Abstract
Pfizer/BioNTech (BNT162b2) is a messenger RNA (mRNA) vaccine that is highly effective in preventing the most severe outcomes of COVID-19 infection. Nucleoside-modified severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) mRNA vaccines induce effective stimulation of T follicular helper (TFH) cells, leading to a robust germinal center B cell response. Side effects from the BNT162b2 vaccination, including significant lymphadenopathy, have been reported previously. Here, we present a case of angioimmunoblastic lymphoma (AITL), a rare, peripheral T-cell lymphoma with RHOA-G17v-mutated gene developing in a patient following BNT162B2 vaccine with a plausible explanation. A 60-year-old Asian female received her first dose of Pfizer BNT162B2 mRNA vaccine in August 2021. Right after her vaccination, she developed right axillary lymphadenopathy. She received her second vaccine dose in September 2021. Thereafter, she developed lymph node (LN) enlargement in her neck and groin. She underwent left posterior cervical and left groin LN excisional biopsy in April 2022 due to persistent palpable lymphadenopathy. Biopsy results then demonstrated benign follicular hyperplasia. For progressive B symptoms, a right axillary LN biopsy was done, which demonstrated AITL, with molecular studies revealing mutation in TET-2, IDH-2, and RHOA-G17v genes. Progression of AITL following BNT162B2 mRNA vaccine is limited in literature. Our case demonstrates a plausible correlation between the diagnosis of AITL following mRNA vaccination due to the malignant transformation of the TFH cells in patients who have a predisposing mutation of RHOA-17v. Given the rarity of AITL and the heterogeneity of molecular findings, more studies are needed to establish such an association.
Introduction
Through their primary effect on T-follicular helper (TFH) cells, mRNA vaccines stimulate and maintain a strong germinal center response.1,2 However, clonal mutations in TFH cells can lead to unregulated growth and oncogenesis in predisposed individuals, manifested as angioimmunoblastic lymphoma (AITL) and other T-cell lymphomas. Angioimmunoblastic lymphoma is a subset of peripheral T-cell lymphomas that is believed to arise from a population of peripheral T cells called TFH secondary to rearrangement of T cell receptors, and its proliferation is sustained by the master transcription factor B-Cell Lymphoma 6 (Bcl6).3-5 In this case report, we present a case of plausible clinical development of AITL following administration of a BNT162b2 mRNA vaccination.
Case
A 60-year-old female patient with a medical history of distant, resected melanoma in situ received her first dose of Pfizer/BioNTech (BNT162b2) mRNA vaccine in August 2021. On the same day, she noticed palpable right axillary lymph node (LN) enlargement. She received the second dose of the same vaccine as per the vaccine schedule 4 weeks later. Thereafter, she noticed multiple other LN enlargements in her neck and groin area as well. She visited her primary-care physician for an annual visit around 6 months from her first vaccine dose. Because of persistent LN enlargement and nonspecific B symptoms, the physician recommended an excisional biopsy of the enlarged posterior cervical and left inguinal LNs. The LN pathology revealed benign follicular hyperplasia with positive cells for Epstein-Barr virus (EBV), with no evidence of malignancy. However, due to persistent LN enlargement and B-symptoms, an oncology referral was sent. Oncology evaluation and workup revealed diffuse lymphadenopathy noted on contrast-enhanced computed tomography (CT) chest, abdomen, and pelvis. Her initial laboratory work-up was normal including hemoglobin, white blood cell count, and platelet counts, except LDH 525 U/L.
Around 2 weeks later from her oncology appointment, she presented to the emergency department (ED) with acute onset intractable nausea and vomiting associated with generalized weakness and fatigue. In the ED, vitals were stable, but lab work-up revealed her hemoglobin 6.1 g/dL, hematocrit 13%, platelet count 356,000/ μL, LDH 737 U/L, total bilirubin 6.0 mg/dL, indirect bilirubin 5.3 mg/dL, and undetectable haptoglobin. The initial peripheral smear showed significant agglutination rendering nonanalysis of reticulocyte count and direct Coombs testing. The patient was accordingly diagnosed with acute hemolytic anemia (AIHA) and admitted for prednisone 80 mg per day, rituximab, and blood transfusion as needed. After discussion with inpatient oncology services, she underwent an excisional biopsy of the right axillary LN, which was her primary LN which had enlarged following the initial vaccination. Pathology showed effaced nodal architecture by atypical lymphoid infiltration composed of a mixture of small lymphocytes with nuclei that showed condensed chromatin and medium-sized cells with round to irregular nuclei and clear cytoplasm. In the background, there was prominent vascular proliferation and numerous inflammatory cells. Immunohistochemistry on the right axillary LN specimen showed infiltrates that marked as a neoplastic population with CD3 (dim)-positive T cells that co-expressed CD4, CD2, CD5, TCR beta F1, dim to diminished expression of CD7, and high Ki-67 proliferation index. They also expressed the TFH cell marker CD10, some of which associated with focally expanded CD21-positive follicular dendritic cell meshwork. Fluorescence in situ hybridization (FISH) was positive for Epstein-Barr virus-encoded RNA (EBER). The above description was consistent with the diagnosis of angioimmunoblastic T-cell lymphoma as shown in Image 1. Next-generation sequencing showed TET-2, IDH-2, and RHOA-G17V mutations.
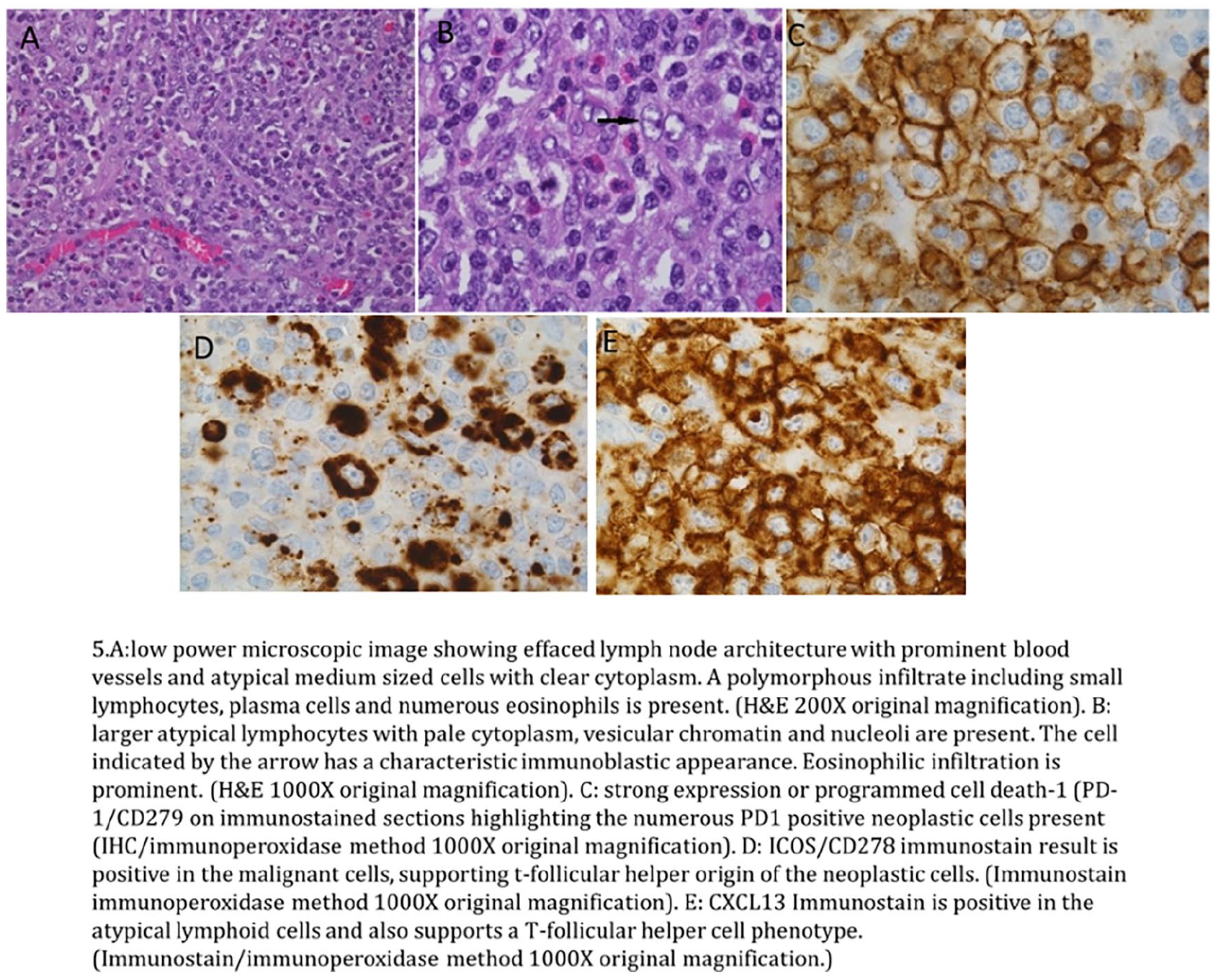
Following the diagnosis of angioimmunoblastic T-cell lymphoma, a positron emission tomography/computed tomography (PET/CT) scan was performed and showed hypermetabolic lymphadenopathy in the involved LNs. There was no evidence of disease in the bone marrow. Following stabilization of her AIHA with steroids and rituximab, the patient was discharged from the hospital. After shared decision-making, she received 6 cycles of CHOEP regiment with cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, vincristine 1.4 mg/m2 on day 1, etoposide 100 mg/m2 iv over 60 minutes daily on days 1 to 3 and prednisone at 100 mg daily PO days 1 to 5 total (CHOEP) as per the National Comprehensive Cancer Network guidelines. Repeat interim PET/CT following cycle 3 showed excellent response with no hypermetabolic lymphadenopathy. She eventually underwent autologous stem cell transplant and has been in complete remission since then.
Discussion
To date, results from the phase III clinical trials showed that both the Pfizer/BioNTech (BNT162b2) and Moderna (mRNA-1273) mRNA vaccines achieved 90% to 95% efficacy in protecting against COVID-19.6,7 It has significantly helped to fight the COVID-19 pandemic. Over the same period, several rare hematological case reports have been reported with their use, such as autoimmune hemolytic anemia, immune thrombocytopenic purpura (ITP), benign lymphadenopathy at the injection site, and progression of hematological malignancies.8,9
The immunity acquired against pathogens and vaccines primarily requires the formation and maintenance of germinal centers, which is driven by and is directly proportional to the degree of TFH cells activation. T follicular helper cells, in turn, induce B cell affinity maturation, class switching, and subsequent development of plasma and memory cells.5,10,11
The essential role of TFH cells is to induce long-lived neutralizing antibodies, and huge efforts have been used to develop vaccines that promote robust TFH responses. It is proven that severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) mRNA vaccines induce well-established germinal center responses, 1 which were shown to induce more potent TFH responses compared to single-peptide vaccines,2,7,12 and with higher responses after each subsequent dose. 1 The germinal center reaction is reflected radiologically and, less often, clinically by lymphadenopathy, 13 and studies on BNT162b2 (Pfizer) and mRNA-1273 SAR-CoV2 mRNA vaccine (Moderna) showed a higher incidence of lymphadenopathy in the former. 14 The reactive hypermetabolic sites were sometimes detected distant from the draining LNs, which were proved later to be benign by revealing prominent germinal centers with reactive changes on biopsy.15,16 It is a special concern with patients who are already receiving treatment for malignancy, where it can lead to misinterpretation of treatment response or disease status.8,17
Our case showed the expected course of lymphadenopathy as described above. Unfortunately, those hypermetabolic responses were not only reactive changes but were associated with the development of AITL. Although the initial biopsy showed benign follicular hyperplasia, but the persistent lymphadenopathy, presence of B symptoms, detection of EBV, and development of AIHA suggested underlying hematological malignancy and prompted to proceed with another excisional biopsy of the right axilla. It is unclear, however, whether AITL appeared de novo or was simply unmasked after vaccination with underlying mutations TET-2, IDH, RHOA G17V. To our knowledge, this is the second case undermining the relationship between BNT162b2 (Pfizer-BioNTech) and AITL. Goldman et al described a case where a patient manifested AITL 6 months after receiving an initial series of 2 BNT162b2 mRNA vaccine doses, administered with a 1-month interval. The presentation initially featured cervical lymphadenopathy and FDG-avid, multifocal hypermetabolic lymphadenopathy both above and below the diaphragm, with subsequent deterioration 2 weeks following administration of the booster dose (the third dose) of BNT162b2 mRNA vaccine, as evidenced by increase in the number, size, and metabolic activity of the previously affected LNs, along with the involvement of LNs in other sites. 8
Angioimmunoblastic lymphoma has a TFH gene expression signature and TFH cell-associated markers. 18 It is well established from multiple systematic reviews of immunohistochemical studies and xenograft experiments that malignant cells in AITL originate from CD4+ cells.18,19 T follicular helper cells are a subset of CD4+ T cells whose differentiation is driven by the transcription factor Bcl6. Excessive TFH cell activity can lead to diseases such as autoimmunity and lymphomas,8,9,19 which was found in our case as presented with AIHA and AITL.
The most common mutations involved are TET-2, IDH-2, DNMT3A, and RHOA G17V. Among these, RHOA G17V is detected in around 50% to 70% of cases. These mutations appear to act partly by dysregulation of DNA methylation.20-22 RHOA G17V mutation was found particularly sensitive to the effect of mRNA vaccines, corresponding to the observation in experimental mice harboring RHOAG17V and TET-2 mutation, which developed AITL after immunization with sheep red blood cells (SRBCs) to induce T cell proliferation in the germinal center. In these experimental mice further, analysis showed expansion of the CXCR5+ and PD1+ T cell population.22,23 Similar findings are evident in our patient as shown in Image.1.
This case shows a plausible association of mRNA vaccine and lymphomatous processes, especially AITL. With having TFH cell signature of AITL and the role of mRNA vaccine to induce robust TFH response and germinal center proliferation, it is possible that individuals harboring these mutations are at a higher risk of developing or unmasking underlying lymphoma. Given the rarity of AITL and the heterogeneity of molecular findings, more studies are needed to establish such an association. It is important to note that the overall benefit of mRNA vaccines in preventing COVID-19, and its complications far outweighs any rare potential risks.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethic Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
