Abstract
Sinistral portal hypertension (SPH), also known as segmental portal hypertension, is a complication of pancreatic disorders and an extremely rare cause of upper gastrointestinal (GI) bleeding. SPH is observed in patients without cirrhosis and arises from splenic vein thrombosis. Unmitigated backflow of blood may cause gastric venous congestion and ultimately GI hemorrhage. Herein, we report a rare case of massive hematemesis due to SPH in a male patient with a history of chronic pancreatitis and pancreatic pseudocyst. Our patient was successfully treated with endoscopic necrosectomy followed by open splenectomy, distal pancreatectomy, and partial gastric resection.
Keywords
Introduction
Pancreatic pseudocysts (PPs) are a common complication of acute and chronic pancreatitis. 1 Strictures, stones, protein plugs, and acute inflammation can cause pancreatic duct disruptions and blockages, and increased intraductal pressure leading to pseudocyst formation. 2 PPs can rupture causing hemorrhage, peritonitis, and sepsis, and may compress on the spleen causing atraumatic rupture, infarction, or splenic vein thrombosis (SVT). 3 Sinistral portal hypertension (SPH) is an infrequent complication of pancreatic disorders and a very rare cause of upper gastrointestinal (UGI) bleeding. 4 Because of its rarity, SPH is an overlooked complication of chronic pancreatitis and PPs.5,6
Case Report
A 45-year-old man with a medical history of diabetes mellitus, chronic pancreatitis, and PP status post cystogastrostomy stent placement presented to the emergency department (ED) after a syncopal episode. Before the event, the patient experienced sharp abdominal pain, nausea, and shortness of breath. He denied having chest pain, palpitations, or seizure-like activity during the event. The patient had normal vital signs in the ED, and the physical examination was significant for epigastric tenderness without peritoneal signs or flank ecchymosis. The rest of the physical examination was normal. Initial laboratory values were significant for hemoglobin of 6.8 g/dL, and the patient was transfused 2 units of packed red blood cells (PRBCs). The liver function tests, CA 19-9 tumor marker, and the rest of the blood chemistry results were unremarkable. A computed tomography (CT) angiography of the abdomen and pelvis showed splenomegaly, SVT, and a gastrocystostomy tube with a large pseudocyst. The patient was admitted to the regular medical floors for observation.
The next morning, a rapid response team was called for multiple episodes of hematemesis. We administered 2 L of normal saline boluses, 2 units of PRBCs, 50 µg of octreotide, and 80 mg of pantoprazole, intravenously (IV). Esophagogastroduodenoscopy (EGD) revealed old blood and blood clots in the gastric fundus and body. Type 1 isolated nonbleeding gastric varices were noted in the gastric fundus (Figure 1), and necrosectomy of the PP was performed (Figure 2). Due to worsening abdominal pain and recurrent episodes of acute blood loss, splenectomy was performed.
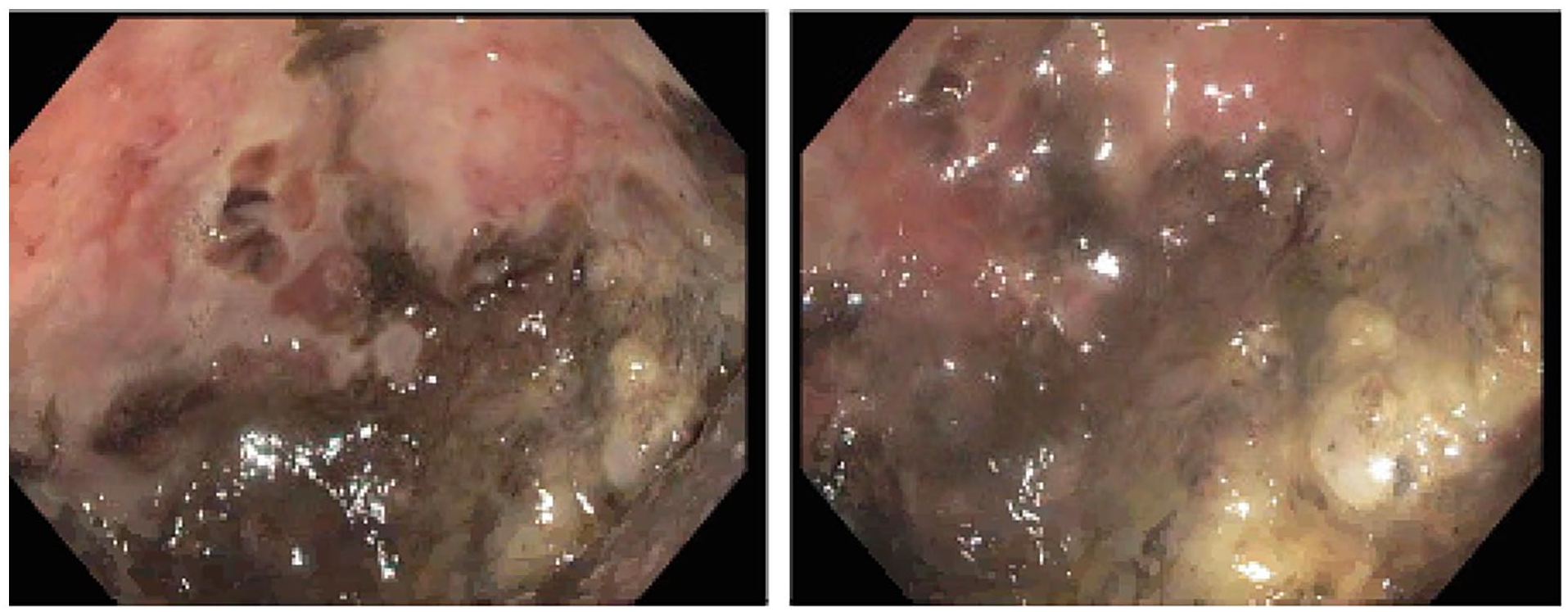
Endoscopic image showing type 1 isolated gastric varices without active bleeding. Old blood and blood clots can be seen in the gastric fundus and gastric body.
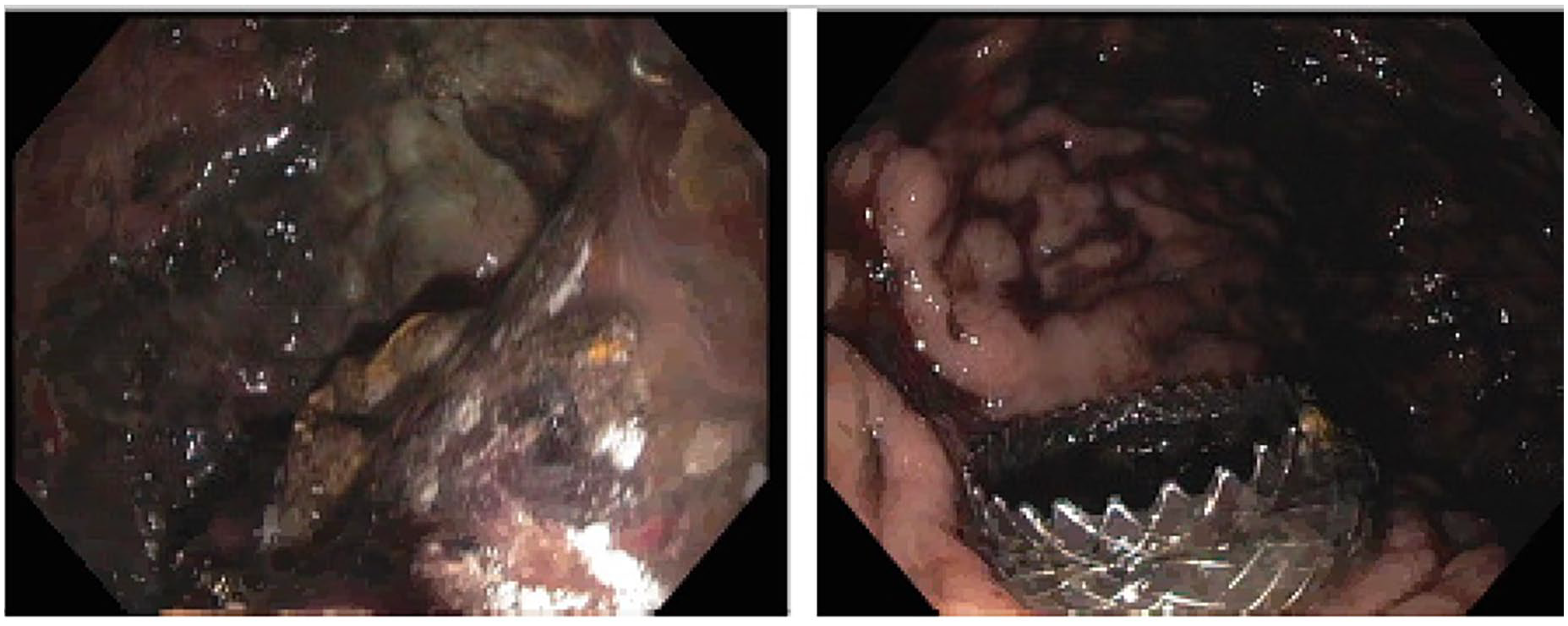
Endoscopic image showing a cyst cavity during endoscopic necrosectomy. Preexisting cystogastrostomy stent can be seen on the right image panel.
Before surgery, we administered PCV20, H. influenzae type b, meningococcal quadrivalent, and meningococcal serogroup B vaccines for prophylaxis against encapsulated bacteria. During surgery, an inspection of the intra-abdominal cavity and laparoscopic mobilization of the splenic flexure showed hepatosplenomegaly with dense adhesions to the spleen. Given the patient’s complex anatomy and location of the pseudocyst, a decision was made to convert from laparoscopic to open surgery. Further inspection revealed perisplenitis and a PP densely adhered to the posterior aspect of the stomach as well as to the spleen hilum. The axial stent was removed and the posterior gastric body gastrotomy secondary to the stent was primarily repaired. An intra-operative EGD confirmed no leaks. The surgeons performed open splenectomy, distal pancreatectomy, pseudocyst resection, partial gastric resection, and repair of the gastrotomy. The congested spleen weighed 580 g and measured 17.0 × 14.0 × 6.0 cm. Adherent blood clots could be seen near the splenic hilum. The patient was transfused 2 units of PRBCs and admitted to the surgical intensive care unit (SICU) for hemodynamic and pain monitoring. Anesthesia was initiated via the erector spinae plane (ESP) block and dilaudid patient-controlled analgesia (PCA) pump to optimize pain control. The patient was transitioned to oral pain medications and transferred out of the SICU.
Wound cultures from the PP grew Candida glabrata and Gram-positive rods, and the patient completed a course of Unasyn (3 g), Q6H, and Micafungin 100 mg, Q24H. The patient had no further complaints at discharge and required at least 1 year of penicillin prophylaxis. The infectious disease team recommended amoxicillin 500 mg twice daily. The patient will also require a meningococcal quadrivalent (ACWY) vaccine and meningococcal serogroup b vaccine 2 months after the first dose. Starting in 2028, these vaccines will need to be administered every 5 years. The patient’s 3-week follow-up at the surgical clinic showed good recovery. Once again, the patient was counseled on medication compliance, follow-up appointments, and animal handling.
Discussion
SPH is an extremely rare clinical entity characterized by elevated splenoportal pressure due to SVT7,8 or obstruction. 9 Unlike conventional portal hypertension, SPH presents with a patent-extrahepatic portal vein6,10 and normal liver function. This disorder clinically presents as hypersplenism, splenomegaly, or UGI hemorrhage.6,11,12 Splenoportal hypertension is commonly associated with pancreatic disorders such as acute pancreatitis, chronic pancreatitis, PPs, pancreatic neoplasms, and pancreatic surgery.7,8,10,11
Owing to its proximity to the pancreas, the spleen is prone to compressive effects from pancreatic tumors and pseudocysts, which can lead to SVT and splenomegaly. Likewise, recurrent pancreatitis may cause splenic vein stenosis resulting in SVT and ultimately SPH. PPs compress the splenic veins causing backflow of blood into the short gastric, gastroepiploic, colonic, and coronary veins.7,10,11 Left untreated, SPH can manifest as UGI bleeding, isolated gastric varices, or renal vein entrapment. 12 Esophageal varices are uncommon in patients with splenoportal hypertension because of the lack of hepatic involvement in SPH pathophysiology. 12
In most patients, SPH is asymptomatic; however, 10 it can manifest as nonspecific abdominal pain and can be misdiagnosed. Very rarely, SPH may present as massive UGI bleeding due to isolated gastric varices, 10 as seen in our patient. Additional signs and symptoms may depend on the primary pathology. Chen et al 6 recommend the routine use of abdominal ultrasonography (US) for the earlier detection of SPH in patients with pancreatic disorders, preventing GI bleeding and gastrohelcosis. No specific blood tests are available for the diagnosis of SPH or PPs. Nevertheless, blood work and pancreatic enzyme levels may help narrow the differential diagnoses. Canbak et al 7 diagnosed a case of pancreatic hydatid cyst-induced SPH due to eosinophilia noted on complete blood count. SVT can be detected using abdominal US and abdominal CT scan. Splenic angiography is preferred because of its higher sensitivity. 13
Symptomatic SPH is a medical emergency requiring urgent intervention. 4 Upper endoscopy is the first-line treatment for UGI bleeding and is often followed by surgical resection of the compressing mass. Splenectomy decreases the inflow of blood into the splenoportal system, 12 resulting in decongestion of the gastric veins. This intervention is preferred as it allows surgeons to concurrently fix the underlying pancreatic pathologies. 14 In our case, PP removal coupled with splenectomy led to the resolution of gastric varices and associated bleeding. Patients with asplenia are at risk of infections due to encapsulated microorganisms, such as Streptococcus pneumoniae. Postsplenectomy sepsis syndrome is a rare but fatal complication of splenectomy. 15 Therefore, patients with SPH are vaccinated before and after surgery. Our patient was vaccinated against encapsulated organisms according to the Centers for Disease Control and Prevention (CDC) guidelines.
Due to a high incidence of perisplenitis in SPH patients, splenectomy is associated with increased blood loss and long operation time.10,14,16 Image-guided interventions such as splenic vein stenting (SVS) and splenic artery embolization (SAE) have been employed in some cases of SPH with good outcomes. 14 First proposed by Poulin in 1993, SAE decreases the splenic volume leading to a better surgical view and negligible blood loss. 14 SAE can be used as a primary intervention for SPH or an adjunct to splenectomy to lower intra-operative blood loss and rates of conversion to open surgery.16,17 A lack of local expertise and high incidence of postembolization syndrome limits the wide adoption of SAE in clinical practice. 14 Postembolization syndrome presents as nausea, vomiting, or abdominal pain.
Splenic vein stenting has the lowest rate of rebleeding and visceral complications when compared to splenectomy and preoperative SAE.14,17,18 Covello et al 17 reported an interesting case of SPH-associated refractory chylous ascites that was successfully treated with transhepatic splenic stenting. Even though SVS has a 92% stent patency rates at 12 months, there are still no standardized guidelines for postprocedural anticoagulation. 17 Similarly, there is no consensus regarding the transjugular versus transhepatic approach of splenic vein stenting. 17
Footnotes
Author Contributions
LB and SO conceptualized the idea for this case report. AA, AM, and GM assisted with writing the manuscript. JE, YC, and WB edited, fact-checked, and proofread the final version of the case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require IRB approval/waiver for case reports.
Consent
The patient verbally consented for the publication of this case report.
Data Availability Statement
Further enquiries can be directed to the corresponding author
