Abstract
Macrophage activation syndrome (MAS) is a form of secondary hemophagocytic lymphohistiocytosis (HLH) when it occurs in the context of rheumatologic disorders. HLH is a rare and potentially life-threatening syndrome characterized by excessive immune system activation. It is mainly seen in children and can be genetic based or related to infections, malignancies, rheumatologic disorders, or immunodeficiency syndromes. MAS can present with nonspecific symptoms, leading to a delay in diagnosis. This report describes a case of a 64-year-old female with marginal zone lymphoma and systemic lupus erythematosus who presented with a purpuric rash and acute kidney injury. She underwent a kidney biopsy and was diagnosed with MAS. This case highlights the importance of promptly recognizing MAS’s symptoms and signs, allowing timely diagnosis and early therapeutic intervention. This potentially fatal condition tends to respond well to rapid treatment initiation with corticosteroids and to address the underlying condition.
Keywords
Introduction
Macrophage activation syndrome (MAS) is referred to as secondary hemophagocytic lymphohistiocytosis (HLH) when it occurs in the context of rheumatologic disorders. 1 HLH is an uncommon and potentially fatal syndrome characterized by hyperinflammation and tissue damage secondary to dysregulated immune activation. 2 It can be a genetic disorder known as primary HLH or triggered by infections, malignancies, mainly malignant lymphoma, or rheumatologic conditions as secondary HLH. 3 Secondary HLH may also manifest as a result of malignancy, either at the initial presentation or relapse, throughout malignancy treatment (chemotherapy) or following bone marrow transplantation. 3 MAS can present as an acute or subacute febrile illness, with nonspecific symptoms, signs, and multiorgan involvement. 4 Common laboratory abnormalities observed in MAS are pancytopenia, elevated ferritin levels, elevated serum transaminases, hyperbilirubinemia, hypoalbuminemia, and an abnormal coagulation profile. 4 Serum ferritin is a valuable diagnostic tool, as this biomarker’s rapid and significant elevation is frequently observed in MAS. 4 The H-score is used as a diagnostic score to estimate the probability of HLH (with points for immunosuppression, fever, organomegaly, cytopenias, levels of triglycerides, ferritin, alanine aminotransferase, and fibrinogen, and hemophagocytosis on the bone marrow aspirate). An H-score greater than or equal to 250 confers a 99% probability of HLH, whereas a score of less than or equal to 90 confers a less than 1% probability of HLH.5,6
We report a case of a 64-year-old patient with marginal zone lymphoma (MZL) and concurrent rheumatologic disorders who presented to the hospital with an acute rash and acute kidney injury (AKI) and was ultimately diagnosed with MAS.
Case Presentation
A 64-year-old female with hypertension, systemic lupus erythematosus (SLE) (diagnosed outside the hospital and left untreated by self-discontinuation of the medication), rheumatoid arthritis (RA), history of deep vein thrombosis, and MZL of the conjunctiva (since 2021) was admitted to the hospital due to severe anemia. She was found to have a right renal mass on a computed tomography scan. She underwent a biopsy of the mass, which showed recurrent MZL. Rituximab was initiated with the continuation of hydroxychloroquine. A month later, the patient presented to the hospital due to a rash for 1 week. The rash started on her left lower extremities and subsequently spread to the abdomen, back, and contralateral side. She had self-discontinued hydroxychloroquine 3 weeks prior to admission. Upon presentation, the patient was hemodynamically stable, with vital signs only remarkable for mild tachycardia, a heart rate of 117 beats per minute. Physical examination revealed a petechial nonblanching rash on the abdomen and lower extremities along with some purpura and blisters on the lower extremities and bilateral lower extremity 2+ pitting edema. The rash was ultimately diagnosed as leukocytoclastic vasculitis (Figure 1).

Petechial rash, purpura, and blisters on patient’s left lower extremity-leukocytoclastic vasculitis.
Initial laboratory results showed mild neutrophilia, mild normocytic anemia, thrombocytosis, elevated blood urea nitrogen and creatinine, metabolic alkalosis, abnormal coagulation profile including elevated prothrombin time and international normalized ratio, elevated erythrocyte sedimentation rate and C-reactive protein, hypoalbuminemia, and remarkably elevated ferritin (Table 1). Viral serology screening, including hepatitis panel and HIV, was negative. The patient was admitted with a working diagnosis of severe sepsis; her inpatient course was complicated by acute hypoxic respiratory failure secondary to cryptococcal pneumonia and pulmonary edema, necessitating mechanical ventilation. Hemodialysis was initiated due to oliguric AKI and volume overload. Kidney biopsy revealed findings with multiple small renal arteries occluded by macrophages leading to ischemic acute tubular necrosis and no evidence of glomerulonephritis (shown in Figure 2). The H-score was 135, conferring a 9% to 16% likelihood of HLH. 5 The patient was started on high-dose steroids, cyclophosphamide 750 mg, etoposide, vincristine, and hydroxychloroquine. There was an improvement in her respiratory status and laboratory parameters as leukocytosis and thrombocytosis resolved. The patient’s clinical condition improved significantly to the point that she was discharged home. She remained oliguric with plans to continue outpatient hemodialysis and chemotherapy.
Initial Laboratory Results.
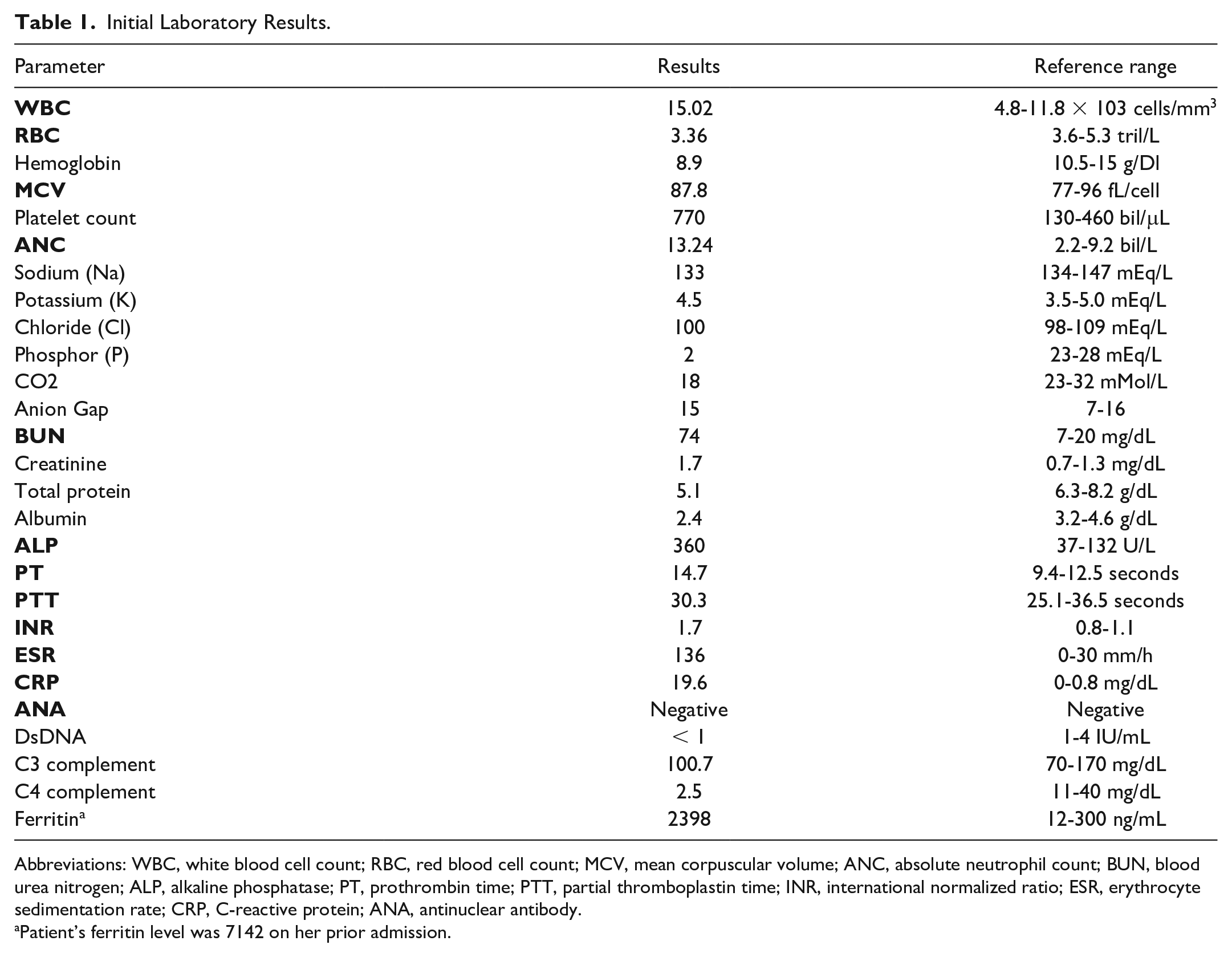
Abbreviations: WBC, white blood cell count; RBC, red blood cell count; MCV, mean corpuscular volume; ANC, absolute neutrophil count; BUN, blood urea nitrogen; ALP, alkaline phosphatase; PT, prothrombin time; PTT, partial thromboplastin time; INR, international normalized ratio; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; ANA, antinuclear antibody.
Patient’s ferritin level was 7142 on her prior admission.

Trichrome stain shows 2 small arteries (arrows) whose lumens are filled with macrophages (pale cells in the lumen).
Discussion
In the adult population, the majority of secondary HLH cases are linked to triggers such as malignancies, infections, immunodeficiency, and rheumatologic conditions. This contrasts with pediatric syndromes, where underlying genetic abnormalities and juvenile RA are predominantly associated with HLH. 7 Among adults, hematological malignancies, particularly non-Hodgkin lymphoma, stand out as the most prevalent malignancy associated with secondary HLH. 7 In B-cell lymphoma, the pathogenesis of HLH may be attributed to the abnormal rise in inflammatory cytokines emanating from lymphoma cells. 7 MAS can occur in individuals with rheumatologic conditions and is often associated with an SLE flare. In patients with a history of SLE, MAS, and an active lupus flare closely resemble each other, making it challenging to distinguish between these 2 conditions. High suspicion for MAS is warranted when the infection is ruled out, or multiorgan dysfunction persists despite treatment of the underlying condition. In this particular case, the patient’s underlying MZL and simultaneous rheumatologic conditions, including SLE and RA, were the most likely triggers for the development of MAS. 8 However, the H-score exhibited a low to intermediate risk. The initial effective therapy for secondary HLH involves a combination of immunosuppressive medications that target hyperactive T-cells and activated macrophages/histiocytes such as etoposide, steroids, and high-dose intravenous immunoglobulin to target histiocytes and high-dose steroids, cyclosporine A, and antithymocyte globulin for activated T-cells. In stable patients with MAS, HLH-specific therapy can be delayed while treating the underlying triggering condition with corticosteroids and disease-specific therapy. 8 Due to the nonspecific nature of the syndrome, some clinicians argue against a definitive diagnosis of MAS/secondary HLH without observing evidence of hemophagocytosis in a biopsy sample. 8 Nevertheless, considering the life-threatening nature of MAS and the occasional absence of hemophagocytosis in biopsy findings, maintaining a high suspicion for MAS and initiating timely treatment is critical. 2
Conclusion
This case highlights the importance of considering MAS in patients with underlying malignancies and rheumatologic conditions, particularly in the setting of lymphomas and untreated SLE. It also shows how a biopsy other than a bone marrow aspirate could reveal histopathologic findings of MAS. As MAS responds rapidly to steroids and immunosuppressant agents, timely workup for early detection is essential to reduce the high mortality rate of untreated cases. Initiating prompt and appropriate treatment in cases of high suspicion could potentially prevent fatal consequences of MAS.
Footnotes
Acknowledgements
The authors express their appreciation to Dr Craig W. Zuppan, MD, for his valuable contribution in preparing the kidney biopsy slide for this case report.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.N. is on the speaker bureau for Calliditas Therapeutics. She is also a consultant for Calliditas Therapeutics and Otsuka Pharmaceutical Co.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
