastatic disease in our case presentation.
Introduction
Merkel cell cancer (MCC) presents usually as a rapidly progressing erythematous or violaceous nodule on exposed skin in elderly Caucasians. Prevalence is increased 2-fold in males compared with females. MCC rarely occurs in un-exposed areas such as oral and genital mucosa where it is associated with poor prognosis. UV radiation and immunosuppression are well-established risk factors. Immunosuppression from lymphoproliferative disorders, HIV, or immunosuppression employed for solid organ transplantation is not only associated with 5- to 50-fold increase in incidence; it is also associated with worse prognosis. Ten percent of patients are found to have metastatic disease at presentation. One third of patients with loco-regional disease will eventually develop distant disease. Survival of patients with distant disease is poor; median survival is approximately 1 year.1,2
Surgery is the mainstay of local therapy; a full skin examination and staging scans are essential prerequisites. Positron emission tomography (PET)/computed tomography (CT) has excellent specificity and sensitivity in staging of MCC. Surgery comprises of wide local excision followed by skin grafting if necessary. Resection margin of 1 to 2 centimeters should be obtained if possible. Adjuvant radiation therapy is employed after wide local excision in local (stages 1 and 2) and regional disease with or without lymphadenectomy (stage 3). A large retrospective study involving 1254 patients demonstrated improved local Hazard Ratio (HR: 0.27) and regional disease (HR: 0.34) control with adjuvant radiotherapy.3-6
Palliative chemotherapy with platinum and etoposide was the mainstay of therapy for metastatic (stage 4) MCC. While associated with excellent response rate 55% to 60%, duration of remission is brief with a median progression free survival of 3 months. Second-line chemotherapy regimens are ineffective (response rate of around 10%), and median progression free survival is only 1.9 months.
Immune checkpoint inhibitor therapy, initially offered for relapsed disease is now recommended for frontline therapy of MCC. This is not only associated with higher rate of response but also durable response.7-14
Case Presentation
A 73-year-old presented with rapidly enlarging erythematous lesion in the left forearm; after initial therapy with antibiotics and attempted incision and drainage, the lesion was biopsied, revealing the diagnosis of MCC. CT scans of chest, abdomen, and pelvis did not reveal evidence of spread to the axillary nodes or metastatic disease. Dermatological evaluation did not reveal any evidence of cutaneous metastasis. Wide local excision and adjuvant radiation therapy was employed as curative therapy. MCPyV oncoprotein antibody was positive. Serial titers were obtained (Figure 1) to monitor the disease status; on account of increasing titers, PET/CT was ordered. PET/CT revealed a soft tissue nodule in the deltopectoral groove. Excision biopsy revealed metastatic MCC. Excision biopsy was followed by adjuvant radiation. Immunotherapy with avelumab (Bavencio) (a fully human antibody directed against programmed death ligand-1) was used as consolidation for oligometastatic disease. Serial MCPyV antibody titers obtained showed decreasing titers which ultimately normalized (Figure 1). CT scans and physical evaluation have not revealed any evidence of disease. Complications from Avelumab include hypothyroidism and ill-defined dyspnea. CT scan did not demonstrate pneumonitis. He completed a year of avelumab therapy and continues in unmaintained remission more than 3 years.
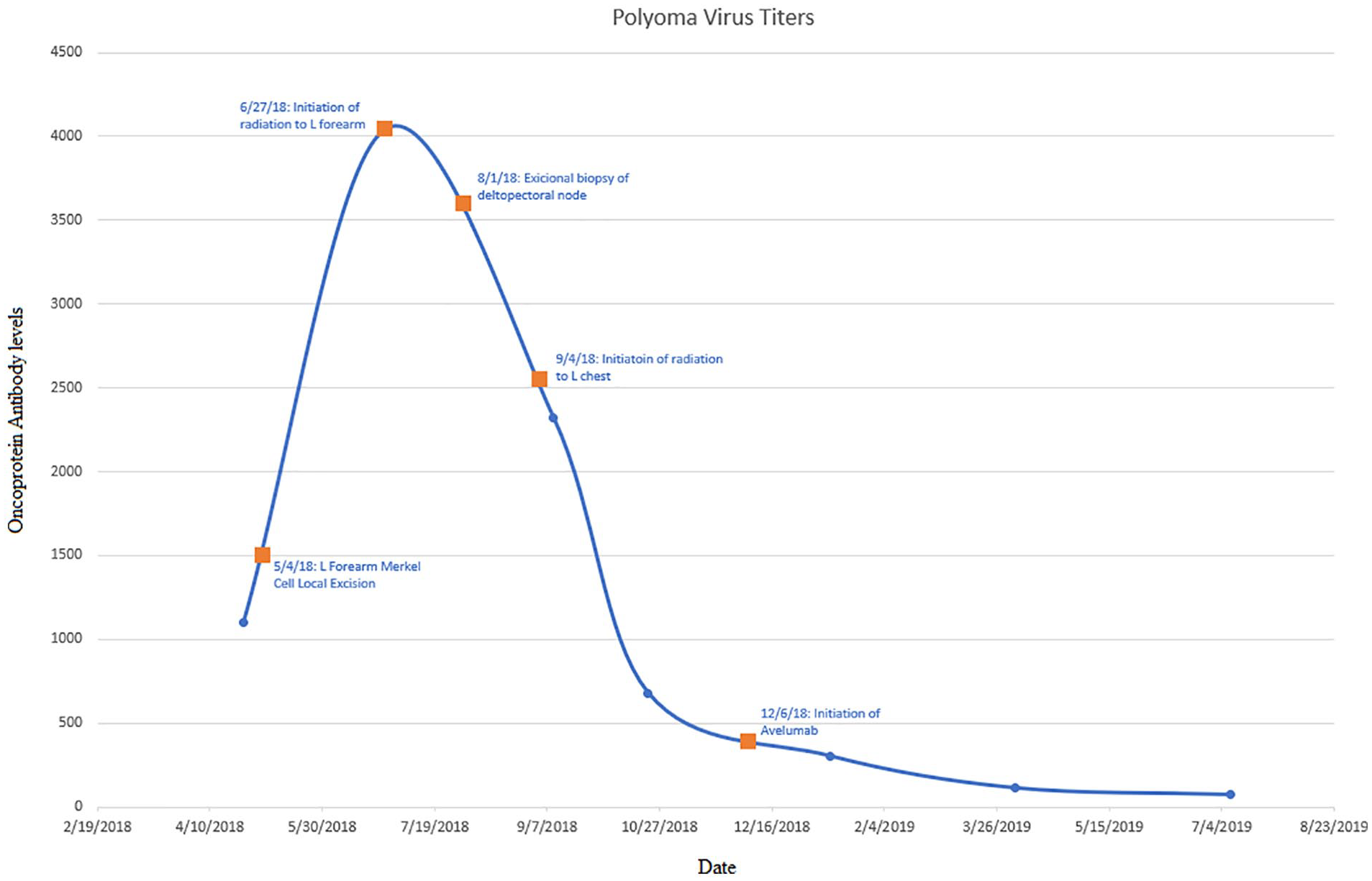
Polyoma virus oncoprotein titers throughout various phases of treatment.
Discussion
MCPyV is a small DNA virus with a small circular double stranded DNA genome of around 5 kilo bases. The viral genome consists of 3 regions: non-coding regulatory region, early and late coding regions. The early coding region contains large T and small T antigens, and late coding region codes for capsid proteins VP1 and VP2. MCPyV genome is normally maintained as a circular dsDNA episome. MCPyV is an abundant virus commonly found on healthy skin; infection is widespread in general population. Primary infection occurs during childhood; once acquired it becomes a life-long component of skin flora. Oncogenesis in MCC includes clonal integration into the cellular genome which results in tumor-specific truncal mutation of large T antigen. This results in loss of viral replication while preserving persistent expression of large and small T antigens important in oncogenesis.
Patients with MCC exhibit antibody production to MCPyV capsid proteins and oncoproteins.
Antibodies to viral capsid proteins are detected in 90% of patients with MCC. High titers are associated with improved prognosis; however, antibodies to viral capsid proteins are detected in 60% of healthy adults. The antibody titer does not reflect the tumor burden and hence not useful in surveillance for recurrent disease. Antibodies to viral oncoproteins are more specific; while this is present in about half of the patients with MCC, this is rarely detected in healthy individuals. Titers of antibody vary with tumor burden and hence useful in detecting recurrent disease. After initial therapy, rapid decrease in titers is observed and becomes negative around 8 months. Increasing antibody levels was predictive of recurrence (positive predictive value of 66%) and decreasing antibody titers had a negative predictive value of 97%. While MCPyV oncoprotein antibody is increased in only half of patients with MCC, this identifies a favorable subgroup in whom antibody titers can be employed for surveillance and avoid frequent imaging.3-6,15,16
Immune checkpoint inhibitor therapy has revolutionized therapy of advanced MCC. Avelumab was initially evaluated in advanced MCC patients progressing after 1 or more lines of chemotherapy. While response was noted in a third of patients, duration of response (median 40.5 months) was significantly better than that observed after first-line chemotherapy. Progression free survival at 24 and 36 months were 26% and 21%, and median overall survival of 12.6 months. Higher response rates have been reported after front-line therapy with pembrolizumab and avelumab. Responses obtained were durable after front-line therapy and associated with manageable safety profile.7-14
Conclusion
MCC is a rare cutaneous malignancy arising from neuroendocrine cells. Significant advances in pathology, pathogenesis, serologic monitoring, and therapy have improved outcomes in this rare cancer. Immunotherapy has significantly impacted the outcome of disseminated MCC and utility as adjuvant and neoadjuvant therapy is being explored. Early detection of recurrence by monitoring viral oncoprotein should improve the outcome like the observations reported in this case study.
Footnotes
Acknowledgements
We thank White River Health System for providing the resources and support to make this project possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
