Abstract
Here, we report a rare case of a 22-year-old female presenting with recurrent chest pain mimicking inferior ST-elevation myocardial infarction (STEMI) but ultimately attributed to an aortic aneurysm. Despite facing initial challenges in diagnosis, such as normal troponin levels and temporary electrocardiogram (ECG) changes, advanced imaging showed a large mass in the chest pressing on the right coronary artery. Prompt multidisciplinary intervention, including surgical resection of the aneurysm, led to successful management and improved outcomes. This case highlights the importance of considering unusual etiologies in atypical presentations of myocardial infarction, necessitating comprehensive evaluation and collaboration among various specialties for optimal patient care.
Introduction
The intersection of cardiovascular pathology presents distinctive challenges in clinical situations. The occurrence of inferior ST-elevation myocardial infarction (STEMI) secondary to an aortic mass illustrates a significant overlap between cardiac and vascular abnormalities.1,2 Although inferior STEMIs typically implicate the coronary artery system, the involvement of an aortic mass introduces a distinctive dimension, necessitating a comprehensive understanding of the complex relationship between aortic pathologies and myocardial ischemia. 2 This complex interplay underscores the importance of a multidisciplinary approach, blending the expertise of cardiologists, cardiac surgeons, and vascular specialists. This introduction sets the stage for exploring the detailed complexities of how an aortic mass can contribute to myocardial infarction, shaping discussions around diagnosis, treatment modalities, and the broader implications for patient care.3,4 As medical understanding advances, delving into specific instances like Inferior STEMI associated with aortic masses becomes pivotal, offering valuable insights into refining clinical strategies and enhancing outcomes for individuals facing this challenging combination of cardiac and vascular disorders. 5
Case Presentation
A 22-year-old newly married woman presented to our emergency department with recurrent, intermittent attacks of retrosternal stabbing chest pain lasting for 1 month. The pain typically lasted a few minutes and radiated to her back and both shoulders, accompanied by nausea. She noted an increased awareness of her heartbeat with physical activity and found relief upon sitting or lying down. Despite 3 previous visits in the last 10 days, during which she received intravenous fluids, paracetamol, and omeprazole, along with reassurance that the pain was musculoskeletal, it persisted, ranging from mild to severe, prompting her return. Her past medical history is unremarkable, and she is a non-smoker and non-drinker. There is a family history of maternal hypertension and paternal type 2 diabetes, but no similar conditions or history of sudden cardiac death, autoimmune disease, or cardiovascular disease in the family. Notably, there are no signs or family history suggestive of connective tissue disorders such as Marfan syndrome, Ehlers-Danlos syndrome, or other genetic aortopathies.
On examination, she appeared conscious, oriented, mildly dyspnoeic but not tachypnic, and anxious due to moderate pain. Physical findings included normal chest auscultation, a grade III diastolic murmur at the left lower sternal border, and no abdominal abnormalities. In addition, she had a height of 158 cm, weight of 55 kg, and a calculated body mass index (BMI) of 22, indicating a normal habitus.
Her vital signs were as follows: oxygen saturation = 96% on room air, pulse rate = 88 beats/min, temperature = 37.9°C, and respiratory rate = 22 breaths/min. Blood pressure was 120/70 mm Hg at the right brachial artery and 126/75 mm Hg at the left brachial artery, with no evidence of radial or femoral artery delay.
Aortic dissection and vasospastic angina were the primary deferential diagnosis. An emergency electrocardiogram (ECG) was conducted, revealing inferior ST-segment elevation at lead II, III, and avF as shown in Figure 1, despite normal serum troponin I level 0.02 ng/mL (0.0.04 ng/mL).

Emergency ECG showing ST-segment elevation at leads II, III, and aVF with reciprocal ST-segment depression at lead aVL.
The catheter laboratory was notified, and the patient was promptly referred to a tertiary cardiac hospital by ambulance. Upon arrival, a follow-up ECG after 30 minutes revealed normal sinus rhythm without ischemic changes (Figure 2), occurring simultaneously with the patient being pain-free and clinically stable.
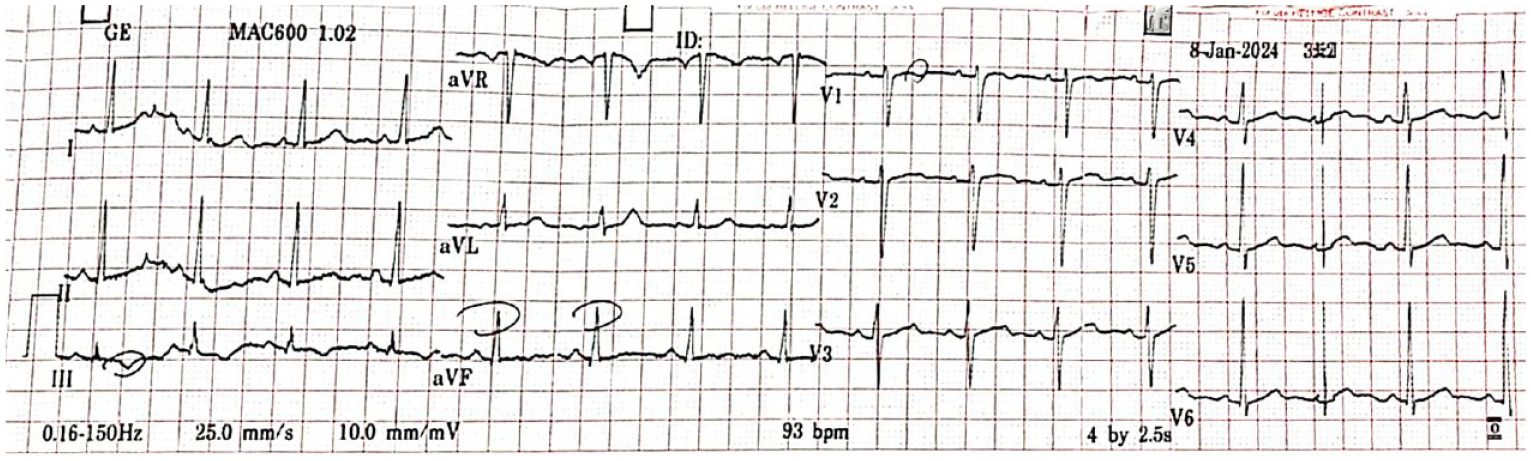
ECG 30 minutes after presentation at the tertiary cardiac hospital revealing normal ST segment and sinus rhythm.
The patient was transferred to the medical ward for further evaluation and follow-up. A chest X-ray was conducted, revealing a large mediastinal mass (Figure 3). Her complete blood count and renal function test were normal.

Chest X-ray PA view showing large mediastinal mass.
A transthoracic echocardiogram revealed a normal ejection fraction and moderate aortic regurgitation without pericardial effusion or regional wall motion anomalies. Based on the nature of the pain and ECG changes, a decision was made to proceed with invasive coronary intervention. The result showed normal blood flow in the main coronary arteries (Figure 4).

(A) Coronary angiography showing normal right coronary artery RCA blood flow (red arrow). (B) Normal left anterior deciding artery LAD (red arrow) and left circumflex artery LAD (white arrow) blood flow.
However, a sac was observed behind the ascending aorta, aortic arch, and descending aorta, mainly compressing the right coronary artery (RCA), resulting in inferior STEMI like future in the ECG (Figure 5).

Aortic injection using a Judkins left (JL) catheter shows a sac behind the ascending aorta, aortic arch, and descending aorta.
We proceeded further with a chest computed tomography (CT) angiogram, which revealed a well-defined anterior mediastinal soft tissue density lesion measuring about 84.9 × 67.2 mm, seen at the aorto-pulmonary window. The lesion exhibited a mass effect, compressing the main pulmonary artery and proximal ascending aorta. It showed a calcified wall with enhancement after intravenous contrast, with 2 different densities within. And it showed close contact, almost inseparable from the ascending aorta. A definite relation with the aorta could not be established, whether it originated from or was invading it (Figure 6).

Chest CT angiogram revealing well-defined anterior mediastinal soft tissue density lesion of about 84.9 × 67.2 mm seen at aorto-pulmonary window with mass effect compression of the main pulmonary artery and proximal ascending aorta (red arrows and cross).
After a multidisciplinary team meeting involving a cardiothoracic surgeon, anesthesiologist, cardiologist, and the patient with her family, it was decided to proceed with open heart surgery.
During the surgery, an aneurysm in the aortic wall near the sinus of valsalva was discovered (Figures 7 and 8).

Aortic defect near sinus of valsalva after aneurysm resection.

Aneurysm tissue sample after resection.
The surgical procedure involved resection of the aneurysm and interposition graft placement (Figure 9).

Aortic interposition graft placement.
A sample of the aneurysm tissue was sent for histopathological analysis, revealing fibrosis, hyalinization, necrosis, and calcification. The patient’s condition improved post-surgery, and she was discharged home with monthly follow-up appointments. Subsequent echocardiography showed normal results, and she later conceived and delivered a single female fetus via normal vaginal delivery.
Discussion
Ascending aortic aneurysms usually occur due to cystic medial degeneration, which causes weakening of the aortic wall leading to dilation and aneurysm formation. It can occur due to aging, and hypertension is known to accelerate the process. 6 Sporadic ascending aortic aneurysm is usually seen in patients by 60 years of age due to reduced elasticity and compliance predisposing to dilation. 7
In the younger age group, ascending aortic aneurysm can be due to familial predisposition and genetic syndromes, such as Marfan syndrome, Ehlers-Danlos syndrome, Loeys-Dietz syndrome, and Turner syndrome. These conditions are characterized by connective tissue abnormalities, including defects in fibrillin-1 (Marfan syndrome) or collagen synthesis (Ehlers-Danlos syndrome), leading to structural weaknesses in the aortic wall. 6
In additionally, congenital anomalies like bicuspid aortic valve (BAV) represent another significant risk factor for abdominal aortic aneurysm (AAA) formation. The abnormal hemodynamic stress on the ascending aorta in BAV patients predisposes them to aortic dilation and aneurysm development. Studies have shown a higher prevalence of AAAs in individuals with BAV compared to those with a tricuspid aortic valve.
Furthermore, inflammatory conditions such as the Takayasu arteritis can lead to aortic aneurysm formation, albeit rarely. The Takayasu arteritis primarily affects the aorta and its major branches, leading to arterial inflammation, stenosis, and aneurysmal degeneration. The inflammatory cascade in the Takayasu arteritis contributes to vascular remodeling and weakening of the aortic wall, culminating in aneurysm formation. 8
Inferior STEMI secondary to an aortic aneurysm is a rare and challenging clinical scenario that demands heightened diagnostic vigilance. 9 The aorta, a vital conduit for systemic circulation, can harbor various pathologies, including tumors or thrombi, compromising coronary blood flow. 10
The challenges arise from the potential for embolic events originating from the aortic aneurysm, leading to downstream coronary artery occlusion and subsequent myocardial infarction. The inferior distribution is a common manifestation due to the proximity of the coronary ostia, complicating the electrocardiographic interpretation. 11
Accurate diagnosis relies on a comprehensive approach. Advanced imaging modalities, particularly transesophageal echocardiography (TEE) and computed tomography angiography (CTA), offer insights into the nature and extent of the aortic aneurysm. 12 Timely recognition of this unique etiology is crucial to guide appropriate management.
Therapeutic strategies include addressing both the aortic aneurysm and myocardial infarction. Surgical intervention may be warranted for the aortic mass, aiming to prevent embolic events and restore normal coronary perfusion. Simultaneously, myocardial reperfusion strategies, such as percutaneous coronary intervention (PCI) or thrombolytic therapy, are deployed to salvage ischemic myocardium. 13
Post-intervention, close monitoring for potential embolic complications, arrhythmias, or recurrent infarctions is essential. Anticoagulation may be indicated to mitigate the risk of further embolism from the aortic aneurysm. 14
Conclusion
This case stresses the need to explore uncommon causes in chest pain resembling myocardial infarction. It highlights the importance of a collaborative, multidisciplinary approach, advanced imaging, and clear communication among health care providers for accurate diagnosis and effective treatment. Ongoing research and education in cardiovascular medicine are vital for enhancing outcomes in challenging clinical scenarios.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
