Abstract
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumors in the digestive tract and arise from the interstitial cells of Cajal in the mesenteric plexus. These tumors can originate in any part of the GI tract; however, a higher burden has been observed in the stomach and small intestines. Mesenteric GISTs are exceedingly rare, with unique clinicopathological features and a poorer prognosis. Herein, we describe a unique case of a 66-year-old female with a remote history of appendectomy who presented to the emergency room complaining of severe abdominal pain and vomiting. On imaging, the patient was found to have a large inflammatory mass associated with small bowel loops, and the pathology confirmed a mesenteric GIST. The tumor was resected, and the genomic test results confirmed the KIT (exon 11) mutation. Although the tumor had a low mitotic rate, the tumor was large enough to warrant the initiation of adjuvant imatinib mesylate for 36 months with regular bloodwork and imaging.
Keywords
Introduction
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumors of the GI tract and arise from the intestinal cells of Cajal or their stem cells.1-4 Gastrointestinal stromal tumors account for 1% of all GI tumors and have an estimated incidence of 10 to 20 cases per million per year.2,3 Necrotic studies have suggested an even higher prevalence, particularly of smaller GISTs.2,5 The term GIST was coined by Mazur and Clark in 1983, but it took 15 years for its etiopathogenesis to be understood.1-3,6-10 In 1998, Japanese scientists found that gain-of-function mutations in KIT proto-oncogene receptor tyrosine kinase were critical for developing GISTs.1,11 This critical discovery paved the way for more molecular studies and the expanded use of imatinib mesylate (IM; Gleevec, Novartis, Basel, Switzerland) in the treatment of advanced GISTs and later as adjuvant chemotherapy. 11
Gastrointestinal stromal tumors typically occur in the stomach and small intestines and rarely arise in the colon, rectum, and esophagus.1,3,4,12 Although infrequent, extraintestinal gastrointestinal stromal tumors (eGISTs) have also been reported, involving the pancreas, uterus, omentum, retroperitoneum, and mesentery.4,13 Gastrointestinal stromal tumors are benign tumors with an indolent course, and only 20% to 30% of the neoplasms carry a malignant potential. 3 Most GISTs are discovered incidentally on imaging, endoscopy, or surgical exploration for an unrelated condition. 11 Some GISTs, however, can present as emergencies warranting prompt endoscopic or surgical intervention. 11 Herein, we highlight a unique case of a geriatric female who presented to the emergency room with severe abdominal pain and emesis. The patient was found to have an inflammatory mass on imaging, and the histopathological analysis revealed a primary mesenteric GIST. The tumor was resected, and adjuvant IM therapy was initiated to prevent recurrence.
Case Report
A 66-year-old female with a medical history of diabetes mellitus and a remote history of appendectomy (40 years ago) presented to our emergency department (ED) complaining of abdominal pain and vomiting for 4 days. The abdominal pain was described as band-like in fashion and was localized to the lower abdomen. The patient reported a low appetite for several weeks and unquantified weight loss. She denied taking any medications for her symptoms or recent changes in medications. She denied acid reflux, dysphagia, melena, hematochezia, diarrhea, fever, chills, dysuria, or gross hematuria. In the ED, the patient was in distress due to pain and nausea. She was afebrile and saturated 94% on ambient air. Vital signs were notable for tachycardia (110 beats per minute).
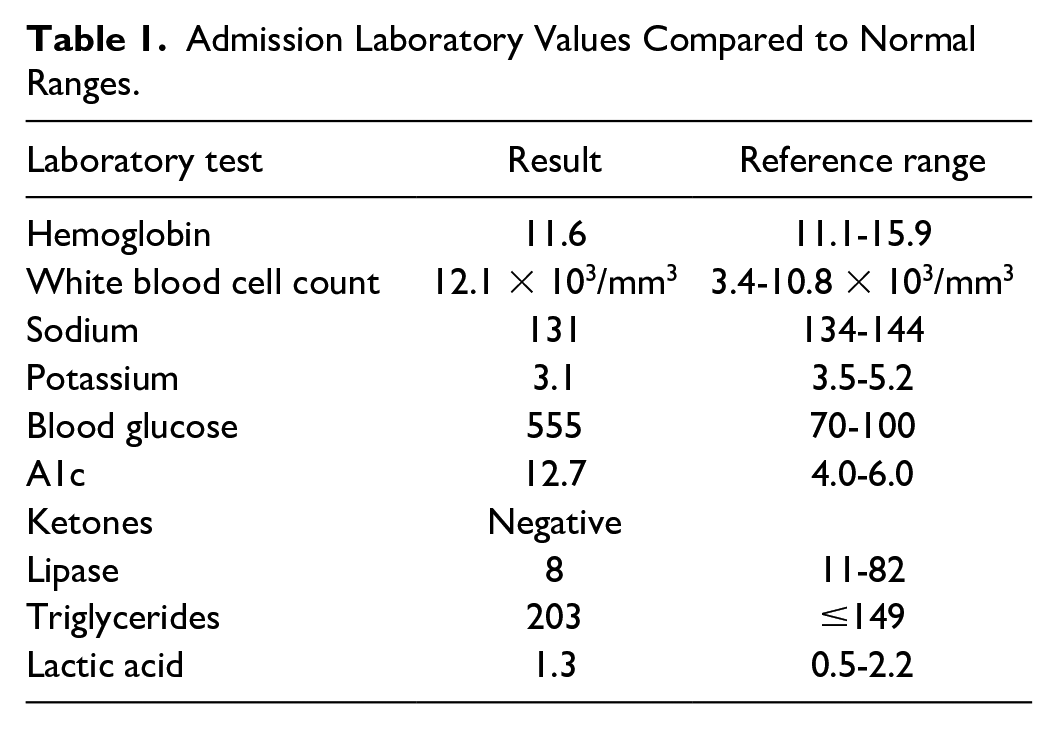
The abdomen was soft, obese, mildly distended, and diffusely tender on light palpation, with mild voluntary guarding. No flank ecchymosis or palpable organomegaly was appreciated on examination. The abdomen was dull to percussion in the lower quadrants with hypoactive bowel sounds. Besides the tachycardia, the rest of the physical examination was normal. Triage bloodwork revealed leukocytosis, anemia, hyponatremia, hypokalemia, and hyperglycemia (Table 1). The lipase level was normal, and no ketones were detected.
Admission Laboratory Values Compared to Normal Ranges.
A computed tomography angiography (CTA) of the chest, abdomen, and pelvis revealed a large inflammatory mass, measuring 17 × 13 cm, closely associated with small bowel loops in the abdomen and pelvis (Figure 1). The mass was associated with a moderate amount of free intraperitoneal air concerning for perforated viscus. Surgery and urology services were consulted for co-management. Because the mass was adherent to the right ureter, a double-J ureteral stent was placed to prevent injury and bruising during resection. General surgeons performed an exploratory laparotomy, resection of the intra-abdominal mass, and small bowel resection with primary anastomosis.

Axial view of a computed tomography angiography of the chest, abdomen, and pelvis showing a large inflammatory mass closely associated with small bowel loops in the abdomen and pelvis measuring 17 × 13 cm.
Intraoperatively, a 15-cm mass was noted adherent to the small bowel and the peritoneum in the pelvis (Figure 2). A LigaSure (Ethicon, Johnson & Johnson MedTech) device was used to remove the attachments, and an 80-mm GIA Stapler (Covidien products, Medtronic) was used to transect the small bowel, freeing the mass. This procedure was performed at both ends of the adherent loop of the bowel. The patient was admitted to the surgical intensive care unit (SICU) for postoperative care and management of diabetic ketoacidosis (DKA). The patient’s SICU stay was uneventful, and she was extubated the following day. Given an A1c level of 12.7%, the patient received diabetic education and was transitioned to subcutaneous insulin for glycemic optimization.

An intraoperative image showing mass adherent to the small bowel and peritoneum in the pelvis.
Histopathological examination of the resected mass revealed a high-grade, spindle-cell-type GIST, staged T4NxMx (Figure 3). The immunostaining profile was positive for CD117 and DOG1 (Figure 4), but negative for CD34, S100, Desmin, MART-1, and Pan CK. Prior to discharge, the patient underwent ureteroscopy and stent removal without complications. When seen as an outpatient a week later, the oncologist ordered c-KIT mutation testing and PDGFRA (platelet-derived growth factor receptor A) mutation analysis to establish the prognosis of mesenteric GIST and the patient’s likely response to IM therapy. The genomic test results confirmed the KIT (exon 11) mutation. Although the tumor had a low mitotic rate, the tumor was large enough to warrant the initiation of adjuvant IM therapy (400 mg daily) for 3 years with serial bloodwork and imaging, as per the National Comprehensive Cancer Network (NCCN) clinical guidelines. It has been 6 months since the patient started the chemotherapy, and she only experiences occasional nausea that resolves with anti-emetics.

A histopathology slide showing the proliferation of spindle cells with significant cytological atypia, increased mitotic activity, and necrosis that are arranged in short fascicles, causing focal obliteration of muscularis propria of the small intestine.

Immunochemistry slide showing expression of CD117 and DOG1 markers in tumor cells.
Discussion
Gastrointestinal stromal tumors are the most common mesenchymal tumors of the digestive tract and account for approximately 1% of all primary GI cancers.1-4 These tumors can arise in any part of the GI tract, but a higher burden has been observed in the stomach and small intestines.2,6,7,12,14 Although extremely rare, eGISTs have been identified in the uterus, urinary bladder, retroperitoneum, omentum, mesentery, liver, pancreas, and pleura.14-16 Gastrointestinal stromal tumors are usually found in individuals over the age of 50 years, with a slight male predominance.2,15 Until 1998, GISTs were thought to arise from smooth muscle cells; however, their histogenesis is now better understood.1,15
Mesenteric GISTs are exceedingly rare with unique clinicopathological features and a poorer prognosis.16,17 These tumors have been postulated to arise from the precursor cells of interstitial cells of Cajal in the mesentery, but such cells are yet to be isolated and studied. 16 Some scientists, however, have characterized mesenteric GISTs as intramural tumors that lost connection to the gut wall during growth. 16 These pathogenic mechanisms remain contested, each with its own limitations. Given their indolent clinical course and varied sizes, GISTs are mostly found incidentally on imaging, endoscopy, surgery, or autopsy.2,12,18 Symptomatic patients may present with nausea, vomiting, dysphagia, abdominal pain, abdominal distension, or early satiety, depending on the anatomic location of the GIST. On very rare occasions, patients may present with peritonism or obstructive symptoms due to the compressive effect of the tumor. 6 Ulcerated or ruptured GISTs can present with GI bleeding or bowel perforation, prompting urgent surgical intervention. 15
Diagnosing symptomatic GISTs involves a multifaceted approach to ensure accuracy and tailoring interventions to individual cases. Imaging aids in operative staging and assessment of the extent of the disease.17,19,20 Depending on size, GISTs may appear as exophytic or inflammatory masses on radiographic imaging. Endoscopy provides direct visualization of the GI mucosa, aiding the identification and characterization of GISTs. Pathological diagnosis is a vital part of confirming GISTs. Immunohistochemical (IHC) analysis of GISTs is pivotal for diagnosis, utilizing markers such as CD117, DOG1, and CD34. 19 About 85% to 95% of GISTs are driven by gain-of-function mutations in KIT receptor tyrosine kinase and/or DOG1. PDGRA gene mutations have been identified in 8% to 10% of the GISTs.2,3,16,21,22
The management of GISTs involves a multidisciplinary approach. The primary treatment for GISTs is surgical resection, aiming for complete removal of the visible and microscopic lesions. 17 Lymphadenectomy is not routinely indicated as GISTs rarely metastasize to lymph nodes.7,15,23 Adjuvant treatment with tyrosine kinase inhibitors, such as IM, is recommended for high-risk patients to reduce the risk of recurrence.3,4 Minimally invasive approaches can be considered for tailored resections, and laparoscopic resection is possible if it does not increase the risk of tumor rupture and peritoneal relapse. 17 Patients presenting as emergencies due to obstructive symptoms or GI hemorrhage are managed with surgery and adjuvant IM therapy based on mutational testing results. Our patient underwent tumor resection without complications and is currently in her sixth month of IM therapy. Although the tumor had a low mitotic rate, the tumor was large enough to warrant a 3-year course of IM therapy to prevent recurrence, as per the NCCN clinical guidelines. 24 Gastrointestinal stromal tumors frequently metastasize to the liver and peritoneum, often requiring surgical re-exploration. Imatinib mesylate therapy is the first-line therapy for metastatic, recurrent, and locally advanced GISTs.4,11,17
Sunitinib malate (SM; Sutent, Pfizer, New York, USA) is a tyrosine kinase inhibitor that has been used in GIST patients experiencing resistance or intolerance to IM.2,4,7,18 It is also indicated in patients with unique GIST genotypes, such as KIT mutations at exon 9.2,11 Second-generation tyrosine kinase inhibitors, such as nilotinib, sorafenib, dasatinib, and regorafenib, are occasionally used in patients with IM and SM resistance.4,7 Tumor size, anatomic location, and mitotic count are independent prognostic factors for GISTs.11,25 The site of origin of a GIST is associated with its clinical behavior, and patients with tumors in the rectum or small intestine have a worse prognosis. Successful management of GISTs requires an aggressive and meticulous approach, combining surgical intervention with molecular therapy.
Conclusion
Here, we share an interesting case of an elderly female patient with a remote surgical history of appendectomy who presented to our ED with an acute abdomen. A mesenteric GIST was observed on imaging and confirmed on histopathology. The tumor was resected and IM adjuvant chemotherapy started to prevent tumor recurrence. Gastrointestinal stromal tumors are the most common mesenchymal tumors in the GI tract and arise from interstitial cells of Cajal. Gastrointestinal stromal tumors have an indolent clinical course and often present with non-specific signs and symptoms such as nausea, vomiting, abdominal pain, or distension. Peritonism, GI bleeding, bowel obstruction, and perforation are rare.
Footnotes
Author Contributions
LB conceptualized the idea for this case report. SY, RB, SV, AK, and RB helped with data curation, collection of pertinent patient data, and writing. MK edited, fact-checked, and proofread the final version of the manuscript.
Data Availability Statement
Further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
